Q: Balance the following net ionic equation for redox reaction in basic solution: MnO4-(aq) + NO2-(aq)…
A:
Q: Balance with the half-reaction method in a basic, aqueous environment: CrO2(s) + NaClO (aq) → K¿CrO,…
A:
Q: Balance the following oxidation–reduction reaction in basic solution. MnO4 –(aq) + I–(aq) -->…
A:
Q: Balance the reaction and find the reducing agent and oxidizing agent. NHO3+H3AsO3 -->…
A: We will try to balance the reaction by hit and trial method.
Q: 2Fe + 3CI2 → 2FECI3 Half equation of oxidation Half equation of reduction reaction reaction lonic…
A: Redox reaction:Redox reaction is a type of chemical reaction in which the oxidation states of atoms…
Q: Balance the following reaction under acidic conditions: Mn2+(aq) + BiO3–(s) → Bi3+(aq) + MnO4− (aq)
A:
Q: Balance the redox equations. a. Br2 + H2O -> BrO3- + Br- + H+ b. I- + O2 + H2O -> H2O2 + IO3-
A: A redox reaction is defined as the reaction in which oxidation and reduction take place…
Q: Balance this reaction equation using the lowest possible whole numbers. Be sure to include any '1's…
A: The given reaction can be balanced as follows:
Q: Balance the following oxidation-reduction reactions, which occur in basic solution, using the…
A: Oxidation reaction is defined as the reaction in which a chemical specie loses electron. Reduction…
Q: balance the equation below. Write the ionic and net ionic equations. Finally, write the balanced…
A: In the above Redox reaction Nitrogen is undergoing reduction in which oxidation state of nitrogen…
Q: BALANCING OXIDATION/REDUCTION REACTIONS Cr3+(aq) + Cl-(aq) →Cr (s) + Cl2(g)
A: Redox reaction Oxidation reaction occurs when there is a loss of electron while a reduction reaction…
Q: Balance the following half-reaction in an acidic solution. Cr2O72- → Cr3+
A: Half reactions are balanced using the following steps- Balance atoms other than hydrogen and oxygen…
Q: Balance the following reactions by the half-reaction method in acidic conditions (include phases at…
A: The given unbalanced equation is: Br2(aq) + SO42-(aq) -----> BrO2-(aq) + SO2(g) (Acidic…
Q: Balance the following oxidation - reduction reaction. Identify the oxidizing and the reducing agent.…
A: Oxidation/reducing agent means adding of oxygen and reduction /oxidising agent means removal of…
Q: Zn+H2SO4=ZnSO4+H2. here you need to do a redox reaction I was given a chemical reaction (I sent it…
A: Given, the Redox reaction: Zn + H2SO4→ ZnSO4 + H2
Q: (b) MnO4 Element Oxidation Number Mn i i
A:
Q: Balance the following oxidation-reduction reaction by the half-reaction method. What is the sum of…
A: In the above reaction, Br was in +1 state initially and finally in +7 and S was in +4 initially and…
Q: When dilute nitric acid is poured on a price of copper metal, copper(II) ions and the nitric oxide…
A: Redox reaction is the chemical reaction in which oxidation – reduction occurs. Oxidation and…
Q: Balance the reaction: Mn2+ + H2O2 à MnO2 + H2O (redox)
A: Using redox method to balancing chemical reaction.
Q: Balance the redox equation: A NO3- + B Cu --> C NO + D Cu2+
A: Redox reactions are those in which oxidation and reduction take places simultaneously. Oxidation is…
Q: Mg + 2HCI H2 + MgCl2 Half equation of oxidation Half equation of reduction reaction reaction lonic…
A:
Q: In setting up this experiment, a student noticed that a bubble of air leaked into the graduated…
A: When gas bubbles leaked into the cylinder that shows there is change in the volume of measured…
Q: Complete and balance the following half-reaction in acidic solution ClO3- (aq) -> Cl- (aq)
A: ..
