Q: A diprotic weak base (B) has pK, values of 4.058 (pK) and 7.102 (pKa). Calculate the fraction of the…
A:
Q: 6. For these reactants: (a) Identify the E+, LG, Nu:. (b) Predict the product(s). (c) Is the…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: about heat conduction Task 1: . What happens to the temperature of a well-insulated room, if a…
A:
Q: A classmate asked you to look at a direct synthesis of curcumin using vanillin and 2,4-pentanedione.…
A: Given that, the synthesis of Curcumin from vanillin and 2,4-pentandione. We have to tell whether the…
Q: 7) Label the species being reduced and the reduc'ng agent in the following redox reaction? Zn2+(aq)…
A: Redox reaction: Redox reactions include reactions that involve the loss or gain of electrons.…
Q: if you want to make 1000.0 mL of a 0.150 M solution starting with a standard solution of 0.550 M,…
A:
Q: hat will be the major product for the following transformation? reactions done on benzene ring and…
A:
Q: A reaction has a positive entropy change. What would the sign of AHrxn need to be to make the…
A: Let us consider the equation for gibbs free energy ΔG=ΔH−TΔS Where,δH is enthalpy change (KJmol −1)…
Q: A transition metal chloride (MCI;) combines with pyridine and DMSO to give a metal complex with a…
A: To which atom does the DMSO ligand bond to central M2+ ion.
Q: 3) Balance the following redox reaction if it occurs in acidic solution. MnO4 (aq) + Fe(s) Mn2+(aq)…
A: You follow a series of steps in order: 1. Identify the oxidation number of every atom. 2. Determine…
Q: Draw the structural formula for 2,5,6-trimethylnonane
A: Given-> 2,5,6-trimethylnonane
Q: Use the following atomic weights and quantities to calculate the overall % yield of…
A:
Q: If a copper metal is immersed in an aqueous solution containing Ag+ ions, what would you expect to…
A:
Q: Consider the half-reaction for the reduction of Cd2+ to Cd(s). Cd² + (aq) + 2 e¯ – Cd(s) = -0.403 V…
A: Since you have asked multiple questions, we will solve first one for you. For remaining questions,…
Q: When 1.06 g of a certain molecular compound X are dissolved in 85.0 g of dibenzyl ether…
A: we need to calculate molar mass of the compound X
Q: What are the main components of an electrochemical cell?
A: An electrochemical cell is an apparatus or tool that produces electric current from chemical change…
Q: Give the name for the structure in the image shown:
A:
Q: Q5.Interpret the following pH values of solutions by converting them to concentration of hydrogen…
A:
Q: 1.) Using the axes provided below, draw a plot of free energy, G, for the generic reaction: A(g)B(g)…
A: It is given that for the reaction, K>1, thus forward reaction rate is more than the reverse…
Q: ow many equivalent orbitals are involved in each of the following sets of hybrid orbitals? a. sp…
A:
Q: A laboratory has 12.69 mg Cu2+ available for investigation. If the molar solubility of Cu(OH)2 is…
A: Given: Mass of Cu2+= 12.69 mg = 0.01269 g (Since 1 g = 1000…
Q: 10) If 100 grams of water saturated with potassium chloride, KCI at 50°d were cooled to 20°C, how…
A: The solubility of a substance can be explained as the total amount of the substance that can be…
Q: Write the balanced COMPLETE ionic equation for the reaction when NaC₂H₃O₂ and LiCl are mixed in…
A: Given statement is : Write the balanced COMPLETE ionic equation for the reaction when NaC₂H₃O₂ and…
Q: -1 A certain liquid X has a normal freezing point of 5.40 °C and a freezing point depression…
A: Formula used :- ∆Tf = Kf m Where ∆Tf = change in freezing point = T0 f - Tf Where T0 f =…
Q: H2S has a solubility in water of 4 g.dm–3. Calculate the molarity of a saturated solution.
A: Given-> Solubility = 4 g.dm-3 =4 g.L-1 (1 dm3 = 1 L)
Q: By how many kilojoules per mole is the energy of O2 increased when it absorbs UV radiation with a…
A:
Q: Which of the following statements is/are true for all voltaic/galvanic cells? I. Reduction occurs at…
A:
Q: What mass of NO2 would have the same number of moles of N with 0.402 g of porphyrin, C20H14N4?…
A:
Q: Create an infographic regarding the Imperfections of solids and Diffusion in Solids.
A: Imperfections in solids are the irregularities in the arrangement of atoms around a point in a…
Q: Draw the following functional groups. 4-ethyl-5-isopropyl-2,3-dimethyl-2,4-nonadien-6-ynedial…
A: Since we know that alkene has more priority than alkyne in iupac naming...
Q: Select Draw Rings More Erase Q ame the isomer with 3 carbons in the main chain.
A: Organic chemistry is branch of chemistry in which we deal with carbon-hydrogen bond.
Q: What is the potential after the addition of 50.00 mL of 0.02000 M Mno, to 50.00 mL of 0.05000 M U".…
A: Solutions- Given- conc. of Mn+2 = 0.01 M, MnSO4 = 0.02 M
Q: An AX ceramic compound has an HCP crystal structure. If the radii of the A and X ions are 0.12 and…
A:
Q: Determine the mole fraction of NaF in a 11.7343 wt % aqueous solution of NaF.
A: Weight percent or mass percent :- The mass of solute in grams present in 100 grams of solution is…
Q: A 1,800 ml solution contains 9 moles of solute. What is the molarity of the solution?
A:
Q: Consider the following reactions. 2+ Mg (aq) + 2e 4 Mg(s) E° = -2.36 V 2+ Zn (aq) + 2e s Zn(s) E° =…
A: Standard reduction potential values of metal decreasing order is Cu > Zn > Mg…
Q: Solve the following problems. Show your complete solutions. 1. A cylinder contains 4.18 L of…
A:
Q: [so;] = 3
A:
Q: The bisulfide anion (HS') has a pk, of 19.0. This means that a 0.10 M aqueous solution of HS is…
A:
Q: Please write clearly and go step by step. Please include the correct stereochemistry for each…
A: Here we have to determine the major product formed in the following elimination reaction.
Q: What is the electron-domain geometry for an ABn molecule in which the central atom A has the…
A: Electron Geometry is determined by Hybridization. Hybridization is the overlapping of atomic…
Q: 3+ (aq) + 3 e¯ → Fe(s) E° Fe(s) | Mg²+ (aq) || Fe³+ (ag) | Mg(s) O Mg(s) | Mg²+ (ag) || Fe³+ (aq) |…
A: In galvanic cell, Oxidation takes place at anode and reduction takes place at cathode. Element…
Q: Propanoic acid that, along with its sodium salt, can be used to make a buffer that has a pH of 5.25.…
A:
Q: How can I get the concentration of Acetic Acid from volume?
A: Going back to moles, you can calculate the mass of acetic acid in the solution by multiplying the…
Q: at pressure (unit atm) will
A:
Q: Find the activity coefficients for both ions of AgCN in a 500 mL solution saturated with AgCN(s)…
A: Molar concentration of KNO3 in the solution: =Mass of KNO3FMKNO3×Volume of solution (in L)=5.055…
Q: For a particular redox reaction, BrO is oxidized to Bro, and Fe3+ is reduced to Fe2+. Complete and…
A: In this question we have to balance the reaction in the basic medium.
Q: Properties of Nuclear Radiation Science 10 Property Alpha Decay Beta Decay Gamma Decay Particle…
A: There are mainly following type of Nuclear decays - (1) Alpha decay - In this type of decay ,…
Q: A 0.1824-g sample that contains both NaCI (MW= 58.442) and NaBr (MW= 102.89) is titrated with 0.1052…
A: Given data,Mass of sample=0.1824 gMolecular weight of NaCl=58.442 g/molMolecular weight of…
Q: p-toluic acid is prepared from 3.42 grams of p-Bromotoluene. When p-Bromotoluene was mixed with dry…
A:
Provide the appropriate reagents for the example.
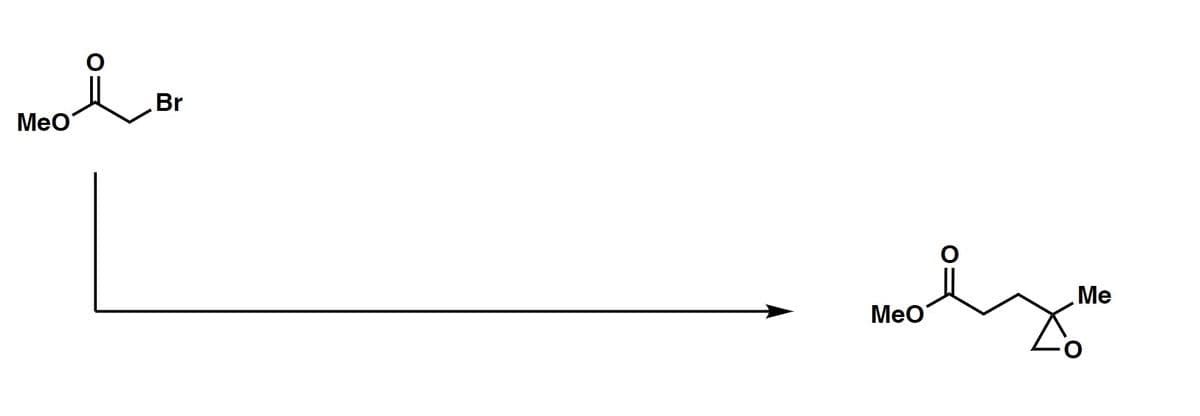
Step by step
Solved in 2 steps with 2 images


