Q: Definition of freezing point depression Use Freezing point depression concept to explain (including…
A: 1. Freezing-point depression is basically drop of temperature at which a substance freezes, caused…
Q: SO₂(g) + Cl₂(g) = SO₂Cl₂(g) ΔΗ° = −264 kJ K = 34.5 At 300 K, a set of equilibrium concentrations are…
A: Here we are required to find the equilibrium concentration of SO2Cl2
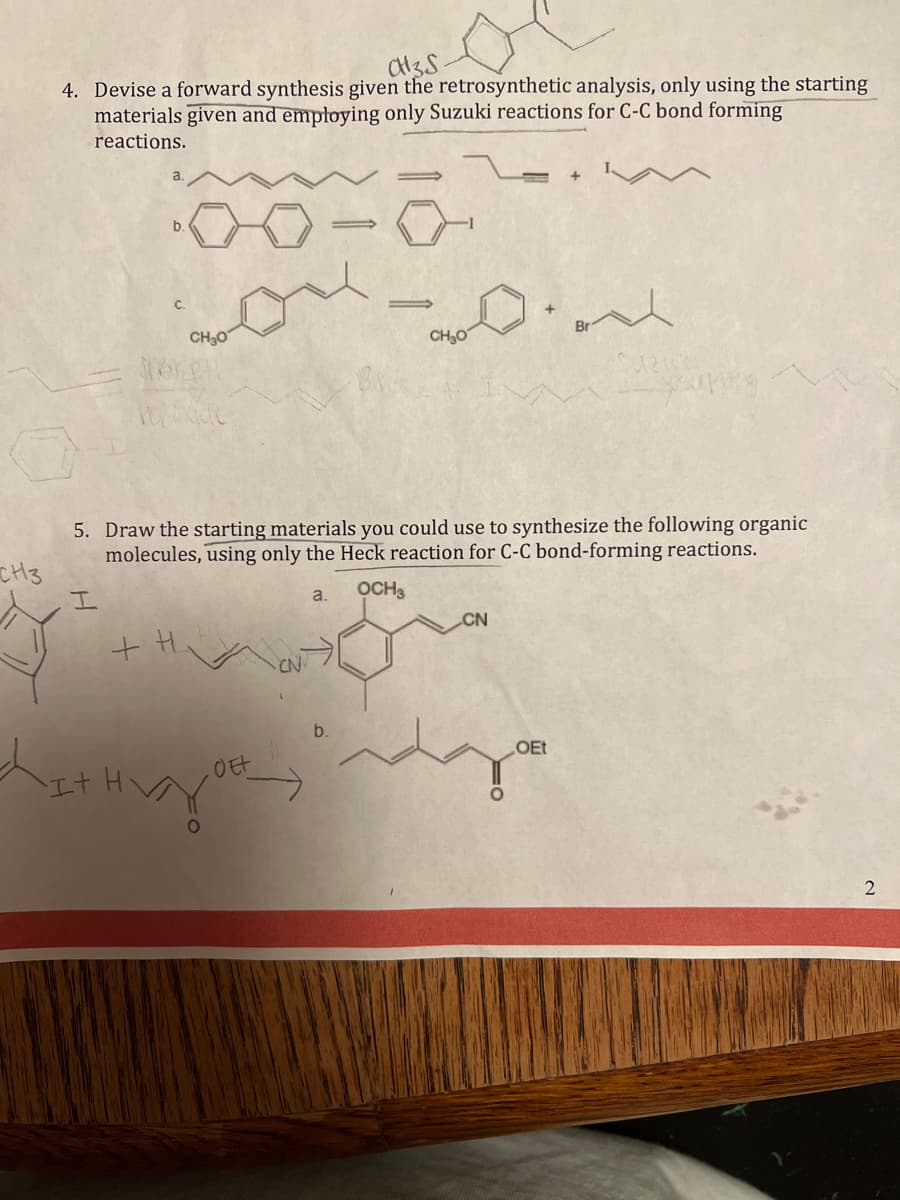
Q: 6. Provide the missing starting materials, reagents, or products, as needed. If you are supplying…
A: Requirement from question: Total eight questions and answers. Major organic products.
Q: H₂O(g) + Cl₂O(g) ⇒ 2HOCI(g) K = 0.0900 @ 298 K The sealed 2.00 L reaction vessel was charged with…
A: Dear student , since you have posted multiple parts questions we are allowed to solve only first…
Q: Circle the molecule in each pair that is more water-soluble. 1 (b) (c) VS. VS. VS. M (e) vs. (f) ï ï…
A: We have to select the more soluble compound
Q: 6. Draw all of the stereoisomers that you would form in the reaction of CHCl3 and KOTBU given the…
A:
Q: A 0.0853 mol sample of an unknown gas contained in a 3.00 L flask is found to have a density of 1.37…
A: Given data : 1. No. Of mole = 0.0853 mol 2. Volume = 3.00 L 3. Density = 1.37 g/L To calculate :…
Q: Calculate either [H,O] or [OH-] for each of the solutions at 25 °C. Solution A: [OH-] = 2.59 x 107…
A:
Q: We start with a pure sample of A(g). The following equilibrium is established: 2 A(g) B(g) + C(g)…
A:
Q: has K, -2.3 x 109. If 0.065 mol NH, is placed in a 1.31 L container, what will the concentrations of…
A:
Q: We have a container enclosing a mixture of N2(g) and O2(g). The total pressure is 4.12 atm. The…
A:
Q: Which molecular formula corresponds to a straight-chain alkane?
A:
Q: Among the butane conformers, which occur at energy maxima on a graph of potential energy versus…
A:
Q: It is desired to inflate a baggie with a volume of 993 milliliters by filling it with neon gas at a…
A: Given Data :
Q: Some chemical compounds are listed in the first column of the table below. Each compound is soluble…
A: Given, BaOH2 , C2H5NH2 , KI and NaOHRequired, Type of compound
Q: What is the major product of th 1)CH₂M 2) IL-O CHL
A:
Q: I need to know what the last 3 sub parts are if their acid base or not
A:
Q: Question 28 Natural water sources can become polluted with toxic metal ions such as Hg as part of…
A: The above question based on the removal toxic of Heavy metal ions from water by precipitation…
Q: Light is emitted from a flame by elements when electrons move from the state to the state.
A:
Q: Directions: Balance the following redox reactions using the change in oxidation number method. a.…
A:
Q: bel the three bold hydrogens in compounds A, B, and C in order of acidity, with 1 being the most c…
A: Acidity can be defined as tendency of a compound to lose hydrogen ion.
Q: A 355 mL container holds 0.146 g of Ne and an unknown amount of Ar at 35°C and a total pressure of…
A:
Q: How many valence electrons are in an element belonging to period 2, group 16? Paragraph v B I U A Ev…
A:
Q: Which of the following statements is true for real gases? Choose all that apply. As attractive…
A: Statements that is true for real gas are as follow.
Q: Calculate the energy required to heat 1.10 kg of water from 49.1 °C to 62.6 °C. Assume the specific…
A:
Q: The following data were obtained from a study of the decomposition of a sample of HI on the surface…
A: A question based on kinetics of reaction in solution that is to be accomplished.
Q: Question 41 of 48 What is the relationship between the two molecules shown below? A) They are…
A:
Q: The decomposition reaction of carbon disulfide, CS2, to carbon monosulfide, CS, and sulfur is…
A: Chemical kinetics is branch of chemistry in which we deal speed at which rate of reaction occurs.
Q: 3. Si + 2F₂ SiF4 4. H₂ + Cl₂ 2HC1 Oxidation Reaction: Reduction Reaction: Reducing Agent: Oxidizing…
A:
Q: Which of the following statements is true for ideal gases, but is not always true for real gases?…
A:
Q: Identify the Newman projection that depicts the eclipsed conformation of butane.
A:
Q: me of the sample increase, decrease, or stay the same? e volume increases or decreases, calculate…
A:
Q: lowing gases in order of increasing average molecular speed a 6, Xe, CCl₂ F2. < < <
A:
Q: Label the following as cis or trans or neither. H H H L Br Br Br CH₂ H CH₂CH₂ -H H Br
A: Incase, disubstituted groups ( But not 1,1-disubstituted) are same side then compound is cis whereas…
Q: An aqueous solution of KCI (molar mass 74.5 g/mol) has a concentration of 1.85 m. If this solution…
A:
Q: 13. The molar conductivity of 0.0300 M HF (aq) is 4.10 mS m² mol. Determine the acidity constant,…
A:
Q: An unknown gas at 73.1 ∘C and 1.05 atm has a molar mass of 44.10 g/mol. Assuming ideal behavior,…
A: Ideal gas equation is the equation which obeys all gas laws under all condition of temperature and…
Q: Basic unit of matter
A: An atom is the basic unit of matter.
Q: opy, mm. The colored 100 The absorbance falls to 0.229 at 35.4 min. Calculate the rate constant in…
A: Given data set: Extinction coefficient = 5100 cm-1 M-1 Wavelength= 520 nm Path length = 1.17 cm…
Q: The rate constant for the second-order reaction 2NOBr(g) → 2NO(g) + Br2(g) is 0.80 M-¹s1 at 10 °C.…
A:
Q: below each structure, write the hybridization state (sp, sp, or sp') =. ALL atoms and lone pair…
A: Hybridization is the overlapping of the atomic orbitals and forms same energy hybrid orbitals.
Q: Provide the correct IUPAC name for the skeletal (line-bond) structure shown here. fr 4- 2,4,4- 2- di…
A:
Q: Draw a line formula corresponding to the following condensed structural form…
A: The line formula of the given condensed structural formula is:
Q: 5. Draw a plausible mechanism for the following reaction. For full credit, show all intermediates,…
A:
Q: Will Ag2 C03 (Ksp = 8.1 250.0ml of 0.0062 m 0.00014 m Na2 Co3 are
A: Solubility equilibrium of Ag2CO3 Ag2CO3(s) ----> 2Ag+(aq.) + CO32- Qsp = [Ag+]2[CO32-] =…
Q: 2CO3) is titrated with 0.5000 N SO4. If the sample weighs 1.100 equires 35.00 mL of the acid fo…
A: Given, Mass of sample = 1.10 g Molar mass of Na2CO3 = 106 g/mol Normality of H2SO4 = 0.500 N…
Q: mixture of oxygen and nitrogen gases contains oxygen at a partial pressure of 279 mm Hg and nitrogen…
A:
Q: the Ksp expression of: Write @ Ag Noz (s) b) Ag2 Sou (s)
A: Given-> (1)->AgNO2 (2)->Ag2SO4
Q: (i) how many π bonds does the molecule have? (ii) how many unhybridized p-orbitals total does the…
A: I have given a detailed solution to the question as follows:
Q: Select the B-hydroxy carbonyl from which the product below was formed via an aldol reaction. A) I B)…
A: 1)Base catalyzed Aldol condensation
Step by step
Solved in 4 steps with 3 images

- The analgesic acetaminophen is synthesized by treating 4-aminophenol with one equivalent of acetic anhydride. Complete the equation for the formation of acetaminophen. a. Acetaminophen: b. Additional Product pls add the name for both, thanks!Using any necessary organic and inorganic reagents, show how you can carry out the chemical conversions shwon below. Please answer parts a, b, and c.Given this organic synthesis, is there any limiting and excess reagents? also describe the procedure is run, how is the reaction monitored?Is the order fo addition important? N-ETHYLALLENIMINE[Aziridine, 1-ethyl-2-methylene-] Submitted by Albert T. Bottini and Robert E. Olsen1.Checked by Thomas H. Lowry and E. J. Corey. 1. ProcedureCaution! This preparation should be carried out in a good hood to avoid exposure to ammonia. The operator should wear rubber gloves and protective goggles because 2-haloallylamines and ethylenimines can cause severe skin and eye irritation. A 2-l. three-necked flask is fitted with a sealed mechanical stirrer, a gas-inlet tube, and a dry ice condenser protected from the air by a soda-lime drying tube (Note 1). The system is flushed thoroughly with dry ammonia, and 32.8 g. (0.84 mole) of sodium amide (Note 2) is added to the flask. The system is again flushed with ammonia, the condenser is provided with dry ice covered by acetone, and 1.2 l. of liquid…
- For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).(a) Illustrate the following name reactions by giving example :(i) Cannizzaro’s reaction(ii) Clemmensen reduction(b) An organic compound A contains 69.77% carbon, 11.63% hydrogen and rest oxygen. The molecular mass of the compound is 86. It does not reduce Tollen’s reagent but forms an addition compound with sodium hydrogen sulphite and gives positive iodoform test. On vigorous oxidation it gives ethanoic and propanoic acids. Derive the possible structure of compound A.To prepare butanoic acid from 1-propanol, which sequence of reagent(s) is/are best employed? a) (1) NaCN; (2) H2O; (3) [H+] b) LiAlH4 c) K2Cr2O7 in acid d) (1) PBr3; (2) NaCN; (3) H2O; (4) [H+]
- Show how m-dibromobenzene can be synthesized from benzene from multiple reactions. Tip: think of diazotation as an important reaction in this process.As per the guidelines, three sub-parts will be answered, please help me with these three parts j, k lloudon 6th edition organic chemistry For each of the following reactions,provide the major product(s) or the necessary reagents to perform the chemical transformations indicated. If no reaction takes place, write NRProvide a synthesis for the molecules below.
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estroneOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?

