Q: H. C-OH H C OH H C-OH H C-OH CH2OH CICICI
A: Cyclic anomers are formed when OH group of C5 attacks the C1 aldehyde group. As mentioned about beta…
Q: CH-(9) + H20(g)
A: The energy of a reaction as,
Q: 69. This compound is caled, CH2 CH3 HạC- N CH -N -CH OO OCH, Resin O Chlorophyll O steroid
A: Identify the given compound by its structure and check whether that substance is correct or not…
Q: CH3 OH SN1 CH3CH- -Cㅡ-CHㅡㅡCH,CH3 + HCI 1 + 2 CH3
A: Given
Q: 29) When C3H8 hydrocarbon combustion reaction occurs the products are: а. 4СО2 + ЗH20 b. ЗСО2 + 4H20…
A: Combustion of organic compound form CO2 and H2O. Combustion of propane forms 3CO2 and 4H2O
Q: 1) H2, Lindlar cat. 2) NBS, heat HBr (1 equiv) ....O. РОСs Me pyridine Me
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: OH CH3 II CH2CH3 НО HO- -H- ČO,H ČO2H II IV CH3 CO2H H- -HO- Но -H-
A: Fisher
Q: ethanol (C2H5OH) are present in 50.0 g-sample?
A:
Q: (a) NaCN (d) NEt H2SO4, 0°C dropwise EINH3 + (b) MEOH (exs.) (e) TSOH, toluene heat 1) Ph3P Br (c)…
A:
Q: Question attached
A: In the presence of strong acids, such as sulfuric acid and heat, alcohol dehydrates to an alkene.…
Q: 1) HO 2) CH3CH2CH2C1 AICI3 CI NaOH, 100 °C ? CH3 H3CT CH3 Ph C Br ? CH3 t-BUOH
A: The following reactions has to be completed
Q: 1. Na s-S Na NaOH / H20 NO2 HCI / H20 HO triethylene glycol 210 °C 2. 3.
A:
Q: 1) ? HO 2) CH3 CH2CH2C1 AICI3 CI- NaOH, 100 °C ? CH3 CH3 Ph H3C. Br ок O K* CH3 t-BUOH
A: Hey since you have posted a question with multiple sub-parts, we will solve first three sub-parts…
Q: acetone H,C но H,C . OH tien is HGl OH H,C
A: The SN2 reaction mechanism involves the nucleophilic substitution reaction of the leaving group…
Q: NaBHa, Oн a) Hg(OAc)2 p-TSCI b) KCN c) ► d) pyridine
A: See synthetic route below.
Q: CH3 H2C=CHCHCH2CO,CH3 (e) 1. LIAIHA 2. H30 CH,CO,COCH,, ? Pyridine (g) CONH2 (h) CO,H 1. LIAIHA 2.…
A:
Q: General formula for a carbohydrate is ---.
A: General formula of carbohydrate
Q: HF он H20 но (A) (В) Он он (C) (D)
A:
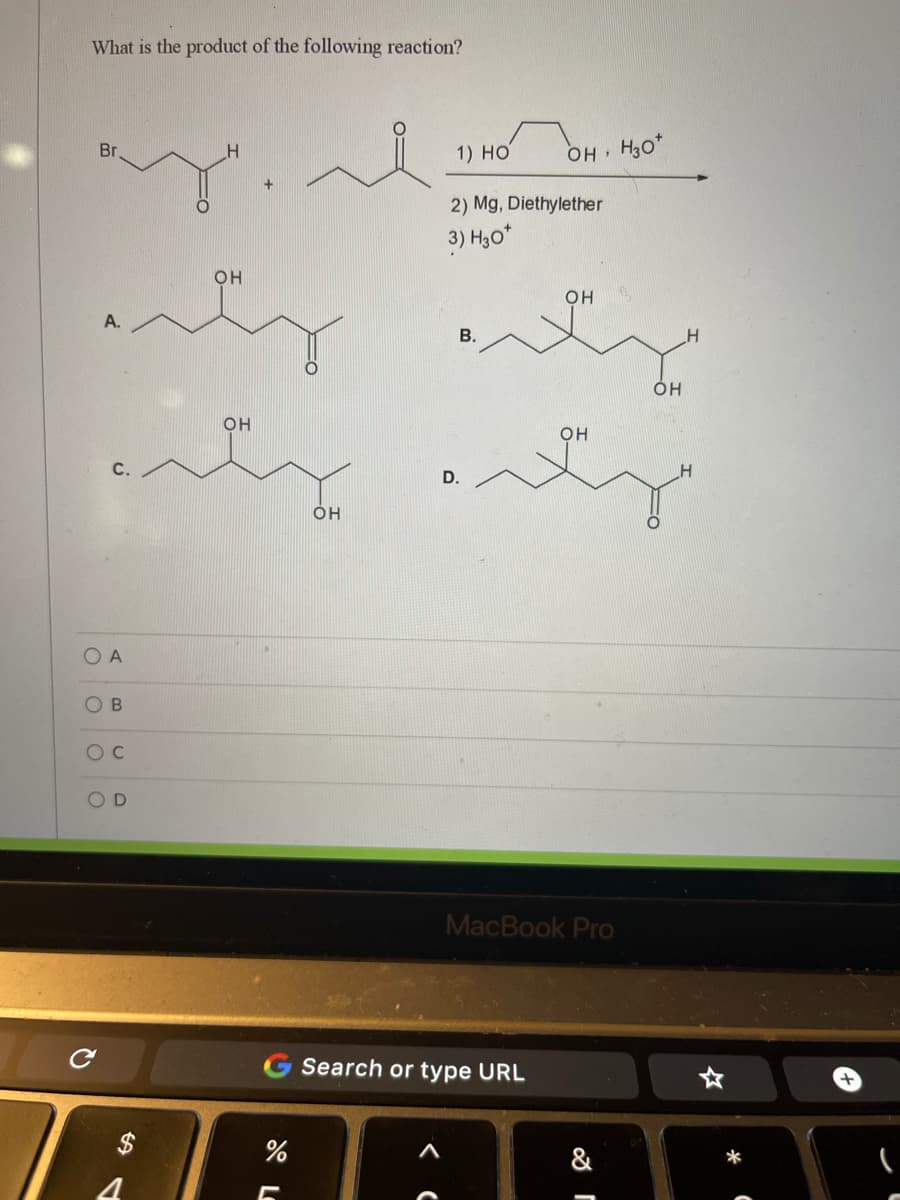
