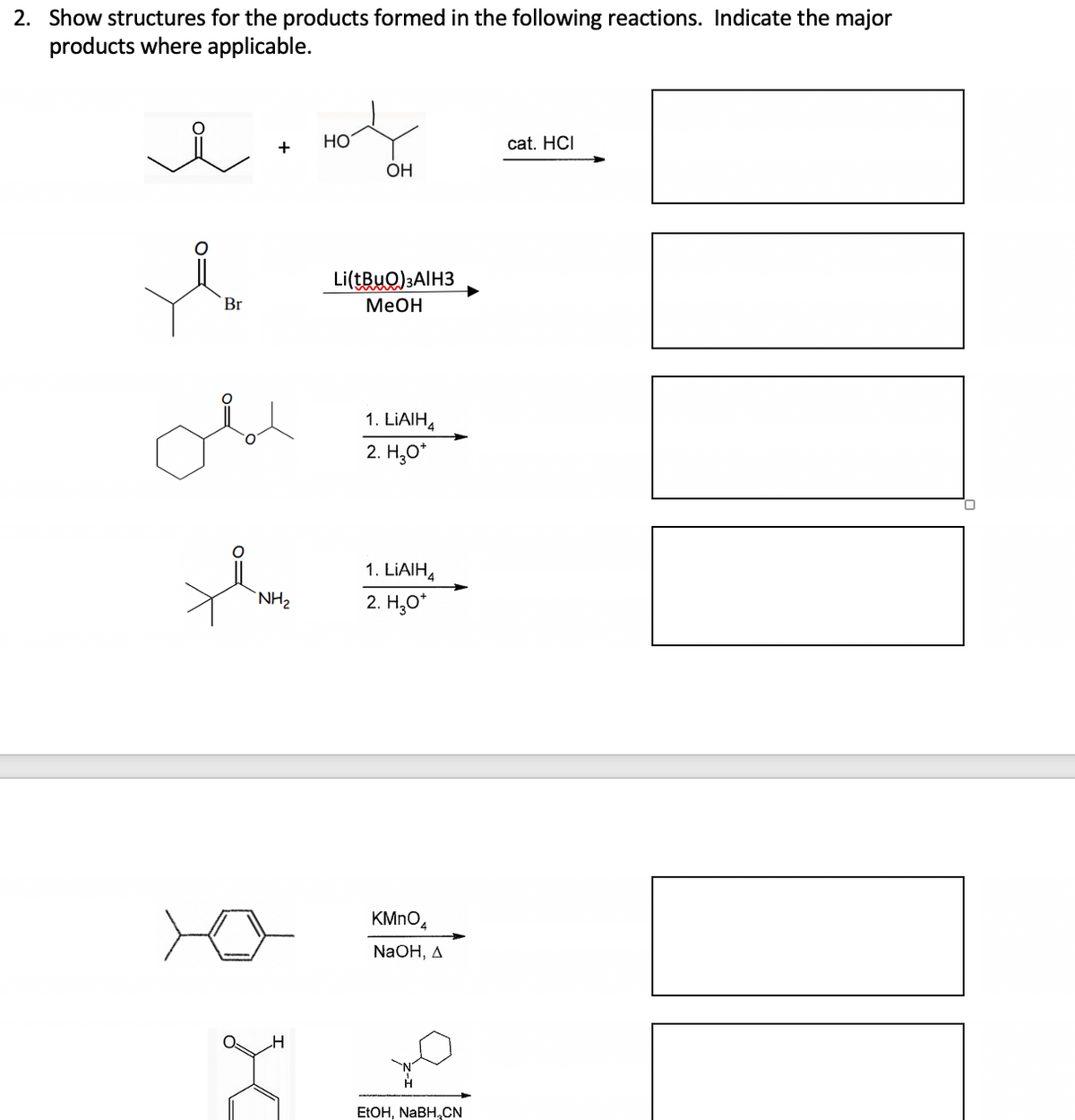
Q: Predict the products for the following single and double displacement reactions, and write balanced…
A: Given incomplete reactions are : a. Aluminum metal + aqueous silver acetate b. Aqueous zinc nitrate…
Q: 2. One of the common methods used to determine phosphorus in urine isto treat urine with…
A: A question based on absorption that is to be accomplished.
