Q: What is the chemical equation involved with the reaction of cyclohexene with cold KMnO4?
A: Alkenes are organic compounds containing carbon and hydrogen atoms only. There is a presence of a…
Q: Write the chemical equations involved for the reaction of cyclohexene with cold KMnO4
A: In the chemical equation, the material or chemical symbol present on the left-hand side of the right…
Q: Draw the product of this reaction. Ignore inorganic byproducts. KMNO4, NaOH cold
A: Oxidation of alkene with cold alkaline KMnO4: KMnO4 is very strong oxidizing agent in alkaline…
Q: (a) Upon the addition of H2SO4 to the reaction, a precipitate is observed. What do you believe the…
A: As per the bartleby expert guidelines, I am allowed to answer first one question at a time. Please…
Q: Write the reaction mechanism between ethane and chlorine in the presence of light. Label each step…
A: To find: the reaction mechanism between ethane and chlorine in the presence of light.
Q: Discuss the role of the following in the liquid-liquid extraction of dyes. A. Vinegar B. Baking…
A: Components are partitioned between an aqueous layer and the organic layer in the liquid-liquid…
Q: In the basic medium, write the compound that gives 1 mol of acetone and 1 mol of acetic acid when…
A: The oxidative cleavage of alkenes is performed in the presence of potassium permanganate. The double…
Q: Propose a methodology for the synthesis in the laboratory of AICI3*6H2O from shot or aluminum foil,…
A: Metals are the substances that have a tendency to lose electrons. Hence, metals have a tendency to…
Q: Account for the relative rates of solvolysis of these compounds in aqueous acetic acid. (CH3),CBr Br…
A: In general, the carbocation formation in bridge heads are not possible because they don’t adopt the…
Q: What is the role of Sodium metal in Lassaigne's extraction?
A: For preparing Lassaigne's extraction, Chemical compound is fused in presence of sodium metal in…
Q: Outline the possible reaction pathways with proper mechanism for the formation of Nylon 6 from…
A: Polymerization: It is a chemical process when a monomer (single unit) combined through various…
Q: Provide the structure of the product and write the mechanism of its formation for the following…
A: Given two reaction are of intermediate chemistry i.e. carbocation formation reaction. Reactive…
Q: OH ОН
A:
Q: Chemical Engineering 7) Propose multistep syntheses for the following molecules. i EtO OEt MeO OMe
A: Note - Since the given question is a multiple question, hence I solved first question according to…
Q: A vessel was shipped to your plant which was only labelled H2S (g). Your colleague named it as…
A:
Q: a) Write down the products that will occur when you withdraw HBr from 2-bromine-3-methyl butane in a…
A:
Q: Which one of the following substances is more likely to dissolve in CC14? O CH3CH2OH O H2O O NH3 O…
A: Given Solvent is CCl4 .
Q: Provide the set of conditions to yield the following molecule. CH3 A. 1. NaNH2, NH, 2. CH,I 3. O, 4.…
A: Alkyne gives a hydration reaction in the presence of sulfuric acid and a mercuric acid catalyst to…
Q: 1. Suggest why it is possible to synthesize many boron-nitride analogs of aromatic carbon compounds,…
A: “Since there are multiple questions and it is not mentioned that which one has to be solved so I am…
Q: Discuss the shape of micelles after c.m.c. with increasing amphiphile concentration.
A: Amphiphiles are synthetic or natural molecules with the ability to self-assemble into a wide variety…
Q: Keeping in mind the leveling effect, can the following species be used as a reactant in ethanamine…
A: For a species to act as a reactant in Ethanamine, it should be a stronger base than ethanamine…
Q: Suggest products for the following reactions with mechanism : 1) Li, NH3(1) (i) →? FOH 2) H3O® 3)…
A:
Q: Mg metal + 6M HCl No reaction Readily dissolved with H2 gases evolved Partially dissolved with the…
A: Magnesium metal reacts with hydrochloric acid to give magnesium chloride and release of hydrogen…
Q: How are we going to know if a chemical equation is to undergo SN1 orSN2 reaction
A:
Q: From the reaction of a hydrocarbon of your choice with hydroxyl radicals, show how NO is oxidised to…
A:
Q: Outline the dissociation reactions for carbonic acid and list the electrical charges of protonated…
A: In this question, we have to write the dissociation reactions for carbonic acid and list the…
Q: 9a. Give a complete reaction scheme for the polymerization of styrene with tert-butylhydroperoxide…
A:
Q: Suggest reagents and reaction conditions for each of the following reactions: (a) (b) (c) (d) O H O…
A: The questions discusses about some of the transformations related to the carbonyl functionality…
Q: You are asked to make a flowchart that describes the process from the completion of the Grignard…
A:
Q: 2. Write suitable detailed mechanisms to account for the formation of the products in the following…
A: We have to give the mechanism for the formation of the product.
