Q: What mass of aluminum oxide can be produced from 100.0 g of aluminum given the reaction: 4Al (s) + ...
A: By using mole-mole analysis in balanced reaction mass of Al2O3 can be calculated.
Q: From the below reactions, indicate which product is the Markovnikov product and which product is the...
A: Markovnikov product The Reaction in which the negative part of the reagent (OH-) is get attached to...
Q: 1) Discuss the factors affecting the sign of AG in the relationship AG= AH - TAS. Give examples for ...
A:
Q: An ether has a lower boiling point than an aldehyde of the same mass (or number of C atoms). Why is...
A: An ether has a lower boiling point than an aldehyde of the same mass (or number of C atoms)
Q: Discuss and compare two possible chair conformations of cis-1,3-dibromocyclohexane compound. Which c...
A:
Q: Chemistry Question
A: Hydrolysis is the interaction of anion or cation of the salt with water to produce an acidic or basi...
Q: Precautions for filter paper
A: Filter paper: It is a paper that acts as a semi-permeable barrier for the mixture of solid and liqui...
Q: Which of the following compounds is SOLUBLE?A. SrSO4B. CaCO3C. BaSD. CaCl2
A: Among all the given compounds SrSO4, CaCO3, BaS and CaCl2, calcium chloride (CaCl2) is highly solubl...
Q: What is the mass of sodium iodide in 50.0 mL of 2.63 x 10 -2 M NaI( aq)?
A: Volume = 50.0 mL = 0.050 L Molarity = 2.63 × 10-2 M Mass of NaI = ?
Q: The overall reaction of the silver oxide battery, which is used in watches, may be written as Ag2O +...
A: Electrode in which reduction takes place is known as cathode and an electrode where oxidation takes ...
Q: 1) What does number of bonding electrons per atom (e/a ratio) means? How does it affect the crystal ...
A: Given: We have to tell how the number of bonding electrons per atom affect the crystal structure in ...
Q: 6. Given that the AH for CaO, H3PO4, Ca3(PO4)2 and H2O, are, respectively, -635, -1267, -4126, and –...
A: Answer (a): ∆H of the reaction is -545 KJ. Here negative sign indicate the release of energy or Heat...
Q: What is the pH of a 0.580 M solution of NaCN (Ka of HCN is 4.9 x 1010)?
A:
Q: What are the [H;O*] and pH of 0.085M weak base C2H5NH2?
A: Consider the solution of the given weak base C2H5NH2 (suppose B) as; As the given base is weak whic...
Q: This is a practice related question
A: Given: N2(g) + O2(g) ----> 2NO(g) ∆H = +180kJ ----(1) 1/2 N2(g) + O2(g) ----> NO2(g) ∆...
Q:
A: Given combustion reaction is : C2H4 (g) + 3O2 (g) --------> 2CO2 (g) + 2H2O (l) Given, Standar...
Q: Acid catalyzed dehydration reaction of 2-methyl-1-butanol produces 2-methyl-2-butene as the major pr...
A: The question is based on the concept of organic reactions. we have to explain the reason of format...
Q: 1) What does number of bonding electrons per atom (e/a ratio) means? How does it affect the crystal ...
A: Given: We have to tell how the number of bonding electrons per atom affect the crystal structure in ...
Q: Draw TWO (2) structural isomers of bromobutane
A:
Q: Calculate the number of moles of HCl in 1.0 mL of a 0.10 M HCl solution
A:
Q: Discuss the factors affecting the sign of ∆G in the relationship ∆G= ∆H - T∆S. Give examples for eac...
A: Negative free energy change accompanies spontaneity of the reaction or process.
Q: indicate if the temperature increase or decrease in the following matter changes: from liquid to gas...
A: 1. from liquid to gas 2. from liquid to solid 3. from gas to liquid 4. from solid to liquid
Q: 9) Adding 1g of benzene, C,H6, to 70g cyclohexane, C6H12, lowers the freezing point of the cyclohexa...
A:
Q: Discuss the factors affecting the sign of AG in the relationship AG= AH - TAS. Give examples for eac...
A: ΔG = ΔH - TΔS G = Gibbs free energy H = enthalpy T = Temperature S = Entropy.
Q: Which of the following does NOT have a carbonyl group? Group of answer choices alcohol aldehyd...
A: Functional group are the atoms or group of atoms which represent the properties of a molecule, diff...
