Q: OH 1) NaOH 2) Etl 1) xs EtMgBr 2) H₂O+ ? ?
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Which of the following is a dominant feature of the outer membrane of the cell wall of gram negative…
A: The aim of this question is to show the dominant feature of the outer membrane of the cell wall of…
Q: 7. Use electronic configurations to account for the stability of the lanthanide ions Ce** and Eu²+.…
A: The electronic configuration of an atom gives the idea of the arrangement of electrons in the atomic…
Q: ● Which one is the product of the Grignard reaction between dimethyl oxalate and excess…
A: The objective of the question is to identify the product of the Grignard reaction.
Q: Which of the following salts will result in a basic solution when dissolved in water? a) NaCI b)…
A: The objective of this question is to determine which of the given salts gives a basic solution,…
Q: The compound in the picture O=C H₂C-0-C-CH₂(CH₂)11CH3 HC-0-C-CH₂(CH₂)13CH3 H_C–0–ệ—…
A: The aim of this question is to explain the properties which shown by given picture. The picture…
Q: Select reagents convert the following compounds: Reagents Available a. BH3, THF; followed by H₂O₂,…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: (b) (i) Ethyl acetoacetate D is a versatile reagent for the synthesis of a wide range of ketones.…
A: The objective of this question is to show the synthesis of E and F compound by using compound D. The…
Q: A closed container with a volume of 6.00 L holds 10.0 g of liquid helium at 25.0 K and enough air to…
A: The objective of the question is to determine the final pressure inside the container.It is given…
Q: 233Np decays by a-particle emission and has a half-life of 2.20 × 10 years. (1 Ci= 3.70 × 10¹0 3.70…
A: mass of sample = 0.230 ghalf-life of Np(t1/2) = 2.20106 yearsMolar mass of Np = 237 g/mol1 Ci =…
Q: CH2023 Q2 (b)
A: The main purpose of this question is to complete the given three reactions which show reduction and…
Q: (b) Describe in detail the growth of MgAl2O4 in the solid state, covering the nucleation and growth…
A: The purpose of this question is to explain the growth of MgAl2O4 in the solid state, covering the…
Q: 4. Give the IUPAC name for (a)
A: Since you have asked multiple questions, we will answer the first question(4) for you. If you want…
Q: Which of the following would not show a C-C signal in an IR spectrum? CI CI CI CI
A: IR active molecules are those in which dipole moment changes after absorption of light.
Q: 8) Write an electrophilic substitution using acetic acid, in a single functional group of the next…
A: The aim is to show the electrophilic Substitution by using acetic acid and other reactant is…
Q: Provide an IUPAC name for the structure shown. H ball & stick H H HH + labels H H H (Use cis/trans…
A:
Q: what functional group ch2ch3 is?
A: given- A functional group CH2CH3 is given.To find-we have to find what functional group CH2CH3 is.
Q: For the structures below, state whether the hydrogens marked Ha and H, are homotopic, enantiotopic…
A: A question based on introduction to organic chemistry. Three dimensional structures of two compounds…
Q: The following fictitious unbalanced Redox reaction is made using some of the 1001GRC teachers'…
A: The objective of the question is to show the oxidation numbers of each given species.
Q: Jord CI 3 Cl heat 10 Lindlar Catast Ha 결 F o=
A:
Q: What is the major product in the following reaction? HBr 40°C I) Br II) Br III) -Br IV) Br
A: Dienes have nucleophilic charater hence, it can give electrophilic addition reaction when reacts…
Q: If the pH of two acids, X and Y, are pH 1 and pH 2 respectively, which of the following is true? IX…
A: The given question revolves around the pH values of two acids, X and Y, where X has a pH of 1, and Y…
Q: The temperature of a sample of water changes from 1000 to 20°C when the sample absorbs 418 joules of…
A:
Q: Two nitro (NO₂) groups are chemically bonded to a patch of surface. They can't move to another…
A: The objective of the question is to determine the change in entropy.
Q: H₂C CH₂ CH₂ CH3 ZI CH₂ water acid
A: The objective of this question is to show the reaction of hydrolysis of amide by using water and…
Q: Given the following calibration data. The standard deviation (sc) for the concentration of the…
A: Sample Concentration(M) pCl-Unknown 4.9804*10-3…
Q: 5. What is the problem with the following reaction scheme and how could it be made to work? HO yo 1.…
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: 10) A solution concentration is 3.5 ppm and is made with a solute that has molar mass equal to…
A: Given concentration is 3.5 ppmMeans 3.5 gm solute present in 106 ml solution
Q: (+) NH3 Br Na OH
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Rewrite this measurement with a simpler unit, if possible. kg m 2 m m 3.4 Note: If you can simplify…
A: A unit of measurement is the magnitude of a quantity that is adopted by convention. It is used as a…
Q: . which is generally not a component of a potentiometric measurement? voltmeter salt bridge…
A: The objective of the question is to identify the component that is generally not a part of a…
Q: I have given you a condensed structure. You need to convert it to an accurate bond-line structure.…
A: The objective of this question is to show the isomerism by moving chlorine atom and other bond line…
Q: (H3C) ₂ со н H 11 CH (CH3)₂ Date : OCH₂ C6 Hz
A: The objective of this question is to show the mechanism of cyclization by using the hydrogen bromide…
Q: Synthesis from benzene, cyclopentadiene and necessary organic and inorganic reagents:
A: The main aim of this question is to synthesize a given molecule from the benzene, phthalic…
Q: Consider the following system at equilibrium where Kc = 1.20E-2 and AHO = 87.9 kJ/mol at 500 K. PCI5…
A: Le-chatelier's principle states that if a system in equilibrium is disturbed by an external change,…
Q: Which of the following statements is TRUE? Select ALL that apply. □logs (+) -logħ(4) Olog(xy) =…
A: The given question involves evaluating the truth of various logarithmic statements. Logarithmic…
Q: Which ion has emission at the lower frequency?
A: given-emission from the n = 5 state to the ground state from U +91 and B +4. to find-Which ion has…
Q: QUESTION 12 What is the molarity of a solution of 10% by mass cadmium sulfate, CdSO 4 (molar mass =…
A: We have to find the molarity of a solution of 10% by mass cadmium sulfate (CdSO4)(molar mass=…
Q: H3C H3C- H3C A -NH₂ (CH3CH2)3N D IZ B (CH3CH₂)2NH (CH3CH₂)3NH* E C
A: The question involves identifying a tertiary amine among several given options. A tertiary amine is…
Q: Question 7 A solution of acids is made by mixing 0.15 M HBr and 0.65 M HF (Ka (HF) = 6.3 x 10-4).…
A:
Q: Select the three possible products of bromination of the following hydrocarbon: Select one or more:…
A:
Q: KMnO (C6H₁00) + (C₂H40₂) hot + (co₂)
A: We know KMnO4 is strong oxidising agent. will oxidise double bond to ketone and aldehy."
Q: Name the alkenes below.
A: The objective is to determine the IUPAC names of the given wireframe structures of alkenes. Alkenes…
Q: Draw the molecular orbital (MO) electron diagram for the Li2 molecular ion. Be sure your diagram…
A: The objective of this question is to explain the molecular orbital diagram of Li22-.
Q: The orbital occupancies for the d orbitals of several complex ions are diagrammed below. [Select]…
A:
Q: The answer is C plese explain and show your work for better understanding Ć 22. Which of the…
A: The objective of the question is to identify which metals will act spontaneously as reducing agents…
Q: 2 By using the same alkene, how do you synthesize the following alcohols? Give the alkene and show…
A: Alcohols are formed from alkenes by the following methodsi. addition of water in the presence of…
Q: Draw the organic product of the Lewis acid-base reaction shown below. Include all lone pairs and…
A: " I'm sorry but i have to draw it on a paper, there's no way i can draw here or copy paste,this will…
Q: 29. What is the value of K, for H₂PO,1 A. 1.3 x 10-12 B. 6.2 x 10 1.6 x 10-7 75 x 10 C D.
A: The objective of this question is to find the kb value of H3PO4-. It is related to ionic…
Q: (Thanks for the help if you can break down the steps and provide drawings(needed) during the answer…
A: The objective of the question is to understand the heme environment in oxymyoglobin and how the…
plesa don't provide handwrittin solution..
What is name of the structure? Give explanation and steps to support your name.
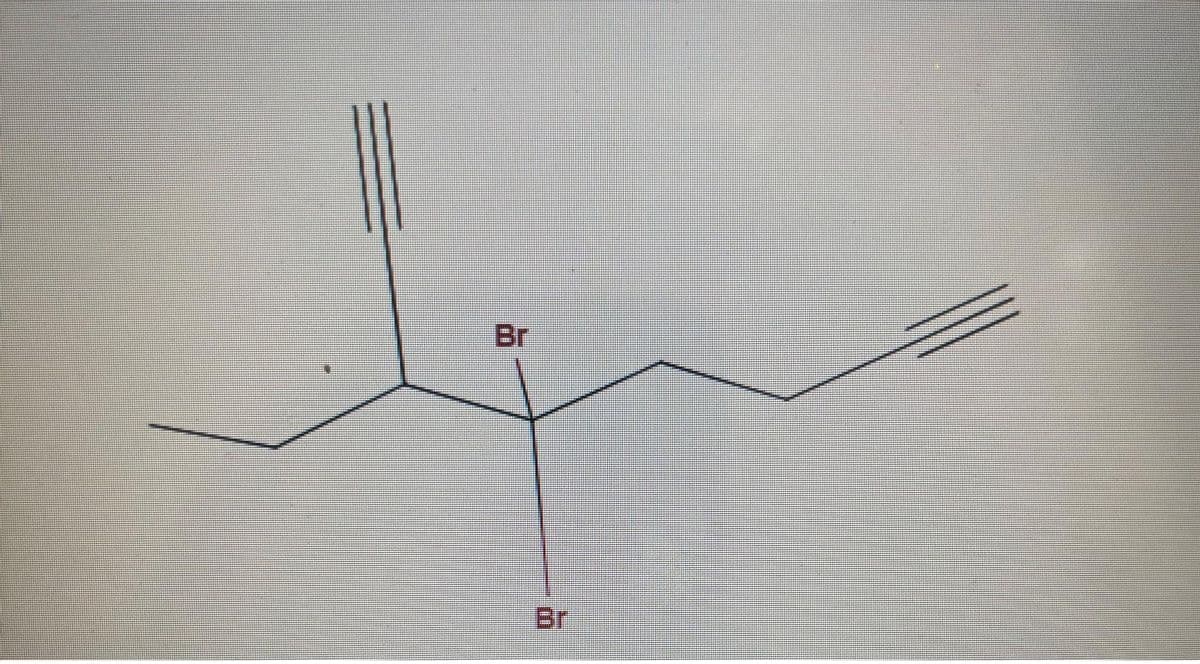
Step by step
Solved in 3 steps with 2 images


