Chapter17: Complexation And Precipitation Reactions And Titrations
Section: Chapter Questions
Problem 17.23QAP
Related questions
Question
9b

Transcribed Image Text:9. The % w/w of I- in a 0.2144-g sample was determined by Volhard
titration. After adding 50.00 mL of 0.04980 M AGNO3 and allowing the
precipitate to form, the remaining silver was back titrated with 0.05322 M
KSCN, requiring 22.76 mL to reach the end point. (MM of I = 126.904
g/mol)
Reactions:
Ag* +I-→ Agl
Ag* + SCN" → Ag(SCN)
Calculate the:
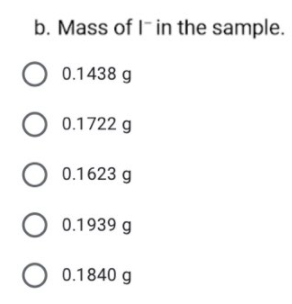
Transcribed Image Text:b. Mass of I- in the sample.
O 0.1438 g
O 0.1722 g
O 0.1623 g
O 0.1939 g
O 0.1840 g
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

