Q: 4. Oxidation state of Mn in MnO2 is a) +2 b) - 2 c) +4 d) - 4
A:
Q: For items 1-8, provide the products for the given reaction. Correctly draw the stereochemistry (cis/...
A: Answer of the question given below,
Q: Methane, CH4, burns in oxygen. a what are the products of the reaction? (Express your answers as che...
A:
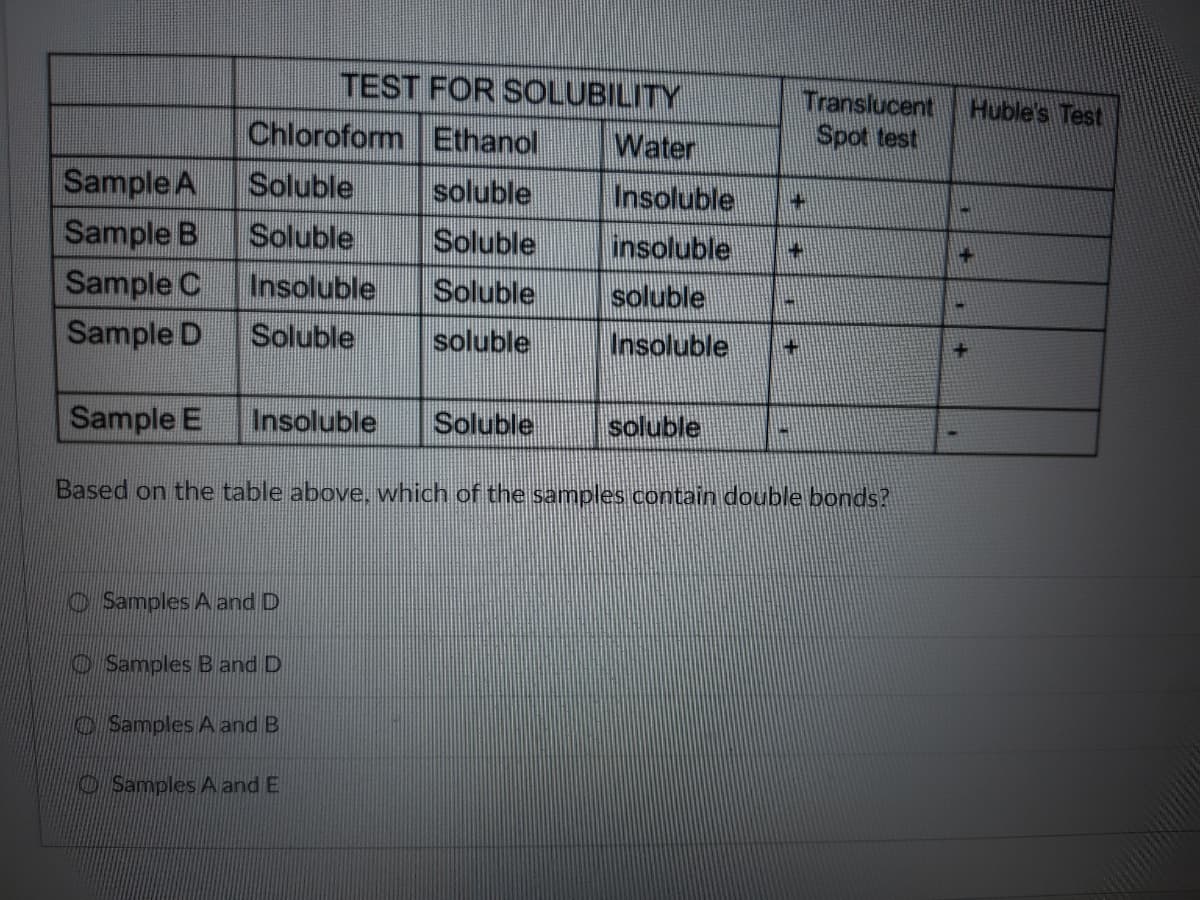
Q: Which of the following will be soluble with water? Group of answer choices Vegetable Oil Desi Ghee...
A: The solubility of a compound mainly depends on the following factors; Temperature Nature of solute ...
Q: Balance the reaction, do not leave any fractions, don't leave anything blank, then answer the questi...
A: In the balance reaction atoms of each element is same on both sides.
Q: Explain how TLC and column chromatography can be used together to ensure the perfect separation of t...
A: Chromatography is a technique for the separation of a compound from the mixture. The column chromato...
Q: Weak base: 25 mL of 0.00255 M hydroxylamine HONH2 (Kb = 1.1 × 10-8) Strong acid: 0.001545 M HNO3.
A: When hydroxylamine is reacted with nitric acid, following reaction occurs: HONH2+HNO3→HONH3++NO3-
Q: Calculate the specific heat (g'C) of gold if it takes 113.3 J of heat to raise the temperature of 29...
A:
Q: Consider the reaction below. 1.7 mol of A and 6.5 mol of B are added to a 1 L container. After a tim...
A: A = 1.7 mol B = 6.5 mol Volume = 1 L Keq = 2.28 * 10-4 [A] = 1.7 M [B] = 6.5 M
Q: Consider an isothermal column of an ideal gas at 25oC. What must be the molar mass of this gas be if...
A:
Q: What is the equilibrium concentration (in M to three decimal places) of HBr for the following reacti...
A: Given, H2(g) + Br2(g) ⇌ 2HBr(g) Equilibrium constant (Keq) at 25°C = 0.000351 Initial concentration...
Q: In Fehling's reaction heat is ? a)Reducer b) Catalyser c)Substrate explain the answer
A:
Q: Measurements show that the enthalpy of a mixture of gaseous reactants decreases by 349. kJ during a ...
A:
Q: Kiko wants to determine the concentration of an unknown solution which produces a green solution. Ho...
A: A question based on analytical process that is to be accomplished.
Q: 6. Why do we need to precipitate from a hot solution? 7. Why is the precipitating agent added all at...
A: Since you have posted multiple questions, the answer for first two question is given below. Kindly r...
Q: DIRECTIONS: Read and analyze each question then choose the letter of the correct answer. Write the ...
A: Vapour pressure is the pressure of vapours on the surface of liquid when they are in equilibrium sta...
Q: er. Br N- OEt N- OE
A:
Q: 3. Predict the products for the following reactions, indicate major and minor where applicable. HNO,...
A: Given reaction is : Predict the major and minor product of the reaction = ?
Q: Consider the following reaction: A (s) B (g). The.molar heat capcities of A (s) and B (g) are 55 and...
A:
Q: Boron and sulfur form two binary compounds. The first contains 0.2247 g B/1.000 g S, and the second ...
A: Atomic Mass of B - 10.811 u Atomic Mass of S - 32.065 u Given, For the second compound 0.1348 g B/1....
Q: please explain each then give answer
A: The solution will be basic if the salt dissolved has a conjugate base derived from a weak acid.
Q: Directions: True or False: Write the word "HEAT" if the statement is CORRECT or “COOL" if the statem...
A: Correct and incorrect statements are given in the following step.
Q: Complete the mechanism for the formation of the major species at equilibrium for the reaction of 3-m...
A: Given,
Q: In reverse phase chromatography, the stationary phase is ___________ and the mobile phase is _______...
A: Answer of the question given below,
Q: The metals industry was a major source of air pollution years ago. One common process involved "roas...
A: Given the reaction is, 2PbS+3O2---->2PbO+2SO2 Mole of PbS is 9.53mol. We have to calculate mole o...
Q: 18. What is the molality of a solution of chlorine and water is 0.0362 m. This solution contains 350...
A:
Q: Provide the answer if the compounds will react. 1. Which compound/s will react with Br2 in water un...
A: A substitution reaction is a reaction where one group is substituted for another group in a single s...
Q: The following mechanism has been proposed for the conversion of tert-butyl bromide to tert-butyl alc...
A:
Q: [2] Write a chemical equation for an equilibrium system that would lead to the following expressions...
A: Answer - (a) According to the question - The equilibrium constant( K ) expression as follows - The ...
Q: 1. If 15.4 g of Cu react with excess nitric acid, how many grams of nitrogen monoxide are produced? ...
A: To find grams of nitrogen monoxide formed, • Convert the given mass of Cu to moles. • Find out the...
Q: LiN(-Pr)2 -78 °C, THF; NaH 25 °C, THF Br 'Br b. H3O* heat MeO OMe С. NaOEt ELOH, heat
A:
Q: Help!
A: Since you have posted multiple questions, the answer for first three question is given below. Kindly...
Q: Calculate the mass percent of Cu in CuCl2. (Mass of Cu= 63.55 g and Cl= 35.5 g) ii) Calculate the nu...
A: Mass percent is the percent ratio of mass of element in the compound to the total mass of the compou...
Q: determine the mass percent hydroxide, oh^-, in cu(h2o)4(oh)2
A:
Q: 7-55 trans-1-Bromo-2-methylcyclohexane yields the non-Zaitsev elimination product 3-methylcyclohexen...
A:
Q: One or more of the monomers below are needed to make the polymer to (CH) -(CH) Choose which one(s). ...
A:
Q: What is the general equation that represents a single displacement reaction?
A: Redox reactions are those in which both oxidation and reduction occurs simultaneously.
Q: Differentiate molecular spectra and atomic spectra
A:
Q: 6. Provide the complete mechanism (arrow, lone pairs, formal charges) for the following transformati...
A: Protonation of alcoholic oxygen Loss of leaving group Carbocation rearrangement for the formation ...
Q: Current Attempt in Progress When 5.64 g of a solid mixture composed of NH,Cl and CaCl, was dissolved...
A:
Q: Question/Problem: What is the % composition of potassium chlorate (KCIO,) in a mixture of potassium ...
A: The percent composition can be calculated as shown below. Percent composition=Mmass of simpler eleme...
Q: Determine the OH- and H+ concentration and calculate the pH, pOH of a solution that is 0.70 M NH3 so...
