Q: If a gas occupies 2.3 lites at a pressure of 1.9 atm, what will be its volume at a pressure of 3.8 a...
A: Solution: From the Boyle's law we have P1V1 = P2V2.
Q: A student reacts 13 moles of iron with 21 moles of oxygen according to the following equation: 4Fe(s...
A: Consider the given balanced reaction is as follows; 4 Fe s + 3 O2 g → 2 Fe2O3 s Consider the given ...
Q: A solution has [H+] = 4.0 x 10-3 M. The [OH"] in this solution is %3D 4.0 x 1011 1.0 x 10 14M 4.0 x ...
A: Given data,[H+]=4.0×10-3M
Q: Name of this compound
A:
Q: How much energy is required to convert 42.0g of steam at 138°C to water at 100°C? O A. 102,000J O B....
A: The amount of heat needed for steam to convert from one temerature to other temerature is given by, ...
Q: A sample of impure benzene has a freezing point of 4.2 C. If the fusion point of pure benzene is 5.5...
A: Colligative properties are the properties which depend on the number of atoms or molecules or ions ...
Q: Treating PGC2 with a strong base such as sodium tert-butoxide followed by addition of acid converts ...
A: Treating PGC2 with a strong base such as sodium tert-butoxide followed by addition of acid converts ...
Q: Youhave 10ml of an 8% aqueous solution of H2Cl6Pt. Calculate the platinum in the solution
A: Given data,Volume of solution=10mL%w/v of solution=8%
Q: If the [OH--] of a solution is greater than [H+], the solution is acidic basic neutral none of the a...
A: Neutral Solution: The condition where [H+] = [OH-] is known as neutrality (or the activities are equ...
Q: Q27
A: To determine the electron geometry and molecular geometry of H2S
Q: Which of the following forms of electromagnetic radiation have the highest frequency? a.Radio w...
A:
Q: What is the name of the compound that forms when Cu+ combines with SO32- ? You can use either the ac...
A: Solution- When copper combines with sulfur-containing ions, the redox reaction occurs. It involves t...
Q: CH3 CH(CH32 H3C, H H
A: The structure of the above given compound can be drawn from the Newman projection given above,
Q: In which compound will ammonium phosphate dissolve? Select one: a. carbon tetrachloride b. oil c....
A: Ammonium phosphate formula is (NH4)3PO4 Ammonium phosphate is a salt form. Ammonium and phosphoric...
Q: Do theatoms in one molecule change places with atoms in another?
A: Yes, when molecules reacts together with one another then the atoms in one molecule changes places w...
Q: The radical step with least bond dissociation energy will be:
A: Bond dissociation energy is amount of energy needed to break a chemical bond formed between 2 atoms....
Q: 1400 Mo [D] H. K [C] Pressure (in mm Hg) 1000 [A] 800 G 600 400 [B] 200 20 60 100 140 180 260 Temper...
A:
Q: A concentration cell is set up using two zinc wires and two solution, one containing 0.250 M ZnCl2 s...
A: Given data,Concentration of ZnCl2=0.250MConcentration of Zn(NO3)2=1.65M
Q: How many grams of oxygen are there in 58.0 grams of HClO2?
A: 1 molecule HClO2 has 2 atoms of oxygen. The molar mass of HClO2 is 68.46 g/mol. The molar mass of ox...
Q: Which property increases as you move from left to right across a row in theperiodic table?(a) atomic...
A: The properties of an element are a periodic property of their atomic number, according to the curren...
Q: 41) In titration of weak acid vs. a strong Base; The Ka value of the weak acid can be determined fro...
A: Henderson-Hasselbach equation shows the relationship between pH of a solution, the acid dissociation...
Q: anomeric carbon.
A: Tetrasaccharide is given
Q: 4. Acetate can be shown as (C2H3O2 ^-) or (CH3COO ^-). * True False
A: Acetate is formed by acetoc acid. When acetic acid loses one H atom, acetate is formed.
Q: A patient is prescribed Tigan capsules for vomiting. The drug is available in the form of 3.0 x 10 m...
A: The patient recieve 1.2 ×2 = 2.4 g Tiagn in 2 days.
Q: A 3H nucleus decays with an energy of 0.01861 MeV. Convert this energy into (a) electron volts; (b) ...
A: Conversion is done to change the given unit to the desired unit by multiplying or dividing with the ...
