Q: Will the reaction conditions below favor SN2 or E2 chemistry? (Simply write SN2 or E2 on the right…
A:
Q: Write the reaction mechanism E2 between 2-bromopropane with sodium hydroxide, NaOH.
A: 2-bromopropane is heated with a concentrated NaOH solution in ethanol to produce propene gas as the…
Q: Write the reactions between acetone and phenol (molar ratio 2:1) in acid catalysis.
A: In organic conversions, appropriate reagent converts the reactants into products. Reagents attack…
Q: Upon completing a successful synthesis (shown below) you are tasked with recrystallizing this solid…
A: In this question, we will select the solvent which is suitable for the recrystallization of the…
Q: 2. Review the E2 mechanism, provide step-by-step mechanism for the following reactions: N2OCH3 a.…
A: This reactions are E2 reaction. Here a base takes a proton which is just anti to the leaving group.
Q: Question 5
A: The nucleophilic addition to C=O compounds (carbonyl) form 1,2-addition or 1,4 addition. The…
Q: Which of the statements is most correct regarding the products expected from the halogenation…
A: Solution: We know alkenes can act as nucleophile due to electron reach Pi bond. They shows…
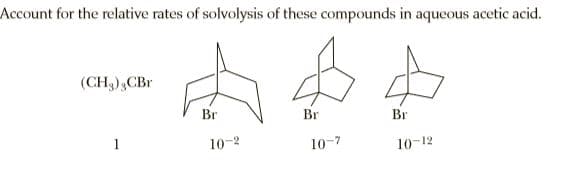
Q: What will most rapidly undergo solvolysis in aqueous methanol? Br Br Br Br
A: An organic reaction in which the leaving group of the reactant molecule is replaced by the other…
Q: 7. In the reactions below predict if the major process is SN1, E2 or SN2 and then write the major…
A:
Q: Predict the structure of the product formed when HCl adds to 1-Bromo cyclohexene. Also explain how…
A: Halogens are electronegative atom , thus it should have act as electron withdrawing group ,in…
Q: What will most rapidly undergo solvolysis in aqueous methanol? Br A. В. Br Br C. D. O B OC OD
A: Solvolysis is a type of Nucleophilic substitution where the nucleophile is a solvent molecule.
Q: Can you list the possible sources of error that can affect the yield of a Grignard reaction followed…
A:
Q: H OSIET3 OSIME3 МеОН H2N + Lewis acid (cat), CH3CN
A:
Q: how how the forward and reverse reaction rates respond to having some HOCl suddenly removed from the…
A: The reaction rate depends on the concentration of the reactant but the reaction rate does not depend…
Q: 3 CH3I NaOH solution NH2 [7] (11) (i) AGOH (replaces I with "OH) (ii) 180 °C [8] (14) (12) (13) 95%…
A:
Q: Discuss SN1 and SN2 reactions , highlighting the key difference between the two reactions.
A: Both SN1 and SN2 are Nucleophilic substitution reactions in which a Nucleophile attacks on the…
Q: State the characteristic of the SN2 and SN1 mechanism towards alkyl halide compounds based on…
A: The substitution reaction of nucleophiles involves the replacement of an electron-rich component…
Q: d. s-OH е. -N `N' TICI4 `N' caffeine
A: The given reactions are acid-base reactions
Q: Does the following reaction take place in a conrotatory or disrotatory manner? Would you carry out…
A: Electrocyclic reactions involves the rotation of bonds in such a manner that the HOMO-LUMO…
Q: (7) Complete the following outlined reactions by providing the major organic product expected in…
A: We have to carry out the given transformation.
Q: Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. For each pair of compounds,…
A: The compounds given to be distinguished are: to be chosen from the options given: I. Halogenation:…
Q: Alkenyl halides such as vinyl bromide, CH2=CHBr, undergo neither SN1 nor SN2 reactions. What factors…
A: SN1 reaction is a nucleophilic substitution reaction. It is a uni-molecular reaction which takes…
Q: A. Compute the amount of reagents which will be used for the preparation of the following reagents.…
A: Hello. Since more than one question has been asked and the first question contains more than three…
Q: What structural and reactivity factors are necessary for E1 reactions to occur?
A:
Q: 7. For each of the following pairs of reactions a. Sketch a comparative Gibbs Free Energy diagram…
A: The Gibbs free energy diagram is obtained by plotting the change in free energy of the reaction…
Q: Write a mechanism for the following reaction. он NANO2 HCI 0°C NH2
A:
Q: Which will react faster in SN2 displacement, 1-bromopentane or 2-bromopentane, and why?