Q: Calculate the maximum expected vapor pressure lowering of a solution made by dissolving 109 grams of...
A:
Q: What are the correct coordination number of diacetyacetonato zinc(II) and dichloro(1,10-phenanthroli...
A: By making structure of these complex compounds we can determine its coordination number.
Q: 3) Explain which is the stronger acid a)HNO3 or HClO4 b) CH2FCOOH and CH2BrCOOH
A: To solve this first we have to know the acid strength which depends upon the release of H+ ion . Th...
Q: Given the following reaction: 2 C2H6(l) + 7 O2(g) → 4 CO2(g) + 6 H2O(l) How many moles of water are ...
A: The given reaction: 2C2H6(l) + 7O2(g) → 4CO2(g) + 6H2O(l) The given number of moles of oxygen = 3.0 ...
Q: Write the oxidation half-reaction and reduction half-reaction, and shorthand electrochemical notatio...
A: Oxidation is process involves loss of electrons. Reduction is a process which involves gain of elect...
Q: Which of the following organic Compounds you think is most readily soluble in water? Explain (a) Tol...
A: Given Compound = Toluene , Oxalic Acid , Benzaldehyde Most soluble in water = ?
Q: An analytical chemist is titrating 56.7 mL of a 0.4900M solution of formic acid (H,CO,) with a 0.750...
A:
Q: How will an organic compound with neutral ph behave on a litmus test.
A: Explanation- The main use of litmus is to test whether a solution is Acidic or Basic. Light Blue li...
Q: The vapor pressure of Substance X is measured at several temperatures: temperature vapor pressure - ...
A: We have different vapour pressure at different temperatures , we wish to calculate the enthalpy of v...
Q: Write a complete IUPAC name for each of the following compounds
A: In this question, the IUPAC name of the provided compounds has to be determined. (i) IUPAC Name: 3,...
Q: Caloulate tOH] at 25° C solutionwidh for a conentrahion of TH30*] equal to 4.5x 10°3 M and a a deter...
A: pH is defined as the negetive logarithm of hydrogen ion (H+/H3O+) concentration. So, pH = -log [H3O+...
Q: Chemistry Question
A:
Q: 10 Discuss the aromaticity of the below compounds? Explain which compound is aromatic, why?
A:
Q: Rb2CO3 (s) + 2 HCe (aq) → 2 RbCe (aq) + H2O (e) + CO2 (g) When 69.3 g Rb,CO3 is allowed to react wit...
A:
Q: Metals and non metals
A:
Q: Discuss the factors affecting the sign of AG in the relationship AG= AH - TAS. Give examples for eac...
A: ∆G can be either positive or negative. A reaction is spontaneous when ∆G is negative and not spont...
Q: How many grams of C,H, must decompose according to the following chemical
A: C6H6 (l) --> 3 C2H2 (g) heat of reaction = 630 KJ 1 mol of benzene ( 78.12...
Q: (d) Arrange the following compounds in increasing order of basicity. NH2 H CH "NH2 H H3C C D E
A:
Q:
A: In first question we have the reduction of carboxylic acid with lithium aluminium hydride followed b...
Q: A fabric reiiant from a burial site has a 1"C/?C ratio of 0.735 of the original value. How old is th...
A: As per regulations, only the first question can be answered.
Q: Chemistry Question
A: Dear student I have given answer to your question in the image format.
Q: Calculate the solubility of silver chloride (AgCl) in g/L. (Molar Solubility)
A: We have to find solubility of silver chloride (AgCl) We know solubility product of AgCl = 1.12×...
Q: Is the following reaction endothermic or exothermic? Reversing the reaction (1) , we get, 2NO(g) ---...
A: The energy difference of the products and reactants is known as the energy change of a reaction. If ...
Q: Common Ion Effect and Solubility Calculate the solubility in g/L of AgBr in 0.0010 M NaBr.
A: Since NaBr is completely soluble in water, it is dissociated into Na+(aq) and Br-(aq). [Br-(aq)] = ...
