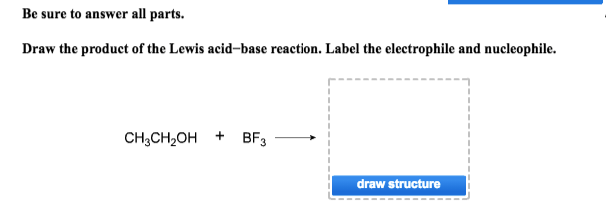
Be sure to answer all parts. Draw the product of the Lewis acid-base reaction. Label the electrophile and nucleophile. CH3CH₂OH + BF3 draw structure
Q: What is the major organic product of the following reaction? Br of CH3ONa methanol heat Draw the…
A: When the polar protic solvent solvent with strong base is provided then E1 elimination reaction…
Q: MgBr 1) BH3. THF 2) H₂O2, NaOH OH OH 2) H₂O excess Na₂Cr₂O7 H₂SO4, H₂O SOCI₂
A: For the above given reactions ; 1) Grignard reagent reacts with carbonyl compound to produce an…
Q: 2. Answer the following: a. What are the possible quantum numbers for an electron in a 4d orbital?…
A: -Principal quantum number (n): Since we are dealing with the 4d orbital, the value of n would be 4.…
Q: How many milliliters of 0.398 M Li2S are needed to react with 15.00 mL of 0.850 M AgNO3? Li2S(aq) +…
A: Given,The reaction:Li2S(aq) + 2 AgNO3(aq) → 2 LiNO3(aq) + Ag2S(s) Molarity of Li2S=0.398 MMolarity…
Q: Required information An ideal gas is contained in a piston-cylinder device and undergoes a power…
A:
Q: What is the pH of 10.00 M of maelic acid where its Ka1 is 1.3 x 10-2 and Ka2 is 5.9 X 10-7? pH =…
A:
Q: For a certain chemical reaction, the standard Gibbs free energy of eaction at 10.0 °c is 106. kJ.…
A: To calculate the equilibrium constant (K) for a chemical reaction using the standard Gibbs free…
Q: SYNTHESIS! Devise a multistep syntheses the same target compound. Limitations: All carbons in the…
A: The multistep synthesis is the reaction which used more than 2 steps. For given reaction we can use…
Q: ou prepare a solution that contains 62.51 g of ethylene glycol (MW 62.07 g/mol) and 790.8 g of water…
A: The depression in the freezing point (∆Tf) is given by the formula, ∆Tf=Tsolvent-Tsolution=iKfm i…
Q: How many milliliters of a 12% (v/v) propyl alcohol solution would you need to obtain 4.5 mL of…
A:
Q: Which statement describes energy in a nuclear reaction? O Energy is destroyed as the nucleus of an…
A: Nuclear reactions: Those reaction in which two or more nucleus combine together to give a large…
Q: Drag each of the following substances to the type of solid structure it would form. 2. 5. NEW…
A: Introduction:Different substances can form various types of solid structures based on their…
Q: What is the molecular shape of CH4 ? tetrahedral O angular O trigonal pyramidal O trigonal planar…
A:
Q: lease calculate the amount expected for the epoxyproduct. Name Molecula Weight Amount Used…
A: We are given mmol and equivalents of the reagents and we have to find the expected amount for the…
Q: Suppose an EPA chemist tests a 250. mL sample of groundwater known to be contaminated with iron(III)…
A: Silver chloride(AgCl) is insoluble in water and forms white precipitate. Using this feature, AgNO3…
Q: Draw the organic products formed in each reaction. [1] NaH [2] | [1] NaNH, [2] Br CI
A: A question based on reactions of alkynes. Two reaction conditions are given for a terminal alkyne…
Q: Provide the IUPAC name the following compounds Cl,CCH,CHO CH3COCH(CH3)2 CH, 0 11 CH₂-CH-C-CH₂-CH₂OH
A: Detail
Q: What is the chemical formula for the compound formed between sodium and fluorine? formula: What is…
A: Given,The compound formed between sodium and fluorine.The compound formed between sodium and…
Q: What is the change in potential energy if the distance separating the electron and proton is…
A:
Q: tive Follow-Up Question for Group Work 20.142 Cl₂(g) + H₂O₂(aq) → Cl(aq) + O2(g) (acidic) Match the…
A:
Q: For the reaction LiOH(s) ⇋ Li+(aq) + OH-(aq), Keq = 4.6 x 10^-3 and [OH-] at equilibrium is 0.042 M.…
A:
Q: In the context of environmental medicinal chemistry, what are the challenges associated with…
A: Developing effective pharmaceuticals that simultaneously address human health needs and minimize…
Q: What is the product of this oxidation? H3C a A b B с H3C CH3 H3C- CH3 Н.С. CH3 FAPE H3C e с d D H3C_…
A: Bleach is a general reagent used for oxidation of alcohols. It oxidises primary alcohol to aldehyde…
Q: What is the Ksp expression for the ionic compound Li2CO3? A. Ksp = [Li+][CO32-] B. Ksp =…
A: The dissolution equilibrium for Li2CO3: Li2CO3(s) ⇌ 2Li+(aq) + CO32-(aq)
Q: Rank the following compounds in order of increasing reactivity with the fe molecule A? O III < II…
A:
Q: 3. Describe Lewis acid and base. Specify following species as Lewis Acid or Base with explanation.…
A: The question is based on the concept of chemical equilibrium. we need to identify Lewis acid and…
Q: Which of the following structures is an ion with a single negative charge? CH₂ 11 H₂C-N-H A) B) C)…
A:
Q: Predict the ground-state electron configuration of each ion. Use the abbreviated noble gas notation.…
A: The electronic configuration is symbolic notation of the manner in which the electrons are…
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: Compounds: AgCl, Ca3(PO4)2, Ba(OH)2 To Determine: Does solubility change with pH pH at which…