Q: 1) 2) H3O* BrMg' OH
A:
Q: So (J/molK) So Organic Substance AHC AG ΔΗρ AGº Substance (kJ/mol) (kJ/mol) (kJ/mol) (kJ/mol)…
A: I had filled all the values is table Using the formula ∆G= ∆H-T∆S
Q: 9. OH CrO3, H2SO4 acetone H. YZA AZY AZY YZA
A: The question is based on the concept of organic reactions. We have to identify the product formed…
Q: .O-H O-H NaHCO3 (exceso) + H;CO3 H20 O-H I. LO Na I. O-H
A: Given : To find the products for the given reaction.
Q: CH1206 (glucose) HCI NH3 H,SO4 O2 II || II IV V
A: Electrolyte is the substance which get dissociate when dissolved in solvent. Strong electrolyte is…
Q: 2. Complete the following reactions ( H2SO3 + Ba(OH)2 > a. b. C4H9OH + 02 > C. Br2 + YF3 >
A:
Q: जष्प क जप्छककnce +vuon oी क्रेतट 1- Q7Gmmdकीट लिमलnmर वीकने० लि किमट ललटकैवकांडन कैट बवेतीनीन्ता से…
A: Tertiary carbocation is mose stable carbocation dye to hyper conjugation and +I effect .
Q: OH OH (1) NaOH - (x) (2) H® Reactant x is : 7. (a) CH3CI (b) CH 2C12 (c) CHCI3 (d) CCl 4
A: Given reactant is phenol and product is salicylaldehyde. This is Reimer-Tiemann reaction.
Q: Draw a stepwise mechanism for each reaction.
A: SOLUTION:Step 1: The mechanism shown here applies to both acetal and hemiacetal formation 1)…
Q: C-H 30% КОн `CH2CH3 CN H,SO. HNO,
A: a. Potassium hydroxide also known as caustic potash. It is used in food items to maintain its pH and…
Q: 1) CH3CO3H 2) H30* OH ...OH ...OH HOH ...OH ОН ОН "OH ÕH A
A:
Q: -OH CrO3 H* HOCH,CH2OH H+
A:
Q: What is the relationship between the two compounds shown? HO HO HO HO- HO- HO- но- HỌ HO- wwwwwww
A: Constitutional isomers have different connectivity of groups. Enantiomers are non-superimposable…
Q: Draw structures for compounds A–F.
A: The given alcohol reaction with phosphorus trichloride in pyridine which gives chlorinated compound…
Q: 1) H*/H20 2) TSC//pyridine 3) CH3S
A:
Q: عالم من Me Me Me cat. H₂SO4 benzene Me Me
A: Here we have to write the mechanism of the following given reaction. The product can be obtained…
Q: Draw the product of each reaction.