A: pH of a solution is a measure of acidity of a solution. pH less than 7 indicate that solution is aci...
Q: Ammonia gas can be prepared by the reaction of a metal oxide such as calcium oxide with ammonium chl...
A: Answer: Limiting reagent is the reactant that exhaust first in the reaction and decides the yield of...
Q: Unsymmetrical bond breaking is a mechanism for electrophilic or nucleophilic substitution reactions....
A: A covalent bond is formed by sharing of electrons between the atoms. When the covalent bond breaks, ...
Q: hypothetical metal with an atomic radius of 0.171 nm has an orthorhombic unit cell for which the a, ...
A: Atomic Radius of metal = 0.171 nm
Q: change in the internal energy
A:
Q: How much energy (in kJ) is required to heat 17.73 g of Au(l) from 2254.1 K to 3080.2 K (this substan...
A: Here we are required to find the heat energy required from gold from 2254.1K to 3080.2K
Q: What is the equilibrium concentration (in M to three decimal places) of NO2 for the following reacti...
A: K is the ratio of product of concentration of products raised to their stoichiometric coefficient t...
Q: The absorbance for a dye sample of unknown concentration is measured and the corrected absorbance is...
A: Given : y = 0.153 x + 0.043
Q: Consider the reaction: AC(9) + 2B(9) AB2 (8) + C9) What is the equilibrium expression for this react...
A:
Step by step
Solved in 2 steps with 2 images

- The following questions all are based on the data in this table: Substance Solvent Temp.,oC Solubility, g per 100 mL X heptane 20 23.5 X heptane 80 27.0 X 1-propanol 20 0.6 X 1-propanol 80 8.7 Y heptane 20 4.0 Y heptane 80 4.0 Y 1-propanol 20 22.0 Y 1-propanol 80 31.0…Table 1. Miscibility of Coconut Oil and Solubility of Cholesterol Solvents Coconut Oil Cholesterol Water Ethanol Chloroform Ether Guide Questions: At which solvents are coconut oil and cholesterol miscible/soluble? Explain why. ________________________________________________________________________________________________________________________________________ During washing oily dishes, it is not enough to just use water. We need to use soap. Explain the function so soap in this situation. ________________________________________________________________________________________________________________________________________ Table 2. Spot Test Substance Observation Inference (Volatile or Non-volatile) Coconut oil Glycerol Ethanol Ether Guide Question: What does it mean when something is volatile?…if ethanol (a liquid hydrocarbon) and chloroform (trichloro methane) are each shaken with caustic soda, which one of them is likely to contain the highest concentration of caustic soda and which one will contain the least
- Table 1: Temp vs. solubility data of ammonium chloride. Test tube g NH4Cl/10 mL H2O Crystallization temp. (ºC) 1 1.0 g/10 mL H2O 10 oC 2 1.8 g/10 mL H2O 20 oC 3 2.9 g/10 mL H2O 40 oC 4 3.8 g/10 mL H2O 65 oC 5 4.4 g/10 mL H2O 95 oC Use the table to plot a solubility curve for ammonium chloride on the graph provided below. For the best fit line, use a smooth curve. "Mass of Solute per 100 mL of H2O," should be written on the y-axis (use increments of 2g for every box). "Temperature (°C)" should be written on the x-axis (use increments of 10 °C for every 2 boxes).Tc-99m sulfur colloid is to be reconstituted with 75 mCi Tc-99m pertechnetate, to be drawn from an eluate with activity concentration of 325 mCi/mL. You will add constituents totaling 2.5 mL to the kit. What volume of eluate and of normal saline should be added to reach 5 mL final volume?Compare picture I and II and choose: A = If the indicated property is greater in picture I than in II B = If the indicated property is less in picture I than in II C = If the indicated property is equal in both picture D = If no comparison can be made due to insufficient information solubility in NaOH
- The colloidal particles has tenth ofmm diameter and easily filtered tru or falseDetermine the solubility of coconut oil (one drop) of the following solvents SOLVENT SOLUBILITY (SOLUBLE/INSOLUBLE) Water Conc. Hydrochloric acid Conc. NaOH (Sodium hydroxide) Alcohol ChloroformSolubility of solids in liquids. In the assignment, you need to elaborate the following points: world limit.(1000-1200) * Effect of Temperature. * Modification of molecular structure. * pH. * Common ion effect.
- Table sugar table salt water water with fully dissolved salt water with fully dissolved suragUsing the Solubility Data for Report below answer the question: Compound Cold water Solubility Hot Water Solubility Benzoic Acid 0.42 g per 100mL of water 5.79 g per 100mL of water Impurity 0.61 g per 100mL of water 4.62 g per 100mL of water An impure sample contains 1.14 g of impurities and 5.14 g of benzoic acid. The sample is dissloved in 100mL of water and heated. The solution is then cooled. How many grams of impurity will crystalize when the solution has cooled?A solute readily dissolves in a solvent. It means that the solute-solvent interaction is greater than the solvent-solvent interaction. Question options: True False

