Chapter3: Mechanisms
Section: Chapter Questions
Problem 96EQ
Related questions
Question
For eaach image, predict the MAJOR product(s). Show stereochemistry where applicable and draw out ALL stereoisomers formed (as major products) in each reaction. State the mechanism(s) by which the major product(s) are formed (SN1, SN2, E1, and/or E2). Reagents are NOT present in excess.
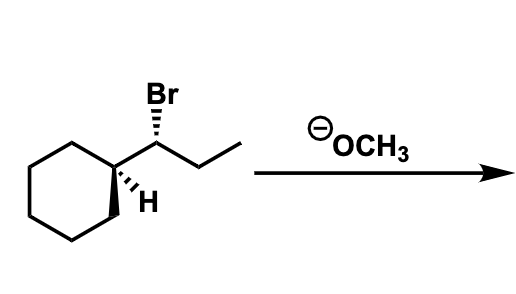
Transcribed Image Text:Br
OOCH3
.א1וו0
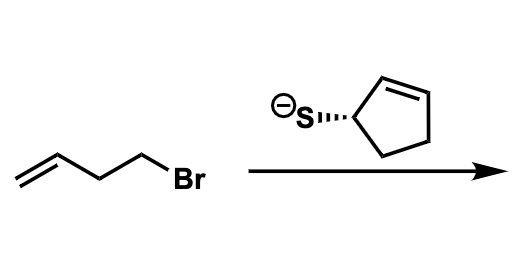
Transcribed Image Text:Br
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