Q: 3. Show the synthetic steps needed to make cis-1-ethyl-2-methylcyclopropane from acetylene. Show…
A:
Q: Provide the correct three-letter abbreviation for the following amino acid: O || H₂N-CH-C-OH I CH₂…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: What is the results of mixing calcium nitrate with sodium carbonate? Group of answer choices A…
A:
Q: A synthesis reaction produces ammonia: 3H2 (g) + N2(g) -> 2NH3 (g). What is the maximum amount of…
A:
Q: Galaulate the PH and the Pott FOR 9.6M solution OF MH₂ инз K. FOR NH3=1.97105
A: Here we are required to find the pOH and pH of 0.6M of ammonia solution
Q: 7. What volume in Litres does 85.4 g of nitrogen gas occupy at SATP?
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: Which conformation is most unstable?
A:
Q: Pb(NO3)2(aq) + KI (aq) Write the complete molecular equation Write the complete ionic equation Write…
A:
Q: Question 2 I, we looked at the structure of Nirmatrelvir, half of the Pfizer COVID antiviral pill…
A:
Q: Which of the following must be true for an optically active compound?
A:
Q: 21. What volume of stock solution is needed to prepare 100.00 mL of 0.125 M solution from 2.50 M…
A:
Q: ✓ The molecular formula of this molecule is NH
A: chemical formula gives the information about the total number of atoms of each element in the…
Q: Please do ring flip of this and determnine both if trans or cis. Thanks
A: Ring flipping in cyclohaxane is the conversion of one chair conformation into another chair…
Q: How many orbitals are there with n = 3 in an atom?
A: Since you asked multiple image questions , so as per Q&A guidelines of portal I solved first…
Q: 1. Provide the correct IUPAC name for the following amines. CH3 H3C. H3C. NH H3C CH3 H. CH3 CI N H…
A: Given compounds are : Provide the correct IUPAC names of the following amines The general…
Q: Quvon (aq) 2 Fecs) + 3 F₂ cg) → GF - (99) + 2 Fe ² + in which reaction, which is oxidising the agent…
A: Oxidation reduction reaction (Redox reaction) It can be defined as the chemical reaction in which…
Q: 15. A 25.0 mL solution of 0.250 mol/L methanoic acid, HCOOH(aq), is titrated with a 0.100 mol/L…
A:
Q: Which ions are spectator ions in the following chemical equation? NaCl + NH4NO3 --> NaNO3 + NH4CI 1)…
A: Spectator ions in a chemical equation
Q: 21.80 Prednisolone is the synthetic glucocorticoid medicine most frequently prescribed to combat…
A: this given structure is structure of glucocorticoids
Q: Calculate the tare weight for an object placed in a 10.9114 g container with a total mass of 16.4545…
A: Container that when subtracted from gross weight yields the net weight is regarded as tare weight.…
Q: ΔHsolution for most salts is positive. Why then do most salts dissolve in water anyway?
A: DELTA H VALUE FOR SOLUTION is positive because when we add salt in water it require energy( lattice…
Q: xn A Part A: Balanced Chemical Equations (must include physical states) Balanced Total Equation…
A:
Q: Fill in the orbital energy diagram for phosphorus. 3s 2s 1s 3p- 2p
A:
Q: 3. Determine mass percent of nitrogen and iron in Fe(NO3)3.
A:
Q: Determine the Miler indices for the direction represented by an arrow and the planes as shown in…
A:
Q: Draw a structural formula for the major organic product of the reaction shown. CH3CH₂ CH₂CH₂CH3 ● H…
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide.
Q: Please assist. I have been stuck on this lab data for some time now. Concentration Absorbance 15.20…
A: A question based on absorption that is to be accomplished.
Q: The members of which pair of compounds are NOT related as isomers? A. H₂C-CE -CH,`NH-CH3 and…
A: The two or more molecules which have same number of atoms and same molecular formula but different…
Q: 4. The ease of in a carboxyl group makes it an acid, due to A. Gain of an electron, steric effect B.…
A: An functional group will be acid when it's conjuated base will be stable.
Q: What is the relationship between the following compounds? O Different compounds O Identical…
A:
Q: Discuss the mechanistic approaches to the degradation of polychlorinated compounds.