Q: Which of the following reaction(s) DO NOT take place (under the given conditions) or/and DO NOT give…
A:
Q: 20. a) Write and name of the mechanism when chloroethane is refluxed with ethanolic potassium…
A:
Q: 2. Write suitable detailed mechanisms to account for the formation of the products in the following…
A:
Q: State TWO (2) organometallic reactions that involve the concept of dn-pn bonding.
A: The above question describes one of the important reaction in organometallics i. e. the reaction…
Q: If you put methylene blue and disperse red 1 mixture into salad dressing, predict what will you…
A:
Q: Provide a short paragraph that relates the most essential elements of theory behind the Wittig…
A: Wittig reaction is used to prepare alkenes form carbonyl compounds specifically aldehydes and…
Q: Sodium borohydride (NABH4) is said to be a chemoselective reducing agent. Which of the following…
A:
Q: 5. () Between compounds I and II, say which one will undergo nitration more readily than the other,…
A: If a group attached to aromatic compound is electron withdrawing than electron density in the ring…
Q: Define the mechanism of acid–catalyzed halogenation ?
A: Halogenation is the chemical reaction which involves the addition of one or more halogen atom to a…
Q: Br PPH3 -A diethyl Ether
A:
Q: 1. What conclusions can be reached given the following information? [Be specific regarding kinetic…
A: The explanation of kinetic and thermodynamic controlled reaction along with ΔG at equilibrium and at…
Q: write the reactions in which one of the following compounds acts on the ammonia solution of silver…
A: Write the reactions in which one of the following compounds acts on the ammonia solution of silver…
Q: Predict the products for the following reactions, indicate major and minor where applicable MgBr…
A: In a given question we have to find out the major and products.
Q: 9ー
A: The given reaction is an example of a pericyclic reaction. In this reaction a cycloaddition reaction…
Q: To ensure that the decolorization of crystal violet reaction takes place within the linear range of…
A: Given: 10 mL of stock solution is diluted twice. First with 10 mL of distilled water and then with…
Q: Briefly describe mechanism involved during formation of precipitate
A:
Q: Predict the major products of the following reactions and give the structures of any intermediates.…
A: There are two types of addition in organic reactions - Markonichoff Addition - In this most…
Q: Why is the reaction mixture extracted with sodium bicarbonate? Give an equation and explain its…
A:
Q: 4. In a hydrolysis reaction, 1-chlorobutane (9 mL) gives 1-butanol (7 mL); what is the per cent…
A: • We know that 22.4 L of volume = 1 moleGiven,1-chlorobutane → 1-butanol. Volume of 1-…
Suggest reagents and reaction conditions for step 1
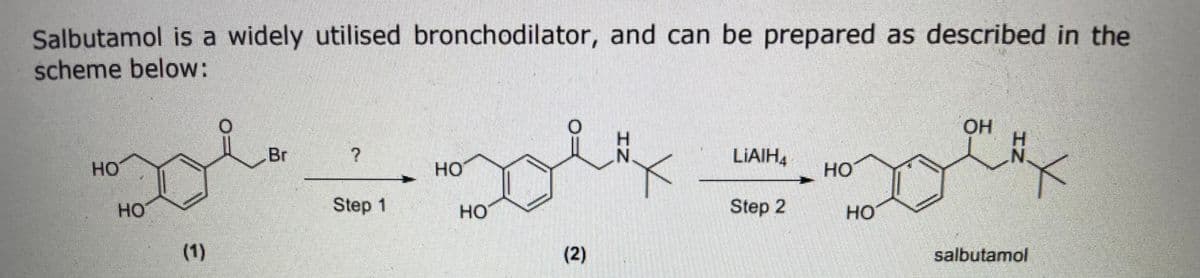
Step by step
Solved in 4 steps with 1 images

- A mixture of A, B, and C was added to a separatory funnel containing CH2Cl2and 10% aqueous NaOH solution. Which compound(s) are present in theaqueous layer, and which compound(s) are present in the organic layer?What are the nonprotein nitrogen components of the blood. Give their chemical structures and relative physiologic concentrations and describe their biosynthesis and excretion. Kindy insert your references. Thank you!Polyunsaturated fatty acids (PUFA's) in oils 0.g. sunflower oil are prone to rancidity because they contain conjugated double bond systems which are easily oxidized in the presence of light or peroxides. BHT Is used as an antioxidant in oils to prevent oxidative rancidity. How do BHT prevent oils from reacting with peroxides so that they do not become rancid? Show the mechanism.