A: The epoxide can be easily opened up by treating with a different reagent as it is a strained ring.…
Q: Complete the following reactions. OH e) KMn Oq Ho Hz C-0- CHz CH2 HI
A: The given reactions are represented as follows:
Q: CM2 ヤ (frace) methanot
A:
Q: Br NaCN A H30* SOC2 В 1. → D 2 1. LIAH4 2. H20 CH3OH, H* 1. DIBAH-H F 2. H20 1. LIAH4 2. H20…
A: We have to give the products B, D, F, and J.
Q: OH он 1. CI-TBS, py H;C. 13 2. NaN, DMSO HSO4, A 14 -CH3 но HO CH2 1. LDA -Et -Et 16 15 2. CHJ
A:
Q: HO, H20, A Product(s)?
A: We have given that Aldehyde reactant react with itself in presence of base to give two different…
Q: Br CH3 CH30H A(61) procincf major minor CH3OH (1) Hg COACla i) (2) NaBHý
A: The question is based on the concept of organic reactions. we have to identify the Product formed…
Q: H-C-H H HI
A: Hydrocarbons are the compounds consist of carbon and hydrogen. General formula of alkane = CnH2n+2
Q: a) CH3OH + H2S04 = ? o) CH3OH + NaNH, = ? :) CH3NH3+ Cl- + NaOH = ?
A: When acid release h+ in the solution and base gain h + from the acid then conjugate base and acid…
Q: 132.1 334.8 19 2.160 20 1.674 H Aromatic Ke 3 6. 8 7 4.25 1.35 1.00 2.35
A: 1HNMR spectrum identified number of chemically non-equivalent protons in molecule.
Q: 1) m-СРBА 2) МеO"/ MeOH Br2 .C=N Me-C Me CH3 МеОн gentle heat Br Me +
A: Concept: A) m-CBPA: it is used for epoxidation of Alkene. Further epoxide opening will be via SN2…
Q: (i) (1) BH3 THF B (ii) H2O/HO (ii) Br SH Br2 (i) POCI, HO E CH2CH3 Pyridine (iv) (i) R2BH (ii) H2O2,…
A: Four Organic chemistry problems, which are to be accomplished.
Q: OTBS PCC F) DCM, 25 °C 1) Hg(OAc)2. THF/H20 2) NABH4. Na OH TIPSOTI, pyridine H) DCM, 25 °C
A:
Q: CH,OH + CH3C C Nat?
A: The hydrogen bonded to the sp hybridized carbon becomes very acidic in nature. In the given problem…
Step by step
Solved in 2 steps with 2 images

- Complete the following reaction equations :(i) C6H5N2Cl + H3PO2 + H2O →(ii) C6H5NH2 + Br2 (aq) →I need help on letter 4E only. I drew the structural form already*there are organis questions can you solve e quesiton with mechanism e = 2-bromo-2-methyl hexane Keep questions specific and include all details
- wt of flask = 98.4669 wt of product = 20.7868 wt of flask and product = 119.2537 weight of water collected = 4.4 mL wt of water expected 4.5 mL percent yeild of ester = 71.58% 19.0 mL of 0.25 M n-propanol 18.5 mL propionic acid plz answer questions below(CH3)2C=CHCH2CH3 and Ozone (o)3 /Zn product?Organic Chemistry 1 Question. EA24. Please show why the answer is B. letter , and not D. letter . Thank you
- How many different alkenes can be formed from the haloalkane in Figure 20? Take stereoisomerism into account. * 1 2 3 4 This haloalkane will not form an elimination product.Answer all of question 1 pls 1.i. Give two examples each of Unsymmetrical alkenes and reagents.ii. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition productsWhich of the following is the most volatile? 1-ethoxypropane (MW = 88) 1-pentanol (MW = 88) 1- aminopentane (MW = 87) butanoic acid (MW = 88)
- can u please find the type/ name of Reaction occurs in each step along with name of products formed in each step.TRUE OR FALSE I. The reaction of BH3 with 2-methylbut-2-ene results in the attachment of a hydroxyl group, OH, on the third carbon since this carbon is less substituted. II. The reduction of an alkyne using a strong base such as LiNH2 gives an alkane when 2 equivalents of hydrogen gas is added. III. Hydrobromination of an alkene follows anti-Markovnikov’s rule in the presence of a peroxide.Which of the following statements about Figure 26 is not true? * A- The two products are formed by two competing chemical reactions. B- The alcohol is formed in the greatest amount. C- The alcohol is formed by an Sɴ1mechanism. D- The alkene is formed by an E1 mechanism. E- All of these statements are correct.

