Q: Volume Acid: 20.0 mL Volume of Base added: 14.20 mL Molarity of Base: 0.10 M 1) From the…
A: HCl reacts with NaOH to form sodium chloride and water. The equation for the balanced chemical…
Q: calculate the molarity of a sulfuric acid(H2SO4) solution if 30.10 mL of o.6210 M NaOH is required…
A:
Q: Pb(NO,),(aq) + 2 KCI(aq) PBC1, (8) + 2 KNO,(aq) acid-base neutralization CH, (g) + 20,g) → CO,(g) +…
A: ->In acid base reaction acid and base reacts to form salt and water. ->In redox reaction one…
Q: 1) Potassium permanganate solutions used in oxidation-reduction titrations are often standardized…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: You have measured 1.231 g of unknown acid on the balance. Final burette reading is 23.63 mL Initial…
A: Molarity is defined as number of moles of solute present in 1 Litre of the solution
Q: Suppose you are titrating a sulfuric acid solution of unknown concentration with a sodium hydroxide…
A: Answer:- This question is answered by using the simple concept of stoichiometry which will give the…
Q: Suppose you are titrating vinegar, which is an acetic acid solution of unknown concentration, with a…
A: Given Volume of NaOH ( V1 ) = 34.71 mL Molarity of NaOH ( M1 ) = 0.1245 M Volume of acetic…
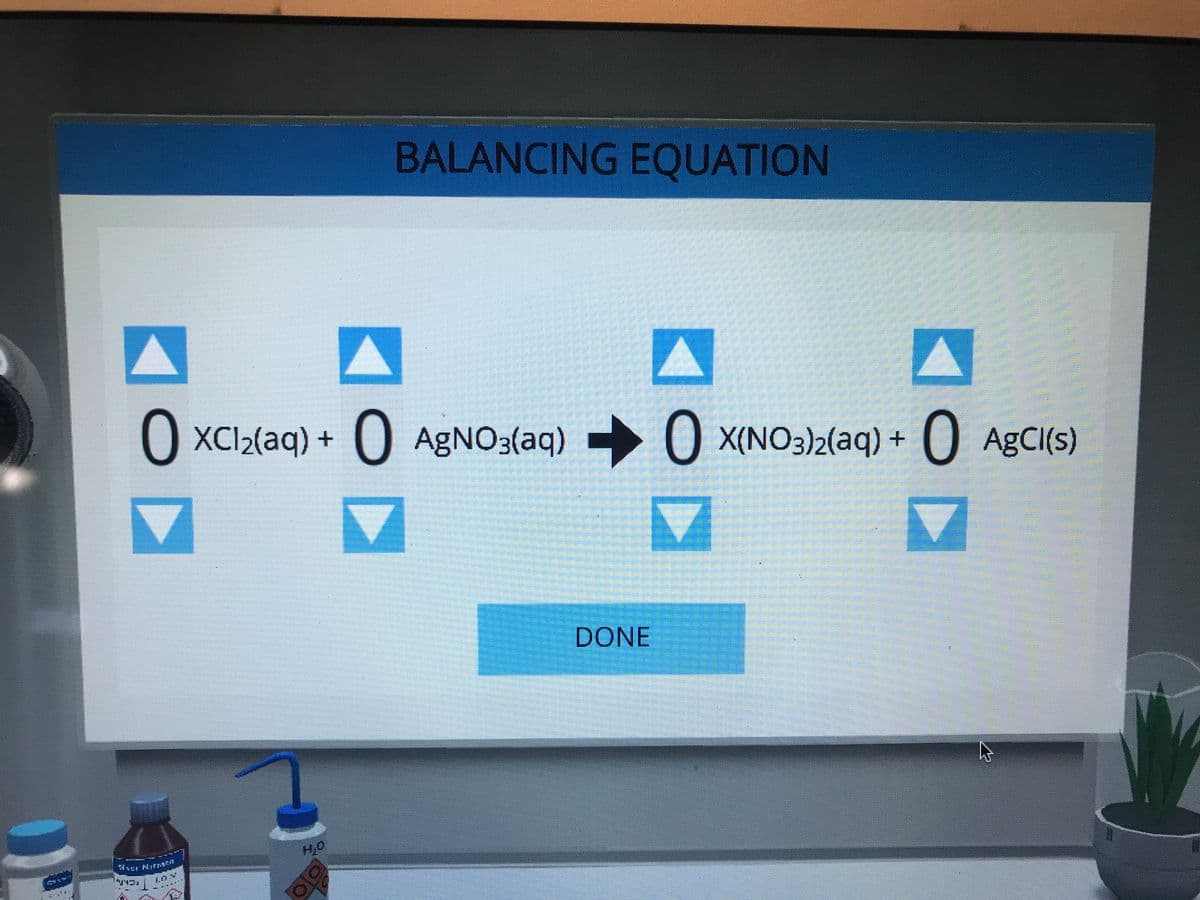
Q: BALANCING EQUATION 2 XC₁₂(aq) + 2 AgNO3(aq) DONE OX(NO3)₂(aq) + AgCl(s)
A: For a balanced equation number of atoms of each element must be same on both sides.