Q:
A: ∆ G = -nF Eocell

Step by step
Solved in 2 steps with 2 images

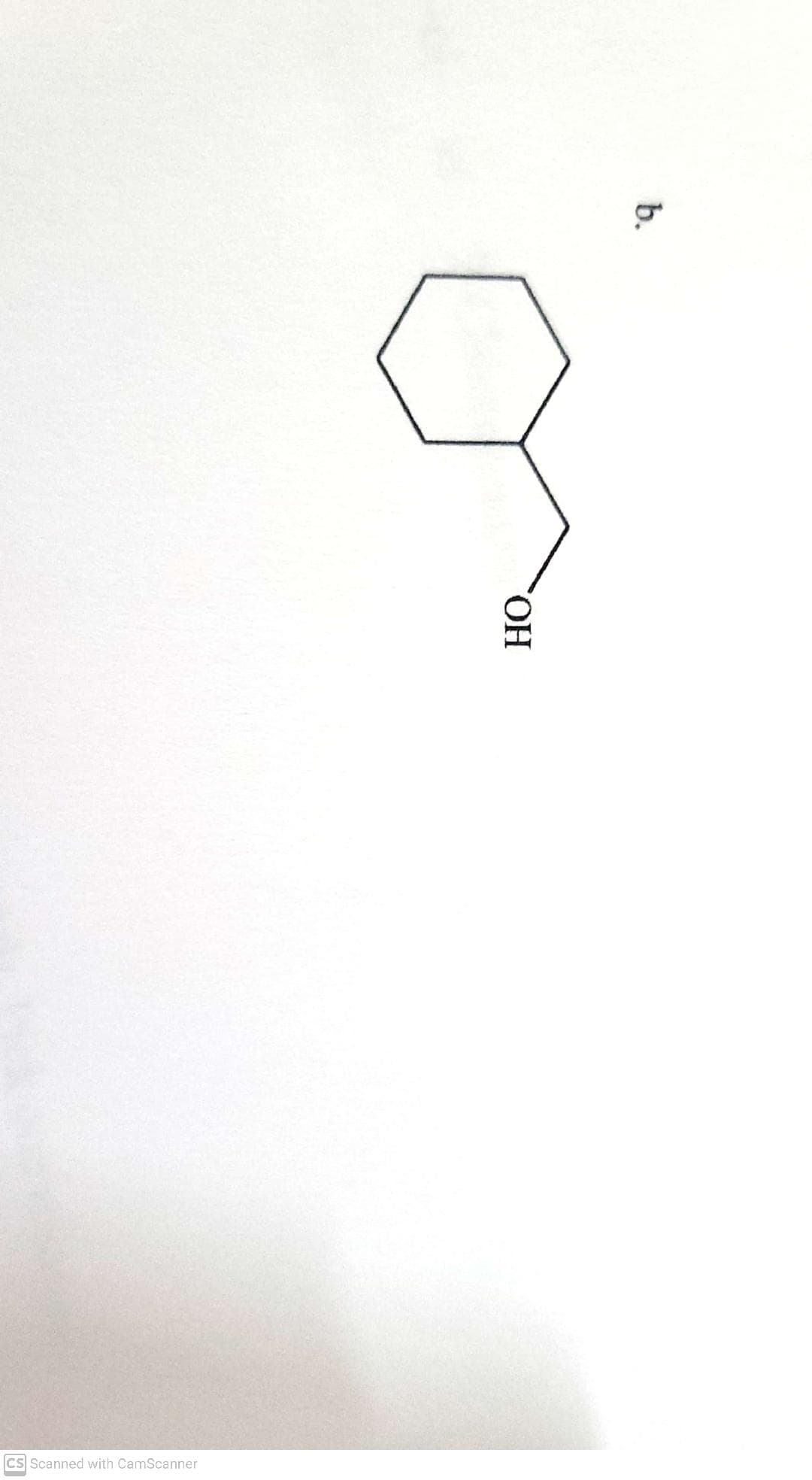
- Propose how you could synthesize the following compound, using a substitution reaction. Propose a nucleophile, a solvent, and an electrophile. Show the curved-arrow mechanism of the reaction. Finally, draw the transition state of the reaction.Propose a detailed mechanism for the reaction below (in the attached picture), showing the structure of thestable intermediate and using curved arrows to indicate electron flow in each stepWhat is the mechanism for the entire reaction below?
- Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpBelow is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?Several reagents and several organic structures are shown below. Construct a multistep synthetic route from the reactant 2-methyl-1-butene to the product 3-bromo-2-methyl-2-butanol by dragging the appropriate pieces into the bins. Note that each bin will hold only one item, and not every given reagent or structure will be used.