Q: There are 103.0 g of chlorine gas (Cl₂) and 36.0 g of helium (He) mixed in a container with a total…
A:
Q: 2. Neglecting any effect caused by volume changes upon addition of sodium hydroxide to a dilute…
A: Ionic strength is described as the measurement of the ions that appear in the solution. The ionic…
Q: What is the smallest whole-number coefficient for H₂S when the equation H₂S + MnO₄⁻ → Mn²⁺ + SO₄²⁻…
A: Recall the given reaction, H2S + MnO4- → Mn2+ + SO42- We have…
Q: Which of the following statements is false when referring to the oxidation state of carbon? a)…
A: Oxidation: It involves the addition of oxygen or removal of hydrogen. Oxidation also involves the…
Q: Consider the following reaction: 2 SO2(g) + O2(g) ⇋ 2 SO3(g); Keq = 4.34 at 600°C An equilibrium…
A:
Q: Consider the following equilibrium: 2NO₂ (g) N₂O4 (g) - AG=- = -5.4 kJ Now suppose a reaction vessel…
A: Reaction : 2 NO2(g) ⇌ N2O4(g) ∆G°= -5.4 kJ
Q: Using the data in the tables and Coulomb's law, calculate the energy change for this reaction (per…
A:
Q: 0 F: ROOR FIND PRODUCT 40 Br tr Brz A Bv AX
A: Br2 and ROOR are good free radical reaction initiators. In presence of high temperature, they…
Q: H Pocl3 DMF H2O NaOH HC=NM₂₂ HC=0 N H H
A: This is example of Vilsmeire Hack reaction of indole. In this reaction indole reacts with dimethyl…
Q: What happens to the kinetic energy of Substance X at 3.5 atm as the temperature is decreased in two…
A:
Q: concentrated phosphoric acid solution is 78.9% H3PO4 by mass and has a density of 1.54 g/mL at 25°C.…
A: Given -> Concentration of H3PO4 = 78.9% by mass Density= 1.54 g/ml T = 25°C
Q: Pls help ASAP
A: Solubility is the maximum amount of solute that can dissolve in a solvent at the given temperature.…
Q: 5 A 22.02-mL solution containing 1.615 g Mg(NO3)2 is mixed with a 28.64-mL solution containing 1.073…
A:
Q: Bond Bond energy (kJ/mol) T HH Average Bond Energies (per mole of bonds) C-H C-C C-C 0-H 2528 kJ/mol…
A: Bond energy of a molecule = Sum of the bond energy of each bond
Q: Determine the geometry around all second-row elements in each compound. :0: a. CH3-C-CH3 b.…
A: A question based on molecules. Two sets of molecules are given that are to be identified for bond…
Q: Show the Claison product and then the reaction mechanism when (CH3)3CCO2CH3 reacts with CH₂CH₂CO₂CH3…
A: As per our guidelines We are supposed to answer only the first question, hence kindly repost the…
Q: a) Write the condensed structural formula for 2-ethyl-1,3-hexadiene b) Draw the structural formula…
A: A question based on introduction to organic chemistry. 3 subdivision questions are given that are to…
Q: 1. Consider the following equilibrium: 2NO() + Cl2) 2NOCI () AH = -77.07 kJ b) Chlorine gas is green…
A: Le chatelier's principle states that the equilibrium shifts in a direction that nullifies the…
Q: 1. Calculate the energy (in kJ) required to heat 0.891 grams of water from -26.9 ℃ to 21.6 ℃. 2.…
A: Specific heat is the amount of heat needed to increase the temperature of 1 gm of any matter by 1…
Q: 3. In the following reaction, indicate any conjugate AB pairs. HSO4 + HCO3 SO4²- + H₂CO3 →>>
A: conjugate acid–base pair consists of two substances that differ only by the presence of a proton…
Q: 1. Assign formal charges to the elements in each of the structures below. Note: Count oxygen atoms…
A: In a molecule, an atom is given a formal charge (FC), which is determined by the assumption that…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- What will be the product of the following reactions? Draw the Lewis-acid base reaction (including the arrows indicating the movement of electron pairs). Label which molecule is the nucleophile and which is the electrophile.Draw the product of the reaction where methoxide behaves as a base and also as a nucleophile.2. Write down the specific form in which bromine (other than molecular bromine) could act as (i) an electrophile : ………………………………..(ii) a free radical : …………………………………(iii) a nucleophile : ………………………………….
- In the following reaction, which chemical species is acting like a nucleophile?How does this mechanism work if OH- is a nucleophile? Would the electrons from the bond be transferred to oxygen after the nucleophile attacks the electrophile? Or is it a simultaneous process?Identify Electrophile & Nucleophile and know the difference How to identify them in a chemical reaction
- Define the following terms: a. nucleophile b. electrophile c. leaving group d. addition reaction e. anhydridePropose a mechanism for the following reaction (remember to use curved arrows to show the movement of electrons from the nucleophile to the electrophile):CHOOSE THE LETTER OF THE CORRECT ANSWER. Which atom in the reaction mechanism is the nucleophile?* Reaction mechanisms of organic molecules (alcohol to carboxylic acid) a. Chloride anion b. Hydroxyl group c. Carboxyl carbon d. Methyl group

