Q: Which chemical equation below is balanced? EQUATION A: HCl(aq) + Ca(OH)2(aq) ----> H2O +…
A: In a chemical equation, reactants are present on left side and products are present on right side of…
Q: 5. Reaction Ni + AGNO3 → : Type of reaction Balanced equation
A:
Q: balance the chemical equation for each reaction HCl04 + P4010----> H3P04 + Cl207
A: The reaction taking place is HClO4 + P4O10----> H3PO4 + Cl2O7
Q: T24 (This question uses the equation that was balanced in the previous question). Now give the…
A: A redox reaction is the one where both oxidation and reduction occurs simultaneously. Balancing of…
Q: balanced molecular equation (ME) write the balanced complete ionic equation (CIE) and write the…
A:
Q: Ionic Equation: 2Na+ (aq) + SO2 (aq) + Ca2+ (aq) + 2Cl- (aq) - CaSO4(s) + 2Na+ (aq) + 2Cl- (aq)…
A: Ans: Na+ and Cl- Explanation: The spectator ions, which do not participate in chemical reactions…
Q: BaCl2 + (NH4)2CO3 o BaCO3 + 2NH4Cl Balanced Equation: Total Ionic Equation: Net Ionic Equation:…
A: Applying concept of ionic and net equation.
Q: Which coefticient will balance the equation when inserted in the _?_spot in the equation? 3Cao 2H;P…
A: Balance the given above reaction with suitable coefficient --
Q: Balance and write the net ionic equation
A: According to the Law of conservation of mass " all atoms of various elements must be balanced on…
Q: need assistance with revising with what I have so far with these chemical balance equations: 1.…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: For the unbalanced chemical reaction shown below. __ Na2SO4(aq) + __ CaCl2(aq) ----> __ NaCl(aq) +…
A: The steps to write a net ionic equation are:- Step 1:- Write the balanced molecular equation. Step…
Q: Question attached
A: A new substance formed will have different chemical formula than the reactants. a) 2NH4NO3→2N2 + O2+…
Q: A. Balance the equation: - MnO2s) + Al(s) Mn(s) + Al2O3(9)
A: Chemical equations are balanced to follow "law of conservation of mass". A balanced chemical…
Q: NaCl (s) forms according to the reaction below between sodium and chlorine: Na (s) + Cla (g) → NaCl…
A:
Q: EQUATION
A: A reaction is said to be chemically balanced if the number of atoms involved in the reactant side is…
Q: 4. Consider the following unbalenced chemical equation to answer the following questions. FeCl3 (aq)…
A:
Q: 15. Balance this equation. CH4 + O2 --> CO2 + H2O
A:
Q: (c1) Tin (gives Sn2+) Balanced molecular equation: ? Edit (c2) Tin (gives Sn2+) Balanced ionic…
A: A chemical reaction can be represented by using symbols and formulas of the participating species.…
Q: NAME: CHEMICAL REACTION AND BALANCING PART 1: Determining the product of a chemical reaction…
A: Parts : 1 1) Reactions : C6H14 + O2 → CO2 + H2O Type of Reactions : combustion reaction 2)…
Q: Although none of the double replacement reactions below are balanced (none have any coefficients),…
A: A Double Replacement Reaction is a chemical reaction where two ionic compounds (reactants) exchange…
Q: Write a balanced molecular equation for the reaction .of hydrofluoric acid and calcium .A Ca(s) +…
A: Reaction is called balanced when atoms are equal in both reactant and product side.
Q: Which of the following is the correct balanced chemical equation for dissociation of aluminum…
A: Balanced chemical reaction: A reaction in which all the elements of the compounds in the reactant…
Q: Balance thh equation NH4 NO3(s) --> N2(g) + 02(g) + H2 O(g)
A: A chemical reaction is said to be balanced when the number of atoms on the reactant and product side…
Q: balanced molecular equation (ME) write the balanced complete ionic equation (CIE) and write the…
A:
Q: 65. Balance each of the following equations that describe decomposition reactions. a. CaSO4 (s)…
A: NOTE : Since you have posted a question with multiple subparts . we will solve the first 3 subparts…
Q: What chemical test could be used to confirm that the gas produced in a chemical reaction is carbon…
A: Chemical test Pass the gas through lime water ( Solution of CaO). The originally lime is…
Q: The ions formed when Sr(C₂H₃O₂)₂ dissociates in water are: A) Sr2+, C4-, H+, and O2- B) Sr+ and…
A: 1) The compound given is Sr(C2H3O2)2.
