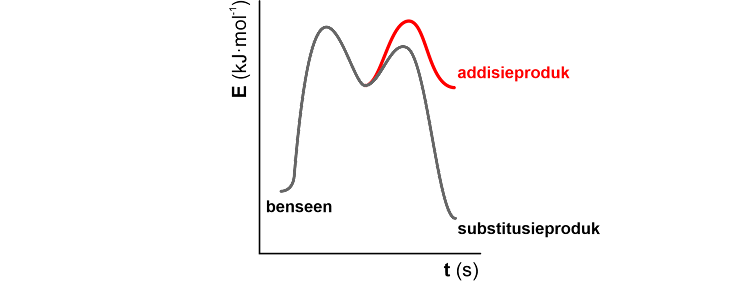
Benzene has pi bonds and should in theory undergo additional reactions such as alkenes. However, we know that benzene only participates in substitution reactions. Figure 16 compares the energy diagrams for the possible addition reactions and substitution reactions of benzene. Mark the correct statements. a)Addition and substitution reactions of benzene proceed through the same reactive intermediate. b)The substitution reaction is faster than the addition reaction. c)Addition and substitution reactions of benzene are exothermic. Both addition and substitution reactions of benzene proceed through two transition states. d)The substitution product is more stable than the additive product due to the stability of the aromatic ring.
Benzene has pi bonds and should in theory undergo additional reactions such as
a)Addition and substitution reactions of benzene proceed through the same reactive intermediate.
b)The substitution reaction is faster than the addition reaction.
c)Addition and substitution reactions of benzene are exothermic. Both addition and substitution reactions of benzene proceed through two transition states.
d)The substitution product is more stable than the additive product due to the stability of the
Step by step
Solved in 2 steps with 2 images


