Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 75AP: Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring...
Related questions
Question
I have attempted most of it and I was wondering if youd be able to verify my answers and help me with the last one. Thank you!
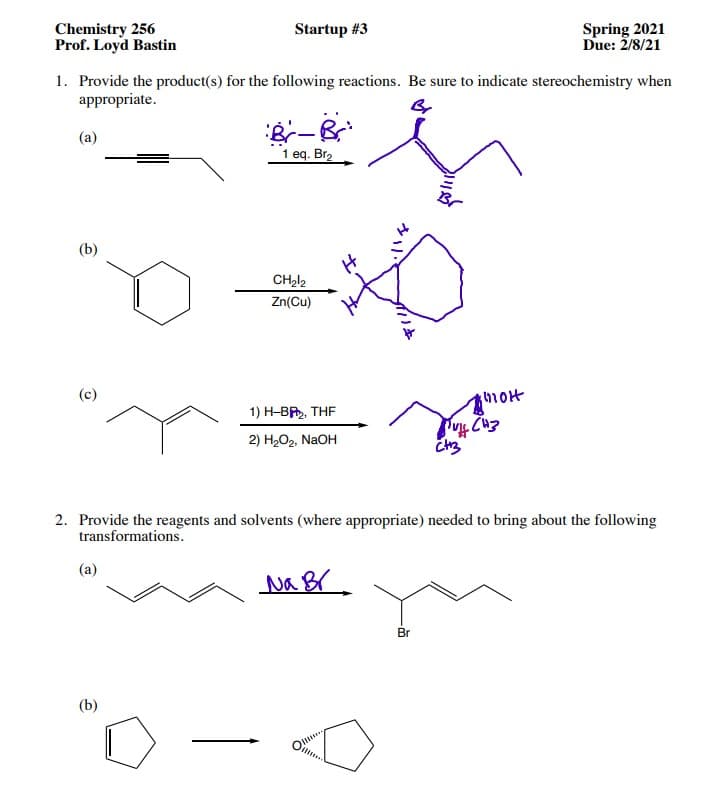
Transcribed Image Text:Chemistry 256
Prof. Loyd Bastin
Startup #3
Spring 2021
Due: 2/8/21
1. Provide the product(s) for the following reactions. Be sure to indicate stereochemistry when
appropriate.
(a)
1 eq. Br2
(b)
Zn(Cu)
(c)
hot
1) H-BR, THE
2) H-О2, NaOH
2. Provide the reagents and solvents (where appropriate) needed to bring about the following
transformations.
(a)
Br
(b)
Expert Solution
Step 1
Since you have posted multiple questions, we can only answer the last one as mentioned by you.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning