Balance each of the chemical equations. Fe(s) + 02(g) → Fe203(s) _Na3PO4(aq) _NH4NO3(s) – а. b. _NaNO3(aq) Pb(NO3)2(aq) – N2(g) + Pb3(PO4)2(s) + _H2O(g) C. --- ____02(g) +
Balance each of the chemical equations. Fe(s) + 02(g) → Fe203(s) _Na3PO4(aq) _NH4NO3(s) – а. b. _NaNO3(aq) Pb(NO3)2(aq) – N2(g) + Pb3(PO4)2(s) + _H2O(g) C. --- ____02(g) +
Introductory Chemistry: A Foundation
8th Edition
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter6: Chemical Reactions: An Introduction
Section: Chapter Questions
Problem 58AP
Related questions
Question
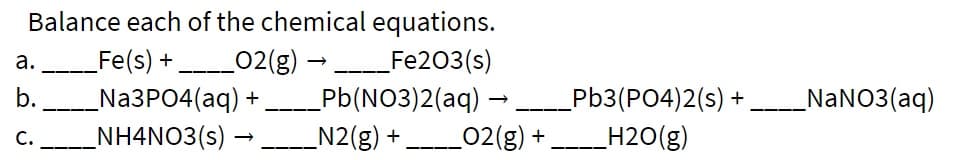
Transcribed Image Text:Balance each of the chemical equations.
Fe(s) + 02(g) → Fe203(s)
_Na3PO4(aq)
_NH4NO3(s) –
а.
b.
_NaNO3(aq)
Pb(NO3)2(aq) –
N2(g) +
Pb3(PO4)2(s) +
_H2O(g)
C.
--- ____02(g) +
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
