Q: но 1 eq. MeOH LOME cat. HCI
A: Carbonyl compound reacts with alcohol to form hemiacetal and acetal. The reaction is reversible and…
Q: trongacid, strong base, weak acid, weak base, organicwhich is neither
A:
Q: Which statement is correct regarding the acid-base equilibrium system below? OH HN. CH HC I.…
A: In an acid base reaction the equilibrium lies in the side having weaker acid.
Q: Part A Arrange the solutions in order of increasing acidity. Rank solutions from least acidity to…
A:
Q: What is the conjugate base of H;PO,?
A: A conjugate base is that which is formed when a acid releases a H+ ion in the solution And a…
Q: The hydride ion, H-, is a stronger base than the hydroxide ion, OH-. What is/are the product(s) of…
A: When Hydride ion H- reacts with the water, it gives following reaction. H-(aq) + H2O(l) → H2(g) +…
Q: Which is a stronger base, PO4 3- or AsO4 3-? Explain.
A: An acid is a substance that gives H+ ions in its solution whereas a basic substance gives OH- ions…
Q: Which statement is correct regarding the acid-base equilibrium system below? CH . OH HN I.…
A: Answer III spontaneous in reverse direction because H3C-CH2-NH3+…
Q: Write the reversible reactions for CH3NH2 with water and identify each species as acid (A), base…
A: Interpretation- To write reversible reaction between CH3NH2 and water and identify the species as…
Q: Which of the following statements is correct about the reaction below? HAsO42- (aq) + H2O (liq) →…
A: To solve this problem we will use the Bronstead-Lowry acid-base theory .
Q: d. Write an equation for the reaction of each acid with NaHC03.
A: NaHCO3 is a basic salt ,it has pH value above 7. Thus it reacts with acid and form salt and water…
Q: CHCI,COOH CH,CH,COOH CH;CICOOH CH3COOH Strongest acid Weakest acid
A:
Q: Arrange the following in order of increasing acid strength, starting with the least acidic:…
A: Two major factors that influences acidity are resonance effect and -I Inductive effect. Acidity…
Q: Acid A - ka is 1.2 x 1015 Acid B - ka is 2.3 x 10 15 Which is a stronger acid?
A: NOTE: Since you've posted multiple questions, we'll solve only the first question for you. If you…
Q: Which of the following is properly identified? a. NH3, strong base b. H2CO3, weak base c. HNO3,…
A: Acidic strength of various acids can be identified by using their pKa value. Acidic strength is…
Q: which is the weaker acid H2SO4 or HNO3
A: The strength of an acid is determined by the dissociation constant. More negative or smaller…
Q: Complete the balanced chemical equation for the following reaction between a weak base and a strong…
A:
Q: Most Acidic H,PO, H,PO, HPO,? H,O+ H,0 Least Acidic
A: Given that : We have to rank these Bronsted acids according to their acidic character.
Q: 22. The conjugate base of CH3COOH is: A) H3O* B) CH3COO" C) OH D) H2O 23. The conjugate acid to NH3…
A: The correct answer about acid Base system is given below
Q: 22. The conjugate base of CH3COOH is: A) H3O* B) CH3COO C) OH D) H2O 23. The conjugate acid to NH3…
A: The correct answer is given below
Q: In this equation H2O +HF- H3O+ +F-1 H20 is a base and HF is its conjugated acid HF is an acid and F-…
A: Acid is the species which gives H+ ion and after giving H+ ion the species formed is its conjugate…
Q: HO. CI. ОН H3C HO. HO, ОН F F нн нн
A: The given molecules either contain halogens or OH groups at the alpha carbon to the COOH…
Q: Calculate [OH-] given [H3O+] = 4.0 * 10^-9 M
A: An acidic substance is the substance that can give H+ ions. They are corrosive and sour in nature.…
Q: Based on their relative acid strength, is the given comparison correct? HPO42- > H2PO4- O True False
A: An acid is a substance which gives H+ ions in aqueous solution. More number of H+ more will be the…
Q: Indicate the conjugate acid of CH3NHCH3. a. CH3NNCH3 b. CH3N-CH3 c. CH3N+H2CH3 d.…
A: When base takes H+ , then conjugate acid is formed As per our company guideline we can answer only…
Q: Highest acidity Lowest acidity
A: Higher the stability of the conjugate base formed by the removal of proton(H+), higher would be the…
Q: Complete the balanced molecular reaction for the following weak base with a strong acid: NaClO2(aq)…
A: Since we know that when sodium chlorite react with sulfuric acid to produce sodium sulfate, chloric…
Q: 1. MeO in MeOH 2. HCI, H2O
A: Given reaction is shown below:
Q: Which comparison is correct? O NH3 is a stronger acid than HF. O PH3 is a stronger acid than NH3. O…
A: The comparisons given are,
Q: c. N2 H5 * (aq): Oacid Obase conjugate acid conjugate base d. SO. (aq) acid base conjugate acid
A:
Q: Illustrate which is the stronger acid using Lewis structures: а. HNO; or HCI4 b. CH2FCOOH or…
A:
Q: What are the products of the reaction between nitric acid [HN03(aq)] and sodium hydroxide…
A: The reaction of a strong acid with a strong base gives salt and water. This type of reaction is…
Q: Explain which is the stronger acid a)HNO; or HC104 b) CH;FCOOH and CH2BICOOH
A: When two or more acids are compared, then one of them more Acidic while other not. It may be due to…
Q: Idonti fy the Opid, base, Conjugate acid and conjugaic base in The follownnej reachon. HPO t Azo- >…
A: The acid base Reaction given in the question is - HPO42- + H2O ---> OH- + H2PO4- According…
Q: The conjugate acid of HPO,-2 i , H2PO4 А. + O B. H2PO4* Oc. H2PO4-3 O D, PO4-3 O E. H2PO4
A: Conjugate acid-base pair: The acid-base pair that differ by a proton is known as conjugate acid-base…
Q: Between HNO3 or HNO2 which is the stronger acid? Type in formula exactly. C.
