A transition metal chloride (MCI;) combines with pyridine and DMSO to give a metal complex with a formula MCI2(DMSO)»(py). The FT-IR spectrum (in the range from 1425 to 300 cm) of the complex is shown below. L= DMSO MCI, + py + 3L py=N
A transition metal chloride (MCI;) combines with pyridine and DMSO to give a metal complex with a formula MCI2(DMSO)»(py). The FT-IR spectrum (in the range from 1425 to 300 cm) of the complex is shown below. L= DMSO MCI, + py + 3L py=N
Chapter26: Molecular Absorption Spectrometry
Section: Chapter Questions
Problem 26.30QAP
Related questions
Question
4
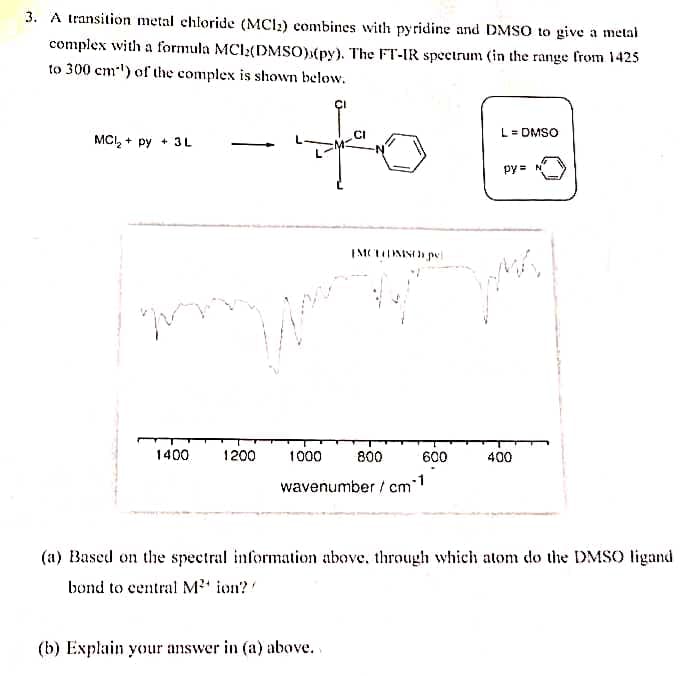
Transcribed Image Text:3. A transition metal chloride (MCI2) combines with pyridine and DMSO to give a metal
complex with a formula MCI2(DMSO)(py). The FT-IR spectrum (in the range from 1425
to 300 cm-') of the complex is shown below.
to
CI
L = DMSO
MCI, + py + 3L
py = N
1400
1200
1000
800
600
400
wavenumber / cm
(a) Based on the spectral information above, through which atom do the DMSO ligand
bond to central M' ion?
(b) Explain your answer in (a) above..
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



