Q: Circle the compound that would have a larger heat of hydrogenation. d) Explain why you circled that…
A:
Q: Classify each transformation as substitution, elimination, or addition.
A: The substitution reactions involve the substitution or replacement of an atom or group of atoms in a…
Q: stereogenic centre
A:
Q: Which compound does not have a conjugated system?
A: The solution is given below -
Q: Locate ONE (1) stereogenic centre in each compound. With detailed working steps, draw and name the S…
A: ( iii ) Stereogenic centre : This is the carbon center , which is sp3 hybridised and is attached to…
Q: Draw the products of attached reaction, and include the stereochemistry at any stereogenic center in…
A: The given primary alkyl tosylate reacts with the strong, unhindered nucleophile cyanide ion via SN2…
Q: Label each heterocycle as aromatic, antiaromatic, or not aromatic.
A: The aromaticity of a molecule is governed by the following factors: Cyclic Planar Delocalization of…
Q: Which box contains a heterocyclic ring?
A: Homocyclic ring which is contains all are same elements. In organic compounds the cyclic ring is…
Q: Draw the product(s) produced (only constitutional isomers, disregard stereoisomers) when the…
A:
Q: Draw the products of attached reaction, and indicate the stereochemistry where appropriate.
A: The given starting material is a fused bicyclic compound in which epoxide is fused with cyclohexane…
Q: # of stereoisomeric products?
A: The products are given below -
Q: Please draw the mechanism for this transformation using curved arrows. CI Na OCH3 CH;OH
A: At first the MeO^- abstract the alpha-H from -CO-CH3 to give the corresponding anion which displace…
Q: Draw the products of attached reaction, and indicate the stereochemistry where appropriate.
A:
Q: Draw the products of attached reaction, and indicate the stereochemistry at any stereogenic center.
A: In this reaction, first alcohol is transformed to alkyl tosylate along with the retention of…
Q: Which reagent(s) complete this transformation?
A: .When secondary alcohol react with oxidising agent then convert into ketone . .Reactant name is 2…
Q: Click the "draw structure" button to launch the drawing utility. What product(s) (excluding…
A:
Q: Click the "draw structure" button to launch the drawing utility. Draw a product formed when the…
A: The reagent i. BH3 ; ii . H2O2, OH-, H2O is used for hyration of alkene where OH adds to more…
Q: Can you help answer this problem and elaborate the steps clearly and show any reagent and…
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: show an arrow pushing mechanism for the reaction between α-phellandrene and maleic anhydride.…
A:
Q: Draw the products of attached reaction, and indicate the stereochemistry where appropriate.
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: Draw the products of attached reaction, indicating the stereochemistry around any stereogenic…
A: 1-D-butanol reacts with HI to give 1-D-iodobutane
Q: Draw all stereoisomers formed in attached reaction.
A: The given reaction is a Wittig reaction. This is a method to convert aldehydes or ketones to…
Q: Labeling Stereogenic Centers with R or S ? Define ?
A: To explain the labeling R and S on the stereogenic Centers.
Q: Which carbocation is destabilized by a substituent?
A: Carbocation is species in which carbon is carrying positive charge. Carbon in carbocation is sp2…
Q: Draw a stepwise mechanism for the attached reaction.
A: In an organic reaction, a nucleophile attacks an electrophile.
Q: Draw the products of attached reaction, and indicate the stereochemistry where appropriate.
A: Organic functional groups can be transformed to another by using appropriate reagents and suitable…
Q: Draw a meso compound for the attached molecules.
A: Meso compounds are the achiral compounds containing a tetrahedral stereogenic center. The…
Q: Use any reagents necessary to complete the folowing transformations. a. b. C. d. OH
A: Applying concept of organic synthesis and reagent.
Q: What type of carbocation is shown? (+)
A:
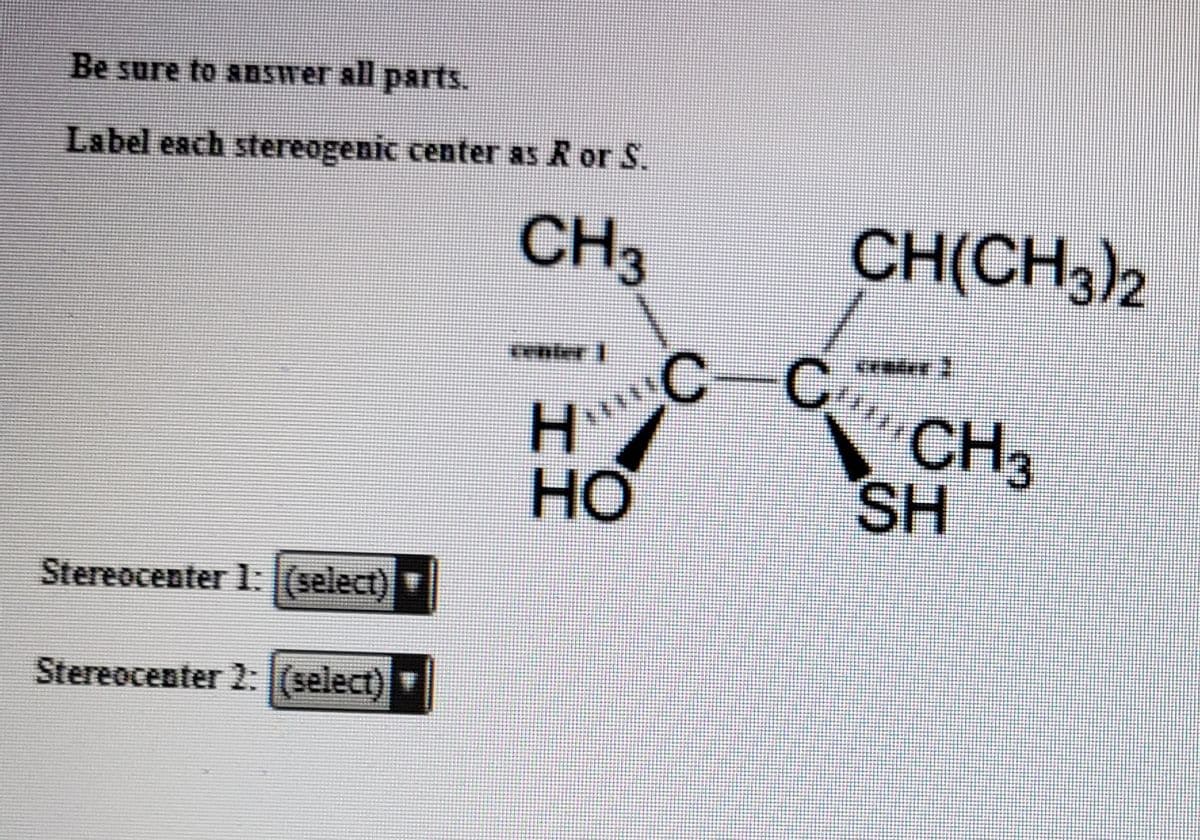
Q: Label the stereogenic carbons as S or R
A: For R and S configuration, Priority order is given as :- Hight the atomic number of attached…
Q: Draw a meso compound for the attached molecules.
A: Meso compounds are achiral compounds containing tetrahedral stereogenic center . Stereogenic center…
Q: reagents you deem necessary. Be sure vour synthesis vields the correct stereochemistry
A:
Q: ion Completion Status: 5). Indicate by an asterisk (*) all stereogenic center(s) in the compound A…
A: Solution: A stereocenter or stereogenic center is any point in a given compound, though not…
Q: Draw enol tautomer(s) for each attached compound. Ignore stereoisomers.
A: Introduction: Those compounds which the same molecular formula but differ in the position of protons…
Q: Draw the products of attached reaction, and include the stereochemistry at any stereogenic center in…
A: Given,
Q: Draw the products of attached reaction, indicating the stereochemistry around any stereogenic…
A: The stereogenic center is the atom of a molecule which will give two different isomer by exchanging…
Q: KOH, CH3CH2OH reflux Please sele Draw Michael Adduct KOH, CH3CH2OH reflux Draw Robinson Product
A: Given ; structure of reactant.
Q: R, S, or achiral
A:
Q: Please label any nucleophilic centers or electrophilic centers.
A: The nucleophilic center is the electron-rich center. The electrophilic center is the…
Q: Draw the products of attached reaction carried out under high-dilution conditions. Indicate the…
A: The products of attached reaction carried out under high-dilution conditions and the…
Q: Draw the products of attached reaction, indicating the stereochemistry around any stereogenic…
A: This reaction follows SN1 mechanism. In SN1 mechanism firstly leaving group leaves and carbocation…
Q: Draw the major product for the reaction, showing the stereochemistry by using wedge bonds. The…
A: When alkene reacts with Cl2 and CH3CH2OH, it gives haloether as the product.
Q: What type of carbocation is shown?
A: As we can clearly see that the positive charge (cation) containing carbon is attached to two carbon…
Q: Label the stereogenic center in attached compound as R or S.
A: The stereochemistry of the stereogenic centre as R or S can be defined by assigning the priorities…
Q: Label each heterocycle as aromatic, antiaromatic, or not aromatic.
A: The aromaticity of a molecule is governed by the following factors: Cyclic Planar Delocalization of…
Q: Draw a stepwise mechanism for the attached reaction.
A: The reaction forms a carbocation intermediate, which undergoes rearrangement to give alkene as the…

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Stereoisomers differ from each other in what respect? A.) Composition B.) Constitution C.) Configuration D.) Steric hindrance E.) NoneMake sure to answer the quesi to chem below Using the priority rule, identify the configuration at each chiral center and the configuration of the double bond: For a chiral center please type EXACTLY one of the following: R or S or neither if it is not a chiral center. For a double bond please type EXACTLY one of the following: E or Z or neither if the double bond has no stereochemistry.Draw all possible configurational stereoisomers for the given molecule below. Designate the R/S on the stereogenic centers and E/Z on the C=C