A:
Q: g. At T = 800 K, what would the value of AG be if the P of CO₂ = 0.1 atm? If the P of CO₂ = 10 atm?…
A:
Q: Given the galvanic cell: SCE || Ag*(aq)|Ag, at 25 °C, and the half-reactions. Agt + e → Ag(s), Eº=…
A:
Q: techniques, in electron paramagnetic resonance, the radiation frequency remains constant while the…
A:
Q: 5. The ease of in an amino group makes is a base due to the formation of A. Gain of a proton;…
A:
Q: Compound A 1. 3100-3080 (medium, sharp) cm-1 2. 3000-2850 (strong, intense) cm-1 3. 1610 (medium,…
A: Let us discuss the peaks to find out the structure.
Q: A solution containing unknown cations is added to three test tubes. The following anions were added…
A:
Q: (c) Calculate the n-electron energy of the pi electrons a pentadiene radical
A:
Q: 10. Assuming that the dissociation products are of equal concentration, classify the pH of the…
A: pH : it can define have the negative logarithm of H+ ion concentration. When pH > 7 solution is…
Q: Chemical Equilibrium: Le Chatelier's Principle Practice Use what you know about Le Chatelier's…
A: The Le chatelier's principle states that , " When any chemical reaction is in equilibrium and a…
Q: -3 The osmotic pressure of 8.46 x 10 M solutions of CaCl₂ and urea at 25 °C are 0.511 atm and 0.207…
A:
Q: The pi bond of an alkyne is O shorter, stronger O shorter, weaker Olonger, weaker longer; equal…
A: Organic compounds are the compounds that contain carbon and hydrogen atoms.
Q: For the equilibrium: A ↔ B, if [A]o = 0.162, [B]o = 0.569, and Keq = 47.8, what are [A]eq and…
A: Recall the given equilibrium, A ↔ B…
Q: Depict the electron configuration for each of the following atoms using noble gas notation:
A:
Q: Calculate the molality of a 10.0% (by mass) aqueous solution of sulfuric acid. Your Answer:
A: * Percent by mass solution = Mass of solute/ total mass of solution * molality = Moles of…
Q: Write the reaction rate expressions for the reaction below in terms of the disappearance of the…
A:
Q: Reactions 1, 2, and 3 are: Reaction 1 Reaction 2 Reaction 3 2N2(g) + O₂(g) = 2N₂O(g) N₂O4(g) N₂(g) +…
A:
Q: What is the result of mixing hydrochloric acid with sodium hydroxide? Group of answer choices This…
A: The reaction of hydrochloric acid with sodium hydroxide is an example of acid base reaction. Where,…
Step by step
Solved in 2 steps with 2 images

- We react 2-propanone with 1º NaCN, H+, 2º LiAlH4 and 3º H3O+. Indicate the final product. Comment reactions.Write the type of reaction that is occurring on the line provided then complete the reaction.Only include the major products and any byproducts (e.g. H2O) but no minor products.In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:
- complete this reaction Fe³= ........agent (from-to) S2O3= .........agrnt (from-to).Give the products of the reactions below and label the major projects.I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?
- For the reaction shown below, 2.00 mL of a 2.00 M solution of bromine, 562 mg of alkene reactant, and excess acetic acid are used to obtain 322 mg of brominated product. What is the percent yield of brominated product? Bromine MW = 159.81 g/mol; Alkene reactant MW = 134.22 g/mol;Acetic acid MW = 60.05 g/mol; Brominated product MW = 328.04 g/molWe have a hydrocarbon, C16H26. The compound has two triple bonds and the acids produced by ozonolysis are CH3(CH2)4CO2H and H2OCCH2CO2H. What is a likely structure for this hydrocarbon?On the axes below, sketch the graphs which show the result of adding: 0.1M NaOH to 25cm3 of 0.1M ethanoic acid. 0.1M NaOH to 25cm3 of 0.1M H2SO4
- Kindly identify the product. Show its stable structure. ty this is rush and the topic is alkenes, alkynes and benzenesFill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.Describe the preparation of 2L of a 0.4 M HCl starting with a concentrated HCl solution (28% w/w, specific gravity = 1.15) -36.5 gm/mol

