boite Teen 4 Based on Table F, which equation represents a saturated solution having the lowest concentration of CI ions? 1 NaCl(s) Na (aq) + Cl(aq) enangan tad Wotorigaorms I to noituloa sbiolrlo ribo2 otulib s arinted A Thaviozzib si obnolrlo muibo2 510m as noituloe ads to thiog grillod sdf basonic 2 AgCl(s) = Ag+ (aq) + Cl¯(aq) 3 NH4Cl(s) NH4 (aq) + Cl(aq)ni miongmiliod odi bre 292091omi aslonaq otulos to imum on sob imiog griliod orb bas 29250 g puloa to odmun T 29281 Ining grilled odt brusasaayoob aslornaq otuloz to n asennoah onion gnitiod odi bre 202coToob zaloinaq otuioz to 150mun T 4 KCl(s) K+ (aq) + Cl¯(aq) in (na) solution increases when
boite Teen 4 Based on Table F, which equation represents a saturated solution having the lowest concentration of CI ions? 1 NaCl(s) Na (aq) + Cl(aq) enangan tad Wotorigaorms I to noituloa sbiolrlo ribo2 otulib s arinted A Thaviozzib si obnolrlo muibo2 510m as noituloe ads to thiog grillod sdf basonic 2 AgCl(s) = Ag+ (aq) + Cl¯(aq) 3 NH4Cl(s) NH4 (aq) + Cl(aq)ni miongmiliod odi bre 292091omi aslonaq otulos to imum on sob imiog griliod orb bas 29250 g puloa to odmun T 29281 Ining grilled odt brusasaayoob aslornaq otuloz to n asennoah onion gnitiod odi bre 202coToob zaloinaq otuioz to 150mun T 4 KCl(s) K+ (aq) + Cl¯(aq) in (na) solution increases when
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter15: Complex Ion And Precipitation Equilibria
Section: Chapter Questions
Problem 71QAP: Consider the equilibrium curve for AgCl shown below. Which of the following statements about a...
Related questions
Question
100%

Transcribed Image Text:boite Haarle
ONLI
4
Based on Table F, which equation represents a saturated solution having the lowest concentration of CI ions? M
1 NaCl(s) Na (aq) + Cl(aq) anged for W 319rgzomma I 16 noitulos obriolrio ribos sulib s anatoosad A
Thavioezib si obnoldo muiboa 010m 28 aoituloe ads to iniog gnidiod sdi bus notule
2 AgCl(s) = Ag+ (aq) + Cl¯(aq)
3 NH4Cl(s) NH4 (aq) + Cl(aq) seponi mioq gailiod odi bre 29209 linq otuloa lo rodmum of 1
panonoob iniog gniliod orb bas 292679m 2015thng tuloa to odmun T
asentoni Ining guillod odt brs 20ass100b 20loinaq otuloz 10 15dmun sil
8528100b iniog gniliod odi bas asesorosb zoloining oiuioz to 150mun s
4 KCl(s) = K+ (aq) + Cl¯(aq)
(na) solution increases when
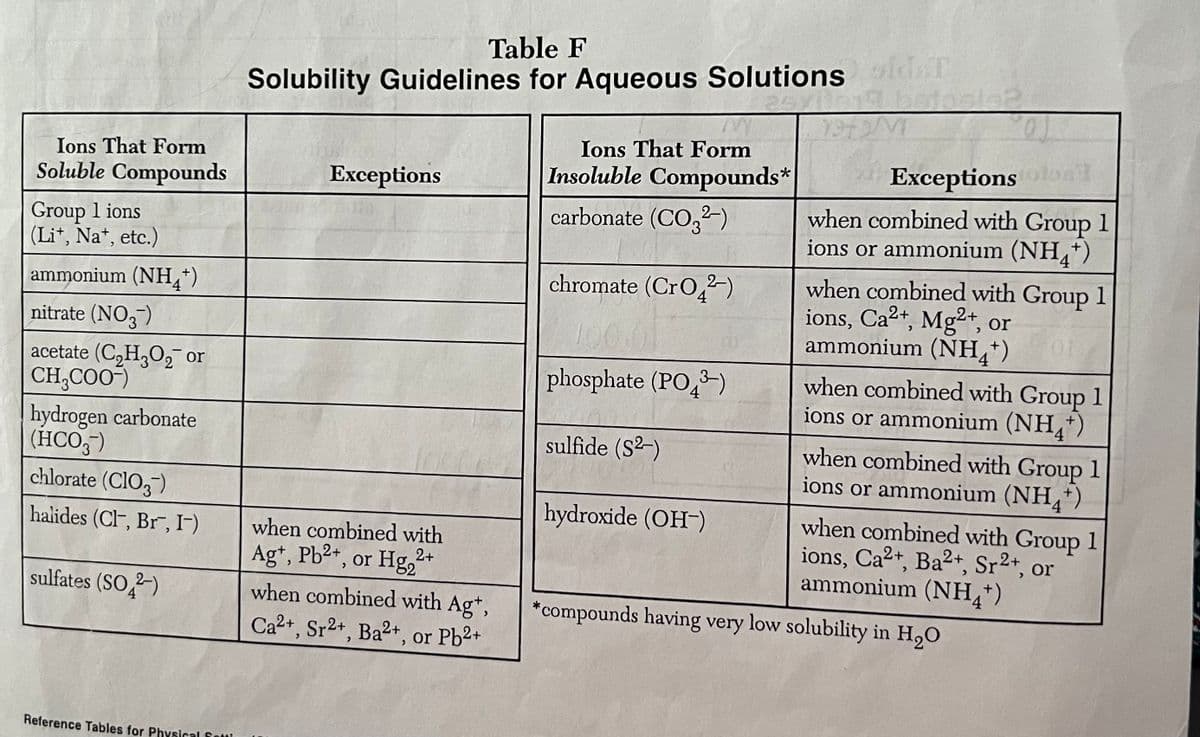
Transcribed Image Text:Table F
Solubility Guidelines for Aqueous Solutions old.T
25xila
M
Ions That Form
Insoluble Compounds*
Exceptions
carbonate (CO3²-)
Exceptions tolon
when combined with Group 1
ions or ammonium (NH4+)
chromate (CrO²-)
when combined with Group 1
ions, Ca2+, Mg2+, or
ammonium (NH4+)
10
phosphate (PO3)
sulfide (S²-)
when combined with Group 1
ions or ammonium (NH4+)
when combined with Group 1
ions or ammonium (NH4+)
when combined with Group 1
ions, Ca2+, Ba²+, Sr2+, 0
ammonium (NH4+)
hydroxide (OH-)
or
*compounds having very low solubility in H₂O
Ions That Form
Soluble Compounds
Group 1 ions
(Li+, Na+, etc.)
ammonium (NH4+)
nitrate (NO3-)
acetate (C₂H₂O₂ or
CH₂COO-)
hydrogen carbonate
(HCO3-)
chlorate (ClO3)
halides (Cl, Br, I-)
sulfates (SO2)
Reference Tables for Physical Sotti
when combined with
Ag+, Pb²+, or Hg₂²+
2+
when combined with Ag+,
Ca2+, Sr2+, Ba2+, or Pb2+
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning