Q: State Kohlrausch law of independent migration of ions. Why does the conductivity of a solution decre...
A: Kohlrausch law of independent migration of ions is introduced by the German chemist Friedrich Kohlra...
Q: 4. Determining electrolytic activity in a dilute solution. Electrolysis of dilute aqueous NaCl solut...
A: Electrolysis of dilute aqueous NaCl solution was carried out by passing 10 milliampere current. Th...
Q: What is 4a?
A: 4 a) The distance of the solvent front can be measured by a ruler. For this, match zero on your rule...
Q: The difference between the boiling points of the ICl (b.p. 97 ˚C; MA: 160 g / mol) and Br2 (b.p. 59 ...
A:
Q: Rank the compounds in each group in order of increasing reactivity in nucleophilic acyl substitution...
A: Carboxylic acid derivatives undergo nucleophilic acyl substitutions on the carbonyl groups. The firs...
Q: Calculate the quantities of lime and soda required to soften 25000 liter of water having following a...
A: Given, (in ppm) CaHCO32 = 4.86MgHCO32 = 7.3CaSO4 = 6.8MgCl2 = 5.7MgSO4 ...
Q: Problem 1: A radioactive material loses 75% of its mass in 16 days. What is its half-life?
A: All radioactive decays are first order reaction.
Q: Show how you could synthesize racemic ibuprofen starting with 1-bromo-4-isobutylbenzene.
A: Given, The structure of 1-Bromo-4-isobutyl benzene is shown below; The structure of ibuprofen is sh...
Q: Which of the following straight-chain organic compounds has the highest acidity? A. C2H4 B. C2H6 C....
A: more S%, more will be acidic in nature. so triple bond have maximum acidic in nature, more % s chara...
Q: Solve this question
A:
Q: sketch how the entropy of water changes as it is heated from -50 °C to 110 °C at sea level. Show the...
A: The entropy changes with the temperatures are shown in the figure below:
Q: What are the products of the following reaction?
A: Given reaction,
Q: For the following hypothetical equilibrium, A + 2 B ⇄ C, what is the value of the equilibrium consta...
A: The equilibrium constant for a reaction is expressed in terms of the ratio of the concentration of p...
Q: The reaction of WCl6 with Al at ~400 °C gives black crystals of a compound containing only tungsten ...
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the ra...
Q: Explain why a much better yield of primary amine is obtained from the reaction of an alkyl halide wi...
A: Alkyl halide reacts with sodium azide to form alkyl azide. Here the formed alkyl azide is not nucleo...
Q: Draw the products of attached reaction.
A: The given reaction results in the formation of resorcinol diglycidyl ether.
Q: The aqueous solution of hydrochloric acid (HCl) with a concentration of 1.12 M has a density of 1.19...
A:
Q: C. Arrange the following alcohols in increasing order of their reactivity toward acid- catalyzed deh...
A: C. Given alcohols,
Q: Identify the Bronsted-Lowry conjugate acid-base pairs in each of the following reactions: (a) H2CO3(...
A: An conjugate acid is a substance that donates proton and a conjugate base is a substance that can ac...
Q: A river is contaminated with 0.65 mg/L of dichloroethyl-ene (C₂H₂Cl₂). What is the concentration (in...
A: According to the Henry's Law: S=KH×P ...... (1) where S is the solubility (or conce...
Q: When reading the volume of water in a burette or graduated cylinder, it is best to read the volume f...
A: The burette is a long graduated glass tube with the markings to measure the volume of the liquid. It...
Q: Draw the general shape of a pH curve for the titration of 10 mL of 5.0 M NaNH2(NH2-is a strong base)...
A: (a) The given titration is acid-base titration, MHCl = 2MNaNH2 = 5VNaNH2 = 10 mL The balanced ...
Q: Suppose you are given two 1-L flasks and told that one containsa gas of molar mass 30, the other a g...
A: Ideal gas law states that- PV = nRT Where P- Pressure of the gas V- Volume occupied by the gas n- Mo...
Q: How many miles of sodium chloride can be produced if 1.2 mol Na reacts with .85 mol of cl2?
A:
Q: Explain in Isomerism in tartaric acid.
A: The isomerism of tartaric acid has to be given,
Q: By using valence bond theory and ligand field theory, prove [Fe(acac)3] is aparamagnetic high spin o...
A: Complexes having unpaired electrons will possess paramagnetic behavior and has come color whereas di...
Q: What would happen if MgCl2 were added to water? A It would dissolve B ...
A: General solubility guidelines for ionic compounds in water are given below
Q: Predict the product C of the following reaction below and briefly explain your answer.[PtCl3(C2H4)]¯...
A:
Q: Which of the following compounds is soluble in water? Select one: a. Pb(HCO,), b. MgS c. Ba,(PO2 d. ...
A: Welcome to bartleby !
Q: For PbI2(s) ⇋ Pb2+(aq) + 2 I−(aq), K = 1.4 × 10−8 at 25 °C and the standard Gibbs energy of formatio...
A: The standard Gibbs energy of formation of PbI2(aq) has to be calculated, The reaction is, PbI2(s) ⇋ ...
Q: It is very difficult to burn carbon in a deficiency of O2andproduce only CO; some CO₂ forms as well....
A:
Q: The following reaction is at equilibrium. N2(g)+ 3H2(g) ⇌ 2NH3(g); Ho= –92kJ Use the Le Chatelier’s ...
A: a) Addition of either H2 or N2 shifts the equilibrium towards right. b) If H2 is removed from the re...
Q: Determine the equilibrium constant for the system N2O4 2NO2 at25C given the equilibrium concentrati...
A:
Q: The Henry's law constant for O, is 1.3 x 10 3 M/atm at 25 °C. What mass of oxygen would be dissolved...
A:
Q: In each of the atoms and ion pairs below, mark which is greater. (8O, 9F, 26Fe, 33As, 34Se, 51Sb) a)...
A: A multiple choice question about atomic size, which is to be accomplished.
Q: In which case is the correct order of increasing the boiling points of the chemical species given be...
A: The increasing order of the boiling points of the following chemical species to be found. Xe, LiF, L...
Q: Draw a molecular orbital diagram to show that CH3¯ molecule is a -donor ligand.
A: Electrons in the highest occupied molecular orbital can be donated and hence CH3- act as donor legan...
Q: Lead (II) chloride, PbCl2, has Ksp = 1.6 x 10–5. (a) Determine the solubility of PbCl2 expressed in ...
A:
Q: What is a general characteristic of a Lewis acid? Of a Lewis base?
A: General characteristics of Lewis acid are discussed as follows, Lewis acid accepts pair of electr...
Q: Give the metric prefix that corresponds to each of the following: 1000 103 10-9 1,00...
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for ...
Q: Which of the following straight-chain organic compounds has the highest acidity? (Note: All numbers ...
A: Acidity of the given organic compounds is as follows.
Q: A mixture of N2, O2, and Ar has mole fractions of 0.25, 0.65, and 0.10, respectively. What is the pr...
A:
Q: Shapes of s ,p ,d ,f orbitals?
A: The orbital names s, p, d, and f stand for names given to groups of lines originally noted in the sp...
Q: One mole of an ideal gas will occupy a volume of 22.4 liters at STP (standard temperature and pressu...
A: 1 mole of any gas will occupy 22.4 L at STP conditions is called as gram molar volume. For gases 1 ...
Q: When cyanide ion, CN, reacts with hydrogen peroxide an oxygen atom is transferred to the ion to form...
A: Lewis structure of cyanate ion: The formal charge of Cyanate ion:
Q: Chemistry Question
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to ...
Q: Draw the mechanism for the following reactions. i. [PtCla]2 + NH3 → [PtCl3(NH3)] +Cl (Association me...
A: In the association mechanism, the binding of a ligand to the metal occurs. The ligand donates an ele...
Q: A vinegar solution contains 5.11% acetic acid. Its density is 1.007g/mL. What is its molarity?
A: The concentration of acetic acid in vinegar solution is = 5.11 % The density of the solution is = 1....
Q: Check the option that gives the correct one which is greater in each of the atom and ion pairs below...
A: Atomic size of an atom increases as we go down the group because electrons are added to different sh...
Q: Consider the following equilibrium: N2O4(g) ⇄2NO2(g). If Kp = 0.144 at 950.0 K, calculate Kc. (no sc...
A:
IUPAC name
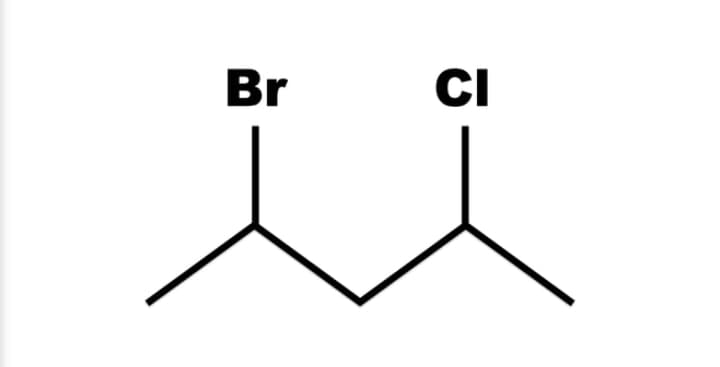

Step by step
Solved in 3 steps with 4 images