Q: units in the first column. Tablet identity (brand) Tums Titration Tablet 1 Mass of tablet/sample (g)…
A: Volume of NaOH = final reading - initial reading = 48.7 - 30.3…
Q: What are the spectator ions in the following ionic equation? 2 NH4+(aq) + CO32−(aq) + 2 Na+(aq) + 2…
A: Spectator ions are those which are present on the both sides of the total ionic reaction. These ions…
Q: Can I please get assistant with the following problem? Write a balanced complete ionic equation for…
A: We would write the ionic equation of compound A2B2 (s)
Q: Given the following information, what is the molar mass of an unknown monoprotic acid? (titration)…
A:
Q: Balance the equations under basic solution. (Show each step) a. SO32- + Cu(OH)2 ➡️ SO42- + Cu(OH) b.…
A: The balanced chemical equation has the number of atoms of the reactant should be equal to the number…
Q: Elc. A JU student was asked to prepare a pH = 4.0 acid solution in which the molarity of H is 1.0 x…
A: GivenDensity of H2SO4= 1.84 g/mlVolume of H2SO4 taken= 1.500ml
Q: (Eqn. 1) 1 OCl− + 3 I− + 2 H+ → 1 I3− + 1 Cl− + 1 H2O (Eqn. 2) 2 S2O32− + 1 I3− + 0 H+ → 3 I− +…
A: Given reactions are Reaction (1): 1 OCl− + 3 I− + 2 H+ → 1 I3− + 1 Cl− + 1 H2O Reaction (2): 2…
Q: If 0.100 grams of unknown solid are dissolved in water and titrated with 4.68 mL of the NaOH, what…
A: Molar mass is grams / moles. We have grams from the weighed mass, we can calculate moles of acid…
Q: Balance equation _____->BaBr(aq)+H20(l)
A: We have to balance the given equation
Q: Suppose a group of students put 2.92 grams of an unknown solid acid into a 250-mL volumetric flask…
A:
Q: First balance equation and then write it as a balanced molecular, ionic, and net ionic equation for…
A: In the Molecular equation , we write the Balanced chemical equation for the Reaction including the…
Q: Suppose you are standardizing a sodium hydroxide solution with KHP (molar mass=204.2 g/mol)…
A: Given, mass of KHP = 0.292 g volume of KHP solution = 250.0 mL and, volume of NaOH…
Q: In the experiment conducted to determine the amount of acetic acid in vinegar, 8.2 mL of a 0.098 M…
A: Molarity of a solution is equal to the number of moles of solute present in one liter of solution.…
Q: F (aq) 4. HF(ag) + H200 > H30*aq) HF (aq) in the equation above is a(n) H3O" (aq) in the equation…
A:
Q: 10.0mL of concentrated H3PO4 (aq) is diluted to the final volume of 250 mL. Then 150 mL of the…
A: Given: Volume of concentrated H3PO4 solution = 10 ml volume after dilution = 250 ml Volume of…
Q: balanced molecular equation (ME) write the balanced complete ionic equation (CIE) and write the…
A: The balanced molecular equation represents the non-dissociated molecular species participating in…
Q: Given the following values, what is the concentration of the acid? (With solution) Initial Volume…
A: Given: Initial volume of acid = 10.150 mL Final volume of acid = 11.620 mL Initial volume of NaOH =…
Q: First balance equation and then write it as a balanced molecular, ionic, and net ionic equation for…
A: The chemical name of AgNO3 is silver nitrate The chemical name of NaOH is sodium hydroxide The…
Q: According to the reaction below: if it required 65.19 mL of HCl with an unknown concentration to…
A:
Q: An unknown vitamin C tablet was titrated with a 0.2030 M KOH solution. Calculate the milligram mass…
A: As vitamin C is the mono protic acid, 1 mole of vitamin C reacts with 1 mole of KOH. At equivalence…
Q: What is the product of the following reaction equation after balancing? If no reaction state that "…
A: Metals react with acid to produce metal salt and hydrogen gas.
Q: 10.0mL of concentrated H3PO4 (aq) is diluted to the final volume of 250 mL. Then 150 mL of the…
A: The solvent is the chemical in the combination that is present in the greatest amount, while the…
Q: balanced molecular equation (ME) write the balanced complete ionic equation (CIE) and write the…
A: The net ionic equation may be explained as the equation in which only those species are available…
Q: Balanced net ionic equation for: 1. CrO42- and 0.2M Ba(NO3)2 2. CrO42- and 0.2 M Pb (NO3)2 3.…
A: 1. Balanced net ionic equation CrO42- + Ba(NO3)2 → CrO42- + Ba+2 + 2NO3- → BaCrO4 + 2NO3-CrO42- +…
Q: - Calculate the equivalent weight and normality for a solution of 6.O M H3PO, given the following…
A: The equivalent weight of the compound can be defined as the ratio of molecular weight by no. of…
Q: This question has multiple parts. Work all the parts to get the most points. a In the equation: 2…
A:
Q: Suppose you are titrating vinegar, which is an acetic acid solution of unknown concentration, with a…
A:
Q: Cu(s) + H*(aq) + | NO3°(aq) NO2(g) + H20(1) + Cu2+(aq) + NO3°(aq) (No species should appear on both…
A: In this reaction , Cu(s) oxidises into Cu+2 , So Cu acts as reducing agent , thus there should be a…
Q: n student worker in the Cal Poly chemistry stockroom is asked to determine the concentration of an…
A: An acid-base reaction's equivalence point is the the stage where the acid and base concentrations…
Q: The tartaric acid, H2C4H4O6 (150.087 g/mol ) in 100.0 mL wine sample was determined using sodium…
A: The question is based on the concept of titrations. we are titrating a weak acid with a strong…
Q: Balance eaxch equation H2O2+ClO2 --> ClO2–+O2 (in basic solution)
A: 1. H2O2 + ClO2 --> ClO2– + O2 (in basic solution) To balance given reaction in basic medium;…
Q: Suppose you are titrating vinegar, which is an acetic acid solution of unknown concentration, with a…
A: The number of moles of a substance is the weight of the substance upon its molar mass. Molarity of a…
Q: Suppose you are titrating vinegar, which is an acetic acid solution of unknown concentration, with a…
A:
Q: When the following reaction is balanced under acidic conditions, what is the coefficient in front of…
A:
Q: A volume of 60 0 mL of aqueous potassium hydroxide (KOH) was titrated against a standard solution of…
A:
Q: In an experiment to determine the content of acetylsalicylic acid in an aspirin tablet, the result…
A: Answer: Given- the calculated mass of acetylsalicylic acid was much higher than the indicated amount…
Q: Balance this equation in basic solution ClO2^- + S2O3^2- Cl- + SO3^2-
A: To balance the equation in basic medium, ClO2- + S2O32- → Cl- + SO32-
Q: What are the spectator ions for the following reaction. LIOH(aq) + HNO3laq) →
A: Spectator ions are those ions that are existing on both sides of a reaction i.e. reactant side as…
Q: Identify the correct net ionic equation for the reaction that occurs when solutions of HF and KOH…
A: HF is a weak acid and does not dissociate completely and KOH is a strong base which dissociates…
