bonds and should in theory undergo addition ver, we know that benzene only takes part in e 16 compares the energy diagrams for the possib itution reactions of benzene. Mark the correct
bonds and should in theory undergo addition ver, we know that benzene only takes part in e 16 compares the energy diagrams for the possib itution reactions of benzene. Mark the correct
Chemistry for Today: General, Organic, and Biochemistry
9th Edition
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Chapter12: Unsaturated Hydrocarbons
Section: Chapter Questions
Problem 12.67E
Related questions
Question
Transcribed Image Text:II
E (kJ mol")
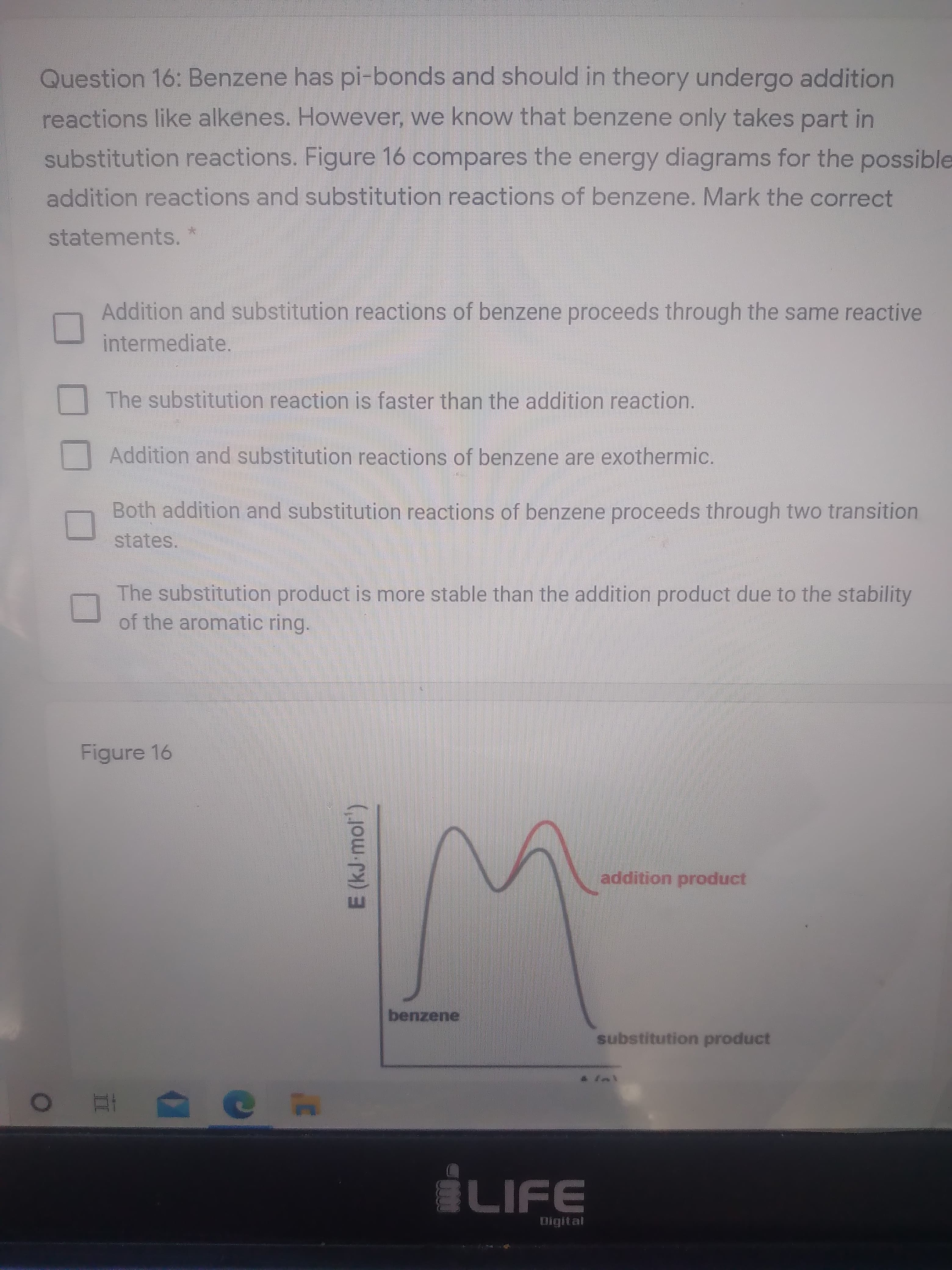
Question 16: Benzene has pi-bonds and should in theory undergo addition
reactions like alkenes. However, we know that benzene only takes part in
substitution reactions. Figure 16 compares the energy diagrams for the possible
addition reactions and substitution reactions of benzene. Mark the correct
statements.
Addition and substitution reactions of benzene proceeds through the same reactive
intermediate.
The substitution reaction is faster than the addition reaction.
Addition and substitution reactions of benzene are exothermic.
Both addition and substitution reactions of benzene proceeds through two transition
states.
The substitution product is more stable than the addition product due to the stability
of the aromatic ring.
Figure 16
addition product
benzene
substitution product
İLIFE
Digital
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning