Q: 2.2 6. (1) Define pH in words. The strong acid HCl has a pH value of 1, use the following equation…
A:
Q: Why is process isolation bad?
A: According to research, "poor overall cognitive performance and poorer executive functioning, faster…
Q: what is the NMR spectrum of benzylcyclohexan-1-ol. explain in detail how to expect to see the nmr…
A:
Q: Complete the following diagram for the carbon molecule (C2):
A:
Q: 2. Write the abbreviated ground state electron configuration for each element. b) Pb a) Ca
A:
Q: 4. Determine which element has the following set of quantum numbers for its outermost electron, show…
A:
Q: Consider the reaction: P4(s) + 6Cl₂(g) → 4PC13 (1) Given an initial mass of 15.75 g P4, an excess of…
A:
Q: How many grams of sodium hydroxide is needed to prepare 500 mL of 0.050 M NaOH solution? atomic…
A:
Q: Balancing Act Name This activity will help you become skilled at balancing chemical equations. 1.…
A: Here, we have to balance the given chemical equations. Note that, in a balanced equation, both sides…
Q: Show the retrosynthesis and the synthesis of the following compounds: OH Target material Starting…
A: Dear student, the given question is a multiple question, hence I solved first question according to…
Q: What is the expected pH for solutions that contain the following initial compositions? a) 1.0x10-5…
A:
Q: The octahedral ion complex [CoF6] 3‒ absorbs at 675 nm. (a) What color is the complex? (b)…
A:
Q: Calculate the pH after the addition of 60.0 mL of 0.3 M NaOH to 50.0 mL of 0.20 M LIN O 10.46 11.86…
A:
Q: Which rock and roll star has the same name as this hexapeptide in single letter code? NH₂ SH NH₂ NH₂
A:
Q: 3 Problem 3: A gas that has a volume of 34 L, a temperature of 318 K and an unknown pressure, has…
A: Here we have to determine the original pressure of gas in the following given process.
Q: 1. What is the specific gravity of N2 * at 80°F and 745 mm Hg compared to air at 80°F and 745 mm Hg?
A:
Q: A solution contains 0.264 M sodium hypochlorite and 0.224 M hypochlorous acid. The pH of this…
A:
Q: Which of the following characteristics is/are the advantage(s) of HPLC over GC? I. Only small…
A: HPLC is high performance liquid chromatography.and GC is gas chromatography.
Q: 4. What are the 3 types of lipids and what are the basic components of each? a. b. C. . 5. Name…
A: There are 3 different types of lipids.
Q: Half a mole of an ideal gas expands isothermally and reversibly at 298.15 K from a volume of 10 L to…
A: The change in entropy (∆S) of an ideal gas is related to heat exchanged (q) in a process at constant…
Q: Draw the cyclic voltammogram of parathion and explain the observed redox peaks. s
A:
Q: Table 2 Absorbance vs CoCl2 concentration Test Tube Number Cobalt Chloride Concentration…
A:
Q: 1.2 2(1) In the contact process: 2SO₂(g) + O₂(g)2SO3(g) AH = -197 kJ mol-¹ Predict the position of…
A:
Q: 2. Write the abbreviated ground state electron configuration for each element. a) Ca b) Pb
A:
Q: Theoretical mole ratio from formula: moles of Cu per moles of CuCO3 Cu(OH)2 = 1.97.
A: Mole ratio =molar mass of particular compound /molar mass of whole compound.
Q: C₂H5OH + 302 2CO₂ + 3H₂O AH=1367kJ/mol How many grams of carbon dioxide are produced when 530. kJ of…
A: Ethanol reacts with oxygen to form carbon dioxide and water. The equation for the balanced…
Q: For CHCl3(g), Cp = 7.05 + 35.60 x 10-³T - 216.9 x 10-7T² cal mole-1 K-1. Assuming this gas to be…
A:
Q: Fractional distillation lab What is the purpose of distillation? What is the theory of fractional…
A: According to bartleby guidelines we're supposed to answer first three subparts of a multipart…
Q: etermine the volume of barium sulfate produced when 00.0 grams of barium reacts with sulfuric acid.…
A: Answer :- given mass of Barium (Ba) = 200g Chemical equation- Ba + H2SO4 -> BaSO4 + H2…
Q: 8.3.01 x 1023 CH₂O molecules moles CH₁₂0 9. 1.00 x 1025 Na atoms moles No
A: Here, we have to find the number of moles of C6H12O6 in 3.01×1023 C6H12O6 molecules and the number…
Q: An ion-selective electrode for fluoride was investigated. The following results were obtained. Plot…
A:
Q: What volume of water must be added to 10 .0 mL of 12.0 M HCl to make the final concentration of the…
A: Given data :
Q: What is the potential that develops at the interface between the saturated KCI solution in the salt…
A: Electrochemistry is branch of chemistry in which both oxidation and reduction reaction occurs…
Q: You tested the buffer capacity of 50 mL of a 40 X dilution of a 0.012 M Glutamic acid solution.…
A: pH+ pOH = 14 pOH = - log [OH-] Also, No of moles = Molarity * Volume (L)
Q: Which of the following units are used to measure heat? Select all that apply. Kelvin Celsius…
A: Heat is a type of energy and the heat of a system can be measured by measuring the temperature of…
Q: Classify the following by the sign of AE for the system. Drag the appropriate items to their…
A: From first law of thermodynamics, ∆E=Q+W When system expands, work is done by the system; and when…
Q: 23 C12H22011+ O2 CO₂ + H₂O Mg(OH)2 + H₂SO4 → MgSO4 + H₂O NaOH + CuSO4 → Na2SO4 + Cu(OH)2 C4H12 + O₂…
A:
Q: 1. What is the specific gravity of N2 * at 80°F and 745 mm Hg compared to air at 80°F and 745 mm Hg?
A:
Q: quation 2 8+8 H₂ 0₂ Reactants Number of hydrogen atoms Number of oxygen atoms -> go H₂O Product…
A:
Q: Consider a helium atom(m=6.6m10^-27 kg) that is confined to mobility along a line with a length of…
A:
Q: Name: Calculate each of the molar masses below. Use the space below in each box to show your work.…
A: Ans : 3) 1 mole of Mg = 24.31 g Mg i e Mg = 1g × 24.31 Total = 24.31 g 4) 1 mole of H2SO4…
Q: The incorporation compound Na2 [Ni (CN) 4] is given. a) What is the name of this association? b)…
A:
Q: Draw the final organic product of the reaction. HC OH CHỊCHINH, heat HC CH What is wrong with this?…
A: Carboxylic acid (Ph-COOH) when heated in presence of primary amine (CH3CH2NH2), a secondary amide is…
Q: A -1 B +2 C +4 E 4
A:
Q: 包 週 (iv) ZH N Η HN H 225°C Pyridine 120°C ?A?A? ?+ ? + H₂ +SO3 (0) KMnO4 (0) alc. KMnO4
A:
Q: How many grams of KI salt (M.M. = 166.01 g/mol) should be weighed and added to 200 ml water (M.M.…
A: Here the solute is KI and the solvent is water. In the case of a 400 dilution solution, for every 1…
Q: 23) trans-1,2-difluoro-ethene undergoes fluorination. Name final product. 24) 1,4-nonadiene…
A: 23) We have to give the name of the final product of the fluorination of the…
Q: Construct a simulated proton-decoupled 13C NMR spectrum for tert-butylamine. Drag the resonance…
A: In NMR, splitting of signal is occurred according to tge presence of number neighbouring nuclei…
Q: Select Draw Erase ||||||||||| H₂SO4 H₂O OH 3 C Draw the products formed when each ester is…
A: In this question, we have to draw the product formed when given compound (ester) is hydrolyzed with…
Q: Question 3 Circle the best answer: a. Dissolving O in Cu: a) O: solvent, Cu: solute b) O: solute,…
A:
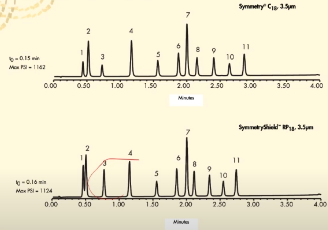
Based on the chromatograms, Symmetry C18 (top) is less selective than Symmetry Shield RP18 (bottom) for the analytes given, true or false?
Step by step
Solved in 2 steps

- Convert kPa/sec to uL/min for the following values. -0.0019 kPa/sec -0.0029 kPa/sec -0.0028 kPa/sec -0.0021 kPa/sec -0.0026 kPa/sec -0.0034 kPa/sec4. Ksp=4.1x10-36 for Pb3(AsO4)2Calculate Eofor Pb3(AsO4)2(s) + 6e- 3Pb(s) + 2AsO43-Pb2+ +2e- Pb(s) Eo= -0.126 VChemistry (i) For T = 298K ΔG° = -8.314 x 298 x ln (2.83x10-3) = 14.524 KJ/mol (ii) For T = 308.15K ΔG° = -8.314 x 308.15 x ln (9.619x10-3) = 11.891 KJ/mol (iii) For T = 318.15K ΔG° = -8.314 x 318.15 x ln (3.14x10-2) = 9.147 KJ/mol (iv) For T = 328.15K ΔG° = -8.314 x 328.15 x ln (8.82x10-2) = 6.621 KJ/mol (v) For T = 338.15K ΔG° = -8.314 x 338.15 x ln (2.42x10-1) = 3.990 KJ/mol Based on the measured ΔG° values, is this equilibrium spontaneous at room temperature? Which factor, entropy, or enthalpy, has the greater impact on spontaneity in this case? Explain your answers.
- Chemistry Fraction no. Elution Vol. (mL) Abs (λ = 410) Fraction no. Elution Vol. (mL) Abs (λ = 410) 1 (PB pH 6.0) 1 0.0050 11 11 0.0041 2 2 0.0032 12 12 0.0060 3 3 0.0040 13 13 0.0064 4 4 0.0064 14 14 0.0063 5 5 0.0067 15 15 0.1109 6 6 0.0067 16 16 2.8902 7 7 0.0050 17 17 0.8500 8 (PB pH 8.0) 8 0.0049 18 18 0.2019 9 9 0.0052 19 19 0.0378 10 10 0.0050 20 20 0.0073 Use the data above to plot an elution profile, in which the absorbance is on Y-axis, and elution volume (volume = fraction number) is on X-axis. The elution profile must indicate at (1) which point the pH change occurred, (2) the fraction(s) which contain the highest myoglobin content, and (3) the maximum absorbance detected.Sample IdentificationCodeConcentration of M%TA=2-log(%T)Q50004.00 x 10-417.90.75Q50013.20 x 10-425.00.6Q50022.40 x 10-435.70.46Q50031.60 x 10-450.20.3Q50048.000 x 10-570.80.15SampleIdentificationCode%TA=2-log(%T)AMQ0210150143.70.359518560.360.000192Q0210150244.10.355561410.360.00018Q0210150343.80.358525890.360.00017Q0210150444.10.355561410.360.00018Q0210150543.80.358525890.360.00017What was their percent error?43%Does Batch 021015 meet legal requirements?No, because it is not between 2.85 * 10(4) and 3.15 * 10(4)Well #DropsBluedye1234567891012345678910Drops 9Distilled water876543210Concne 0.26tration0.52Test Tube #0.781.041.3Solutions3Concentration (M)2.082.32.6Concentration (ppm)1:1 dilution11.82Starting Dilution21.562:1 dilution0A.Zero standard0Was your calibration curve as linear as you expected?B.Did you experience any “drift†of the resistance readings?C.What is the equation of your best-fit line?D.What commercial drink did you analyze?E.Assuming…An analysis of city drinking water for total hardness was done by two students in the laboratory and produced the following results (in ppm CaCO3): Student A: 228.3, 226.4, 226.9, 227.1, and 228.6. Student B: 229.5, 226.1, 230.7, 223.8, and 227.5What is the 95% confidence interval for the mean?
- 5.61 x 10^-12 is the kspQq.1. Subject :- Account1. Which of the following is NOT a true representation of the Lineweaver-Burk equation? Group of answer choices a. y-intercept = -1/Vmax b. x-intercept = -1/Km c. y = 1/V0 d. x = 1/[S] e. slope = Km/Vmax 2. If the y-intercept of a Lineweaver-Burk (double reciprocal) plot = 39.37 (sec/millimole) and the slope = 75.3 L/sec, Vmax equals: Group of answer choices a. 75.3 millimoles/sec. b. 0.0254 millimoles/sec. c. 0.523 millimoles/sec. d. 39.4 millimoles/sec. 3. Which of the following is true regarding allosteric enzyme behavior?Group of answer choices a. Allosteric enzymes are rarely important in the regulation of metabolic pathways. b. An allosteric effector binds reversibly on the surface of the enzyme. c. An hyperbolic representation of the relation initial velocity vs [S] is a specific characteristic of an allosteric enzyme behavior. d. Michaelis-Menten kinetics model the kinetic behavior of allosteric enzymes.
- Station 1 Pressure 10 mL 94.96 kPa 5 mL 203 kPa 7mL 121kPa 15mL 77 kPa 5mL 240 kPa what would be the inverse of these volumes?(6.65x10^5)/(6.60x10^-6) (3.73x10^8) Please Calaculition. Only typed solutionSimplify the following quantities using a unit with an appropriate prefix: (a) 2.5 104 Hz. (b) 8.43 105 mmol. (c) 7.44 104 nm. Show complete solution