Q: Part II: Determine whether the following Single Replacement reactions will occur. Write Yes or No.…
A: A single replacement reaction is a reaction in which one element is substituted by another element…
Q: Which chemical equation below is balanced? EQUATION A: Al2(SO4)3(aq) + BaBr2(aq) ---->…
A: The Chemical reaction follows the law of conservation of mass. According to the law of conservation…
Q: Na Balancing Chemical Equations Balance the following equations using whichever method you prefer:…
A: balance the both side atom reactant side atom must be balance by product side atom these all…
Q: Complete ionic equation for chemical reaction KBr(aq) + AgC2H3O2(aq) =KC2H3O2(aq) + AgBr(s)
A: The reaction given is, => KBr (aq) + AgC2H3O2 (aq) → KC2H3O2 (aq) + AgBr (s)
Q: ) hydrochloric acid and zinc Type of Reaction: Balanced Chemical Reaction: 2) Heating copper (II)…
A: The balanced equation and reaction types are given below
Q: Equations A. Zn + H2SO4 > ZNSO4 + H2 B. Mg + 0, - 2MgO C. Na,S + 2HCI 2NACI + H,S D. H,S + S0, - 3S…
A: If number of atom and charge are balance both side that is products side and reactants side the…
Q: reaction, and balance the equation. Reaction NazC04 + H2SO4 → Type of reaction Balanced equation…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: III. Reacting Aqueous CuSO, with Aqueous NaOH 4 1. Appearance of CUSO, solution 4 2. Appearance of…
A: The reactants given are colourless clear solution of NaOH and blue colour solution of CuSO4 Hence…
Q: Question 12 of 20 Complete the balanced molecular chemical equation for the reaction below. If no…
A:
Q: Question 10 Is this a balanced equation? 2KCI + LizO K20 + 2LICI Your answer:
A:
Q: 13. Using the following reaction, answer the following questions: Balance the reaction. Ba(NO3)2…
A: Note:- As per bartleby guidelines, I need to solve the first question in case of multiple questions.…
Q: T24 (This question uses the equation that was balanced in the previous question). Now give the…
A: To determine the coefficient of permanganate, the reaction needs to be balanced.
Q: With a 0.50 to1 ratio EXPLAIN how closely that relates to the balanced chemical equation. (the…
A: A question based on stoichiometry that is to be accomplished.
Q: Balance the acidic equations: NO2 → NO3− + NO O2 + As → HAsO2 + H2O
A:
Q: Question 4 Predict the products and balance the equation: No Spacing between coefficients…
A: 4.) Sodium reacts with bromine to give sodium bromide(NaBr). Write the equation with the product.…
Q: 1. For the following chemical equations: (a) Predict the products (b) Balance the equation and (c)…
A:
Q: Question 33 Which chemical equation shows decomposition reactions? O Mg (s) + O2 (g) → MgO (s) O CH4…
A: Decomposition reaction is reaction in which one compounds breaks into two or more than two…
Q: upon the complete reaction of 26.4 grams cetrafluoride? silicon tetrafluoride (s) + water (I) -->…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: Balance each of the following reactions and identify each type of reaction: 4. Pb + H3(PO4) > H2 +…
A: Balanced the following reaction and idenyify type of reaction--->
Q: 2+ 2 Mn04 (aq) + 10 Br (aq) + 16 H"(aq) - 2 Mn (aq) + 5 Br2(aq) + 8 H20() How many electrons are…
A: In the given balanced equation, if we count the oxidation state of each species, then for MnO4-,…
Q: write down the corresponding ionic equation. Predict products and balance the equation 1.Nitric…
A: There are five reactione in the question given. The corresponding balanced chemical equations,…
Q: Question 1 Balance the following reaction CO2 + H20 C6H6 -> O2 Question 2 Balance the following…
A: The given unbalanced equations are: _CO2 + _H2O -------> _C6H6 + _O2 _O2 + _S8…
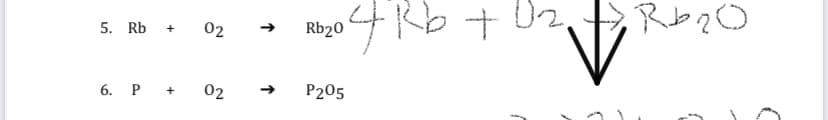
Step by step
Solved in 2 steps with 2 images

- HCl(g) deltaG=-95.3O2(g) deltaG=0 H2O(g) deltaG=-228.6Cl2(g) deltaG=0Write the electronic configuration of Ba (Barium) Select one: a. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d1 0 5s2 5p6 b. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d1 0 5s2 5p6 6s2 c. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d1 0 d. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d1 0 5s2 5p6 6s2 e. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d1 0 5s2Thank you. I'm just confused as what is is the reason 122.1 has to be multiplied by (-45543.23)?
- Describe nuclear magnetic resonance spectroscopy .2. The observed nuclear moments of 209 Bi are: I=9/2; μ = + 4.1 μN; Q=-0.4 x 10-28 m² Determine the expected values for these moments according to the simple Shell model and comment on any significant differences.Food dye spectroscopy 2. At what wavelengths should the readings be taken? What instrumentation will you utilize with a solution of a known dye to determine the wavelength for your analysis?