A: Comparison the acidic strength of HNO3 and HNO2 =?
Q: = relative acidity
A: The resonance effect is more effective than the inductive effect.
Q: Arrange the follwoing in order of decreasing basicity. OH-, CH3COOH, CH3CH2-, NH2-, NH3, Cl-
A: Basicity: Tendency to donate electron pair or simply bases are those that abstracts protons. Greater…
Q: Alcohols can act either as weak acid or as weak base, just as water can. Show the reaction of…
A: Alcohols can act either as weak acid or as weak base, just as water can.
Q: HO. .CI NH HO НО НО HO HO A В C D
A: Given compounds are : Arrange in order of increasing acidity. Explain.
Q: Based on their relative acid strength, is the given comparison correct? HNO3 > HNO2
A: Strength of an acid depends on how many protons are dissociated and form hydrogen ions in…
Q: Which statement is incorrect? I. H2O П. ОН III. H3O* IV. O- A. II is a conjugate acid of IV C. I is…
A: Acid-base pairs that differ by one H+ unit are called conjugate acid-base pairs. Acid gives H+ to…
Q: what is the conjugate base and conjugate acid products with formal charges?
A:
Q: Arrange the following according to decreasing acid strength * Br F CH3-CH-COOH (2) BRCH2CH,COOH…
A: We have to predict the correct order for acidic strength.
Q: 6. Which of the following statements is true about the following acid/base reaction? F (aq) + HSO4…
A: pH is measure of acidity or basicity of a solution.. pH less than 7 indicate solution is acidic. pH…
Q: The common constituent in all acid solutions is O H2 H+ O OH- O H2SO4 O C-
A: When acid is dissolved it form H+ ion
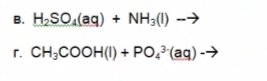
Step by step
Solved in 2 steps

- Consider the reaction below: Pb(NO3)4(aq) + 4 NaOH(aq) ----> _ Compound A + _ Compound B (OR Pb(N O3)4(a q) + "4" N a O H(a q) ----> Compound A + Compound B) One of the the single ions that would be used to form compound A or compound B would be ____.What volume of 0.171 M KOH,0.171 M KOH, ?,V, can be neutralized with 58.5 mL58.5 mL of 1.21 M HNO3? V=What is the reaction that represents ΔHf° for Al2O3 (s)?
- A 10.0 mL sample of household ammonia solution required 25.10 mL of 0.245 M HNO3 for neutralization. Calculate (a) the molar concentration of the ammonia and (b) the mass/mass % concentration of ammonia(17.04 d/mol), given a solution density of 0.985 f/mL. HNO3(aq) +NH3(aq) ---> NH4NO3(aq)A)When working in the lab, you are measuring the pH of a system. The pH meter reads 4.34. What is the concentration of [H3O+] in solution? B)You have created a solution of NaOH by dissolving NaOH in water. You determine that the [OH-] concentration in solution is 4.37 x 10-9. What is the pOH of the solution? C)What is the pH of a solution containing an [H3O+] = 9.67 x 10-3?What volume of 0.441 M KOH,0.441 M KOH, ?,V, can be neutralized with 50.0 mL50.0 mL of 1.64 M HNO3? ?= mL
- Given the mole ratio of HCl to NaOH is 1:1 in the balanced acid base reaction A 15.52mL sample of HCl is titrated to endpoint witg a 32.08mL sample of NaOH, which has a known molarity of 0.976M. How many moles of NaOH were used in this reaction?Phosphoric acid, H3PO4 (aq), is found in soda. Write the balanced chemical equation for the neutralization of phosphoric acid with NaOH.What volume of 0.195 M KOH,?, can be neutralized with 57.0 mL of 1.15 M HNO3? (in mL)