- Repeated laboratory results over the past three weeks suggest that your patient suffers from acidemia. In the most recent visit, blood tests yielded the following results (normal values in parenthesis): pH = 7.34 (7.4) pCO2 = 60 mm Hg (40 mm Hg) [HCO3-] = 31 mM (24 mM) The problem is most likely due to: A) high glucose level in blood B) vomiting and nasogastric suction C) chronic respiratory center depression and central hypoventilation D) diabetic ketoacidosis E) none of the aboveCarbonic anhydrase of erythrocytes (?r 30,00030,000) has one of the highest turnover numbers known. It catalyzes the reversible hydration of CO2.CO2. H2O+CO2↽−−⇀H2CO3H2O+CO2↽−−⇀H2CO3 This is an important process in the transport of CO2CO2 from the tissues to the lungs. If 33 ?g of pure carbonic anhydrase catalyzes the hydration of 0.10 g0.10 g of CO2CO2 in 1 min1 min at 37 °C37 °C at ?max, what is the turnover number (?cat) of carbonic anhydrase? Please provide only typed answer solution no handwritten solution needed allowedCarbonic anhydrase is one of the fastest enzymes known. If 10 µg of this enzyme catalyzes the conversion of 0.30 g of CO2 to bicarbonate in 1 minute at 37˚C. What is the Kcat value for this enzyme in min -1?
- Q5: If the Ksp of Ca3N2 is 6.06 x 10-3, how many moles of calcium nitride (s) could you dissolve in 1.589 L of solvent (assume that calcium nitride (s) dissolves in water to form Ca2+(aq) and N3- (aq))? Enter your answer with at least 3 sig figs. Please provide only typed answer solution no handwritten solution needed allowedIsoleucine, another of the twenty amino acids found in proteins, is metabolized by a pathway that includes the following step. Propose a mechanism.During exercise when the body lacks an adequate supply of oxygen to support energy production, the pyruvate that is produced from the breakdown of glucose is converted into lactate. High lactate levels can lead to acidity in the muscle cells as some of the lactate hydrolyzes to lactic acid. The dissociation of lactic acid to lactate is shown in the reaction. Lactic acid has a p?apKa of 3.86.3.86. A solution containing a mixture of lactic acid and lactate was found to have a pH of 3.24.3.24. Calculate the ratio of the lactate concentration to the lactic acid concentration in this solution.
- The Ksp for AG2SO4 is 1.2x10^-5 and the Ksp for CaSO4 is 4.93x10^-5. How do I find the Ca concentration?When some sugars dissolve in water they spontaneously undergo changes in optical rotation called mutarrotation. The Mutarrotation of D-glucopyranose is catalyzed by acid and bases. 2-Hydroxypyridine is a more effective catalyst than phenol and pyridine for this reaction because: a.Both oxygen and N in 2-hydroxypyridine act as bases increasing the rapid interconversion of sugar b. The OH of 2-hydroxypyridine serves as the base while the current N as the acid. c. 2-hydroxypyridine acts both as a base to remove the proton from the hydroxyl group in the hemiacetal and as an acid to provide a proton to the oxygen in the hemiacetal. d.Phenol and pyridine are very expensive.d-Glucuronic acid is found widely in plants and animals. One of its functions is to detoxify poisonous HO-containing compoundsby reacting with them in the liver to form glucuronides. Glucuronides are water soluble and therefore readily excreted. Afteringestion of a poison such as turpentine or phenol, the glucuronides of these compounds are found in the urine. Draw thestructure of the a- and b-glucuronides formed by the reaction of b-d-glucuronic acid and phenol.