Q: Balance the following oxidation reduction reactions: CIO, + MnO2 --> Cl + MnO4" BrO4 + Br --> Br2
A:
Q: Balauce n 7 steps Oxidation - Reduction MNOLT t Noz-7 MNOZ+ N03 > MNOZ+ NO3
A:
Q: Balance the following reaction under acidic conditions: Cl-1(aq) + MnO4-1(aq) to ClO3 -1 (aq) +…
A: A balanced reaction contains equal number of each atom of each element present on either side of the…
Q: Balance the following redox reactions by the oxidation number method and determine the oxidizing and…
A: A redox reaction can be defined as a reaction in which the oxidation (loss of electrons) and…
Q: Balance Redox Reaction:
A: A chemical formula is a short term notation of a molecule. In a chemical formula the respective…
Q: Balance this reaction:_C«Hs + O:- H:O + CO: * - - -
A: A balanced chemical equation is that which consists of an equal number of atoms on either side of…
Q: Balance the following half reaction in acidic solution. CrO3 --> Cr2+
A:
Q: Balance by half reaction: NO2 + OH- --> NO3- +NO2- + H2O
A: We have to balance the given reaction
Q: Complete and balance the chemical reaction using method of half-reactions. Write your answer in your…
A: The given reaction is - Als + NO2-aq ⇔ AlOH4-aq + NH3aq Oxidation is the loss…
Q: The following skeletal oxidation-reduction reaction occurs under basic conditions. Write the…
A:
Q: Balance the oxidation-reduction reactions that occur in basic solution using the half-reaction…
A:
Q: Balance each aqueous reduction/oxidation reaction.
A:
Q: Balance the following equation under both acidic and basic conditions. Br2 + SO2 --> Br…
A: To Balance the given reaction in acidic and basic medium. Br2 + SO2 --> Br- + SO42-
Q: 5. N2H4 + Cu² → N2 + Cu Oxidation half-reaction: Reduction half-reaction: Complete balanced…
A: Oxidation means loss of electrons and reduction means gain of electrons.
Q: Balance the following reaction using the oxidation state method. Then give the coefficient of sulfur…
A: Using concept of balancing chemical reaction.
Q: Balance the equations according to the half-reaction method. Zn(s) + NO3−(aq) → Zn2+(aq) + N2(g)…
A:
Q: Balance the following half-reaction in an acidic solution: Cr2O72- --> Cr3+
A: The given half cell reaction is as follows: Cr2O72- → Cr3+
Q: Balance the following reaction using the oxidation state method. Then give the coefficient of sulfur…
A: Given that, ___ SO2 + ___ MnO4– + ___ H2O --> ___ SO42– + ___ Mn2+ + ___…
Q: Balance the following equations using the half reaction method. Balance the first equation in an…
A: Balanced the reactio under the given condition--
Q: write balance oxidation reduction reaction 3+ 2+ Fe Mn
A: Given reaction MnO4-aq+Fe2+ (aq)→H+Fe3+ (aq)+Mn2+aq Above reaction occurs in acidic medium ( see the…
Q: Balance the oxidation reduction in an acid solution: ClO4- + S2O3^2- —-> cl- + so4 ^2-
A:
Q: Balance this equation, MnO2(s) + Cl-(aq) —> Mn2+(aq) + Cl2(g) using the half-reaction method.
A:
Q: Balance the following equations, using either the half-reaction method or the oxidation number…
A: Given balance the redox reactions
Q: (d) MnO2 Element Oxidation Number Mn i i
A: Given molecule is MnO2 .
Q: Balance the following equation in basic solution: CN-+ Mn04¯ CNO- + Mn0
A: Since you asked multiple questions so as per Q&A guidelines of portal I solve first question…
Q: Balance this equation Mn²+ (aq) + O2(g) MnO, (s) in basic solution.
A: Balancing the reaction equation: Equalize the no. of same atoms on either side of the equation by…
Q: ions. Write down unbalanced half reaction equations
A: We have to Write down unbalanced half reaction equations and a balanced overall equation for the…
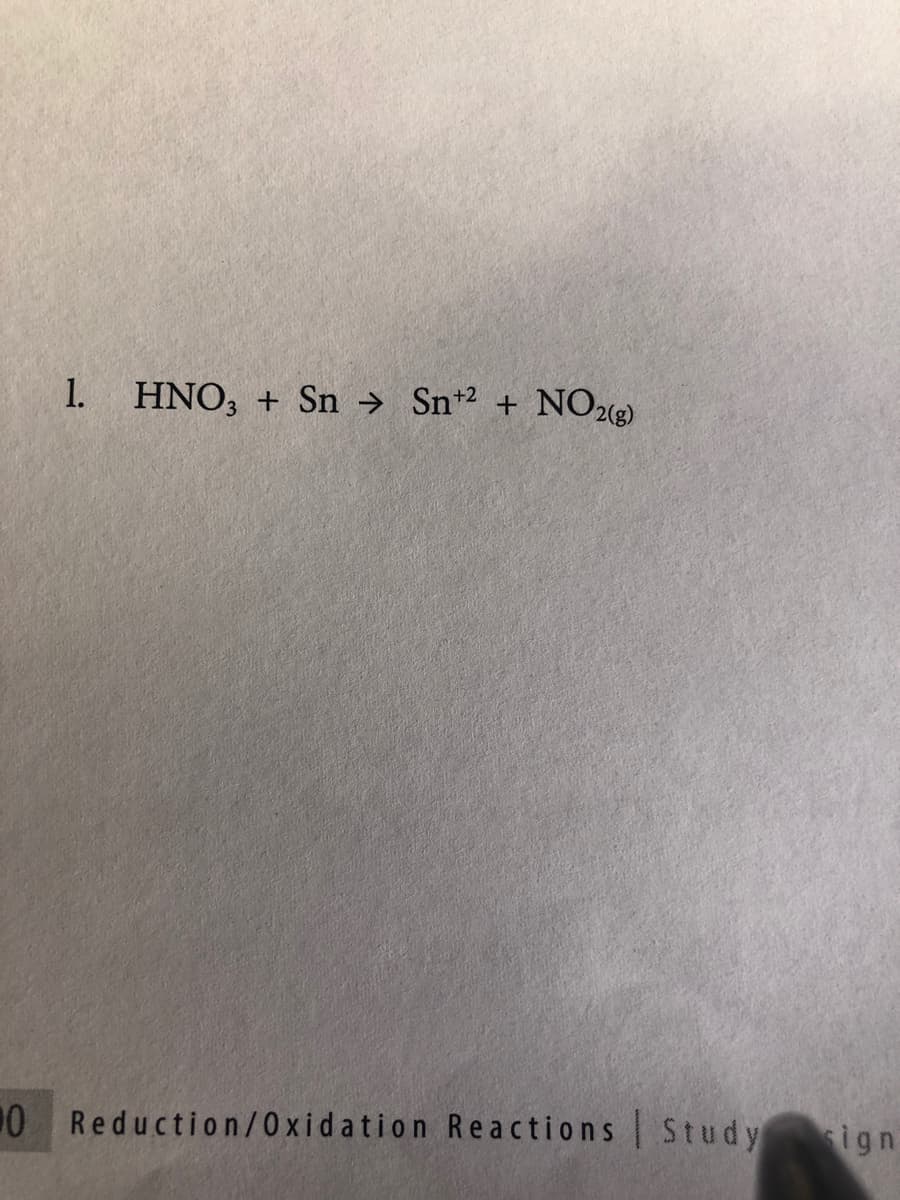
Step by step
Solved in 2 steps with 2 images

- In the reaction Pb(s) + 2Ag1(aq) -----> Pb21(aq) + 2Ag(s) Which species is oxidized and which is reduced?I am struggling to get this question right and would really appreciate any help. Pls make sure you’re anser is corrrecf 100% pls and thank you !loss of electrony -2 in the reaction: CuSO4 (aq) + Mg(s) →MgSO4 (aq) + Cu(s)? privollot o B) Cu (s) C) Mg (s) D) CUSO4 (aq) 3) What species is oxidized A) MgSO4 (aq) Jl reaction?