A: The SN2 reaction is a substitution nucleophilic bimolecular reaction. In this reaction, bond…
Q: Predict the product for the following reactions. Below each question, write a sentence explaining…
A:
Q: HO OH OH OH NH3 (aq)
A: We have to give the possible mechanism of the given transformation.
Q: 5. () Between compounds I and II, say which one will undergo nitration more readily than the other,…
A: If a group attached to aromatic compound is electron withdrawing than electron density in the ring…
Q: How does changing the solvent from DMSO to CH3OH affect the rate of an E1 reaction?
A: E1 reaction stands for unimolecular elimination reaction. As the name suggests these reactions…
Q: Dimerization is a side reaction that occurs during the preparation of a Grignard reagent. Propose a…
A: Please find below the mechanism for the above reaction.
Q: Complete the reaction with writing the suitable elimination mechanism онн CH,0 OCH3 нн CH;OH Base H'
A: We have given the organic reaction and we have to find the major product and the mechanism of the…
Q: HO ОН `ОН H₂CRO4 H2O PCC DCM MnO₂ acetone
A: -> H2CrO4/H2O is reagent used for oxidation of every alcohol. -> PCC/DCM is reagent used for…
Q: What structural and reactivity factors are necessary for an E2 reaction to occur?
A: The concept behind this question is how a elemination reaction proceed and all the factors that…
Q: Provide a chemical formula (AmBn) for the product of the reaction below SH [0]
A: -SH functional group represent a thiol which undergo oxidation to form disulfide.
Q: Br CH,OH
A: Given reaction is sn2 substitution reaction. And we get inversion of stereochemistry in product…
Q: In the presence of water, which would be reactive to the electrophilic addition of Br2?
A: The conversion of a double bond present between two carbon atoms in alkene to a C-C bond is called…
Q: Which one of the following solvents is the best choice for SN2 reaction? cyclohexane water hexane O…
A: The solvent which is used in SN2 reaction is given below.
Q: Provide the structure of the major organic product of the reaction below. H3C CH3 NaOCH,CH3 CH;CH,OH…
A: We have given the organic reaction and we have to find the major product of the reaction.
Q: 1) Complete the following reaction and provide a detailed, step-by-step mechanism for the process.…
A: The given problem is based on the chemical properties of alkenes.
Q: What will most rapidly undergo solvolysis in aqueous methanol? Br A. B. Br Br C. Br. D.
A: Solve Solvolysis reaction follows SN2 mechanism.
Q: Grignard Reactions Provide the structure of the major organic product in the reaction shown below.…
A: The Grignard Reaction is the addition of an organomagnesium halide (Grignard reagent) to a ketone or…
Q: 1. LİAIH4 HO. 2. H30 * HO,
A: Given: Reaction of acetic acid with lithium aluminium hydride To find: Mechanism Solution: when…
Q: 6) Provide the structure of the major organic product in the reaction shown below. NaBH4 CH3 HOCH3
A: The structure of the major organic product in the given reaction = ?
Q: 1. A student wants to synthesize compound C from the compounds A and B under alkaline (basic)…
A: The base is a substance that can furnish its unshared electron pair to other substances. For…
Q: 1. Complete any three of the following reactions and write down their stepwise reaction mechanisms.
A:
Q: HO, H,SO, (cat.) H. HO OH
A: Acetal is a functional group that has two ether bonds connecting the same carbon atom. When…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Question: How do quantum mechanical effects influence the stability and reactivity of molecules with non-classical carbocations, such as the 2-norbornyl cation, and how does this impact the reaction mechanisms and outcomes?For the base-catalysed hydrolysis of 3-bromo-3-methylhexane (i.e. reaction with the nucleophile OH-): State whether the reaction is likely to proceed by an SN1 or SN2 mechanism, and explain why give the likely rate law and explain whyExplain the transition state And effect of non polar solvents of sn2 reaction
- Suggest an explanation for the fact that the order of reactivity of the halides toward n-butylbrosylate in acetone is Cl” > Br” > I~ when (C4H9)4N* is the cation of the halide salt butI” > Br” > Cl” when Li’ is the cationAcid Halide Preparation reaction mechanism: Please use HCl and SOCl2 as reagentsState the characteristic of the SN2 and SN1 mechanism towards alkyl halide compounds based onmolecularity, reaction rate constant, formation of product(s), reactivity of alkyl halides and thestrength of nucleophile.
- Outline the mechanism showing both products formed for the reaction of ammonia (2 mole equivalent) with 1-bromobutane (1 mole equivalent).write the reaction of the action of methyl-2, bromo-2-propane with hot KOH solution. a) Name the formed product b) Explain why the reaction takes place according to the SNI mechanismThe E1 mechanism (unimolecular elimination) of Elimination ?

