Balance the decomposition reaction: Hypothesis: What percentage do you predict for the KCI (potassium chloride) and KCIO, (potassium chlorate)?
Balance the decomposition reaction: Hypothesis: What percentage do you predict for the KCI (potassium chloride) and KCIO, (potassium chlorate)?
Chapter5: Gases
Section: Chapter Questions
Problem 161IP: In the presence of nitric acid, UO2+ undergoes a redox process. It is converted to UO22+ and nitric...
Related questions
Question
Transcribed Image Text:M..
New folder
Genetic drift - Wikip...
4 Algebra Foundation...
nposition of KCO, Lab
prmat Tools
Add-ons Help Accessibilit
Normal text
Comfortaa
14
+.
BIU
Balance the decomposition reaction:
Hypothesis:
What percentage do you predict for the KCI (potassium chloride) and KCIO, (potassium
chlorate)?
Materials:
safety glasses
mass balance
test tube
test tube clamp
test tube tongs
manganese dioide. MaO
mixture of KCO KC
ring stand
Procedure:
1. Bring a dry test tube to your teacher and obtain the manganese dicxide catalyst
2. Weigh the test tube with the catalyst (W,)
3. Obtain a sample of KCI and KCIO, mixture and mix it gently by tapping on the bottom
of the test tube several times
4. Weigh the test tube with all of the chemicals (W.)
5. Place the test tube in the holder (at an angle) and turn on the propane flame (make sure
the flame is on low)
a. Move the flame around to avoid overheating individual portions of the mixture
(the entire process should take about 3 to 4 minutes)
b. Once the oxygen is driven off, turn the flame off.
6. Let the test tube cool
7. Bring the test tube to the scale and weigh it for the last time (W)
Transcribed Image Text:mposition of KCIO, Lab
Format Tools Add-ons Help Accessibilit
Comfortaa
BIUA
Normal text
14
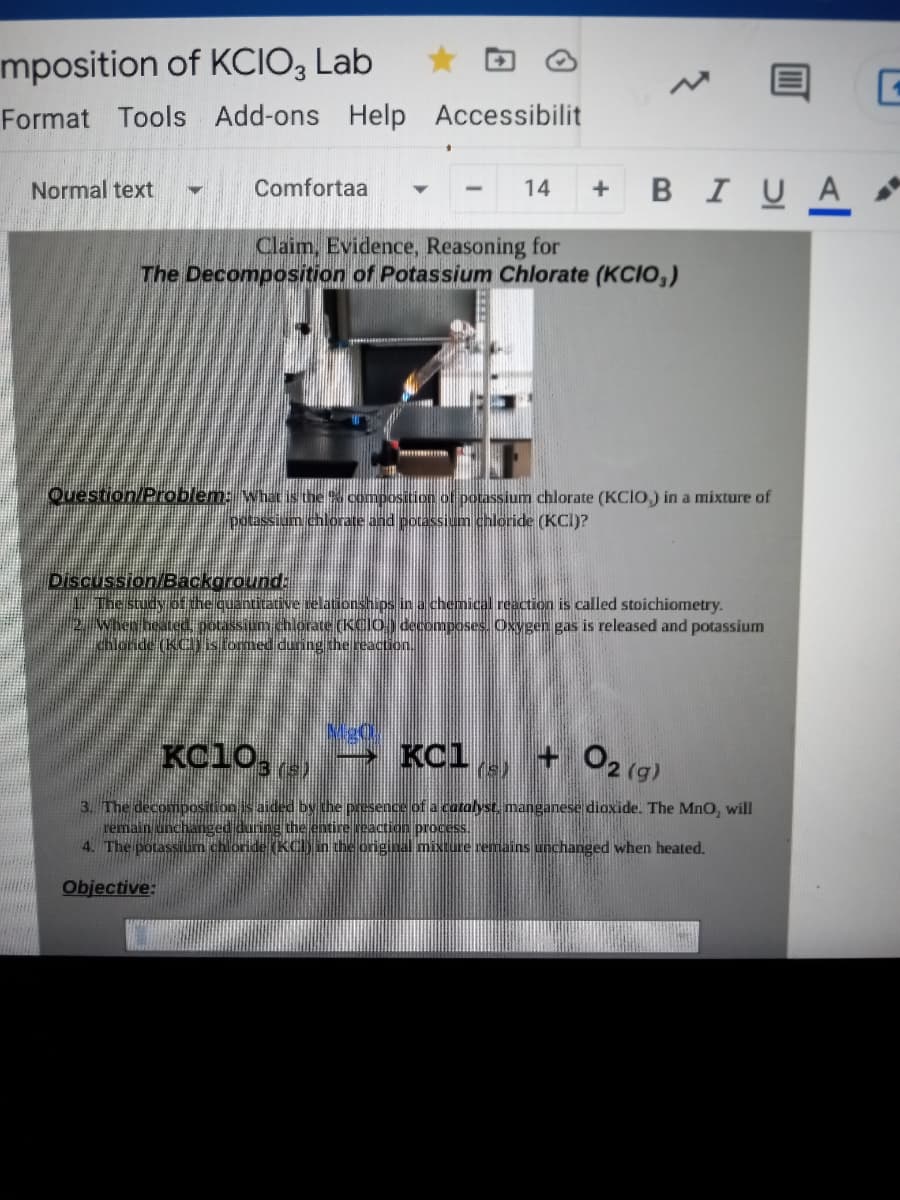
Claim, Evidence, Reasoning for
The Decomposition of Potassium Chlorate (KCIO,)
Question/Problem: what is the composition of potassium chlorate (KCIO,) in a mixture of
potassium chlorate and potassium chloride (KCI)?
Discussion/Background:
ive selationships in a chemical reaction is called stoichiometry.
chlorate (KCIO) decomposes. Oxygen gas is released and potassium
chlond
is formed during the reaction.
KC10, e)
KCl, + O2 (g)
3. The decomposition is aided by the presence of a cotalyst, manganese dioxide. The MnO, will
reaction process.
the original mixture remains unchanged when heated.
remain unchanger
4. The potassium chloride
Objective:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