Q: A sample of 1.916 g glutamic acid (H2C5H,NO4, 147.13 g/mol) is titrated with 41.85 mL solution of…
A: The balanced equation showing the reaction taking place between glutamic acid and NaOH is- H2C5H7NO4…
Q: The Follow-up: Write the balanced molecular equationfor 2c, using K+(aq) as the spectator ion.…
A: CrO2-(aq) + ClO-(aq) →CrO42-(aq) + Cl2 (g)(basic solution) Balanced chemical equation :- Ionic…
Q: Zn(s) + 2 Ag+ (aq) ® Zn2+(aq) + 2 Ag(s) What would happen to the value of Ecell if the…
A: Ecell :- It is the cell potential of a reaction at non standard condition. That means the cell…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Balance the following net ionic equation (there is one mole of (NH4)3PO4●12MoO3 in the balanced equation- do NOT change this coefficient): HPO42-(aq) + NH4+(aq) + MoO42-(aq) + H+(aq)(NH4)3PO4●12MoO3(s) + H2O (l)Balance the following equation. ___ M2CO3 (s) + ____ HCI(aq) --->___ CO2 (g) + ____ MCI (aq) +____ H2O(I) With the chemical equation above the following data was obtained: Mass of unknown + weighing boat - 2.87g Mass of weighing boat - 2.14g Volume of 1.0 M HCI used 19.11 mL What is the # of moles of HCI # of moles of unknown carbonate used The molar mass of unknown carbonate What is the identity of the unknown carbonate?The following reaction occurs in basic solution: _ H2O(aq) + _ MnO4–(aq) + _ ClO–(aq) → _ MnO2(s) + _ ClO4–(aq) + _ OH–(aq) When the equation is properly balanced, what is the sum of the lowest whole-number coefficients? a. 9 b. 10 c. 20 d. 12 e. 6
- when the following equation is balance, the coefficient of H2 is K(s)+H2O(L)-->KOH(aq)+H2(g) answers options (a)1 (b)2 (c)3 (d)4 (e)5Aqueous sodium hypochlorite (NaOCl, household bleach) is a strong oxidizing agent that reacts with chromite ion [Cr(OH)4-] in basic aqueous solution to yield chromate ion CrO42- and chloride ion Cl-. The next ionic equation is: ClO- (aq) + Cr(OH)4-(aq) ------> CrO42-(aq) + Cl-(aq) Unbalanced Balance the equation using the method described in section 1 of the Electrochemistry chapter.Can I please get assistant with the following problem? Write a balanced complete ionic equation for the following ionic solid. Assume A is a metal ion and B is an anion. For example, CaCO3(s) ⇌ Ca2+(aq) + CO32-(aq) A2B2(s) ⇌ x A+(aq) + y B-(aq) What is the value of y?
- Identify the spectator ions in the following balanced molecular equation: KBr (aq) + AgNO3 (aq) → AgBr (s) + KNO3 (aq) Group of answer choicesThe Original volume of unknown NaCl solution = 98.407mL The concentration of Ag + within the 10 mL unknown NaCl solution after AgCl is added [Ag+] = 4.71370e^-10 Calculate Calculate the mass of NaCl in the 10 mL unknown solution. Show your work.? Hint: mole = molarity x L; mass = mole x molar mass AgCl(s)⇌Ag+(aq)+Cl−(aq) Ksp=[Ag+][Cl−]KHP (aq)+NaOH (aq)-->NaKHP (aq)+H2O(l) Trial Vi (mL) Vf (mL) 1 36.19 48.56 2 3 A student masses out 1.096 grams of KHP, which is dissolved in 25.00 mL of DI water. This KHP solution is titrated with NaOH, as shown in the equation above. Calculate the molarity of the NaOH solution.