Q: A solution with a pH of 2.17 is
A: Given :- pH of solution = 2.17 To identify :- Nature of solution i.e. acidic, basic or neutral
Q: Answer questions 3 and 4 please. Subject ia Analytical Chemistry
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want...
Q: A chemist titrates 20.00 mL of 0.2000 M HBrO (Ka = 2.3x10-9) with 0.1000 M NaOH. What is the pH: (a)...
A: Note - Since you have asked a question with multiple sub-parts, we will solve the first three sub-p...
Q: A strong acid is any acid with a pH less than 4. True or false
A: On the pH scale the compounds that have a pH vale of 7 are considered neutral, the ones with pH valu...
Q: What would be the name of the compound S2N6?
A:
Q: Based on the band of stability, what is the approximate neutron to proton ratio for vanadium (atomic...
A: The stability of an atom is determined by the ratio of the number of neutrons to the number of proto...
Q: A 314.0-gram piece of this metal at 80.00°C is added to 120.0g of water at 20.00°C. The final temper...
A: Heat capacity is defined as the quantity of heat absorbed by a substance to raise the temperature of...
Q: I only need 8.93 thanks.
A: In HNC the nitrogen acts as the central atom, the Lewis diagram of HNC and formaL charge on each ato...
Q: Radium has a half-life of 1600 years. How many years does it take for 90% of a given amount of radiu...
A:
Q: FTIR Common Name: Phenylmethanal IUPAC Name: Benzaldehyde BP: 179 MP: -26 MICRONS 2,2 23 24 2526 272...
A: In infrared (IR) spectrum % transmittance vs wavenumber is plotted. A particular wavenumber in the I...
Q: A salt crystal has a mass of 0.12 mg. How many NaCl formulaunits does it contain?
A: Given that : Mass of salt = 0.12 mg = 0.12×10-3 g NaCl units = ?
Q: What is the change in energy (in kJ) when 24.0 g of water at 85.0°C is converted to ice at 0°C? ОА. ...
A: The amount of heat released when the water freezes is also known as the latent heat of fusion and is...
Q: Aluminium has a density of 2.70 g/cm. How many moles of aluminum are in a 13.2 cm block of the metal...
A: Solution- Density :− Density of a substance is its amountof mass per unit volume . So , density is t...
Q: Which of the following statements regarding the Rutherford Gold Foil experiment is false? a. The...
A:
Q: Whatis the molarity of a vinegar (acetic acid) solution if 50.00 mL were titrated to a phenolphthale...
A: In chemistry, there are many ways to check the deviation of experimental data from the accurate valu...
Q: Glycogen... O is a polymer of nucleotides O is a polymer of sugar monomers is a peptide polymer is a...
A: Glycogen is a complex bio-macromolecule in the form of which the reserved food material is stored in...
Q: Which of the following should most greatly increase the rate of dissolution of an ionic salt in a po...
A: Which of the following should most greatly increase the rate of dissolution of an ionic salt in a po...
Q: Question 3
A: When two or more atoms combine with each other, they will form a molecule and those molecules have a...
Q: A mixture of barium chloride and sulfuric acid yield the precipitate barium sulfate and hydrochloric...
A: Interpretation - In the above given question we have to determine the mass of H2SO4 needed in gram ...
Q: 38) for a reaction 2A + B >C, AG° =250. kJ/mol. What is the value for AG for this reaction at 25°C I...
A: Introduction The formula of change in Gibbs free energy is as follows: ∆G=∆G∘+RT ln[C][A]2[B]
Q: ignore the lock 3
A: Given that : Mass (m) = 67 g Change in temperature ∆T = 55°C - 0°C = 55°C Specific heat of copper (C...
Q: Show your work please, thank you!
A: The pH of the solution is determined as- pH = -log [H+] And the relationship between [H+] and [OH-] ...
Q: What is the pOH of a solution with a pH of 8? 2 6. 12 14
A: PH is defined as the negative logarithm of the H+ ion concentration. It is measured in PH scale, it...
Q: Q44
A: Redox reaction is the reaction in which reduction and oxidation takes place simultaneously.
Q: Solid CO2 (dry ice) sublimes at 195 K and its heat of sublimation ΔHsubl = 25.2 kJ/mol. A block of d...
A: In order to calculate ΔS for any particular compound, apply the values of heat of sublimation and te...
Show synthetic strategy to convert 1-bromo-1-methycyclohexane into 1-(2-methylpropyl)cyclohexene. Neither mechanism nor stereochemistry are required for this question.
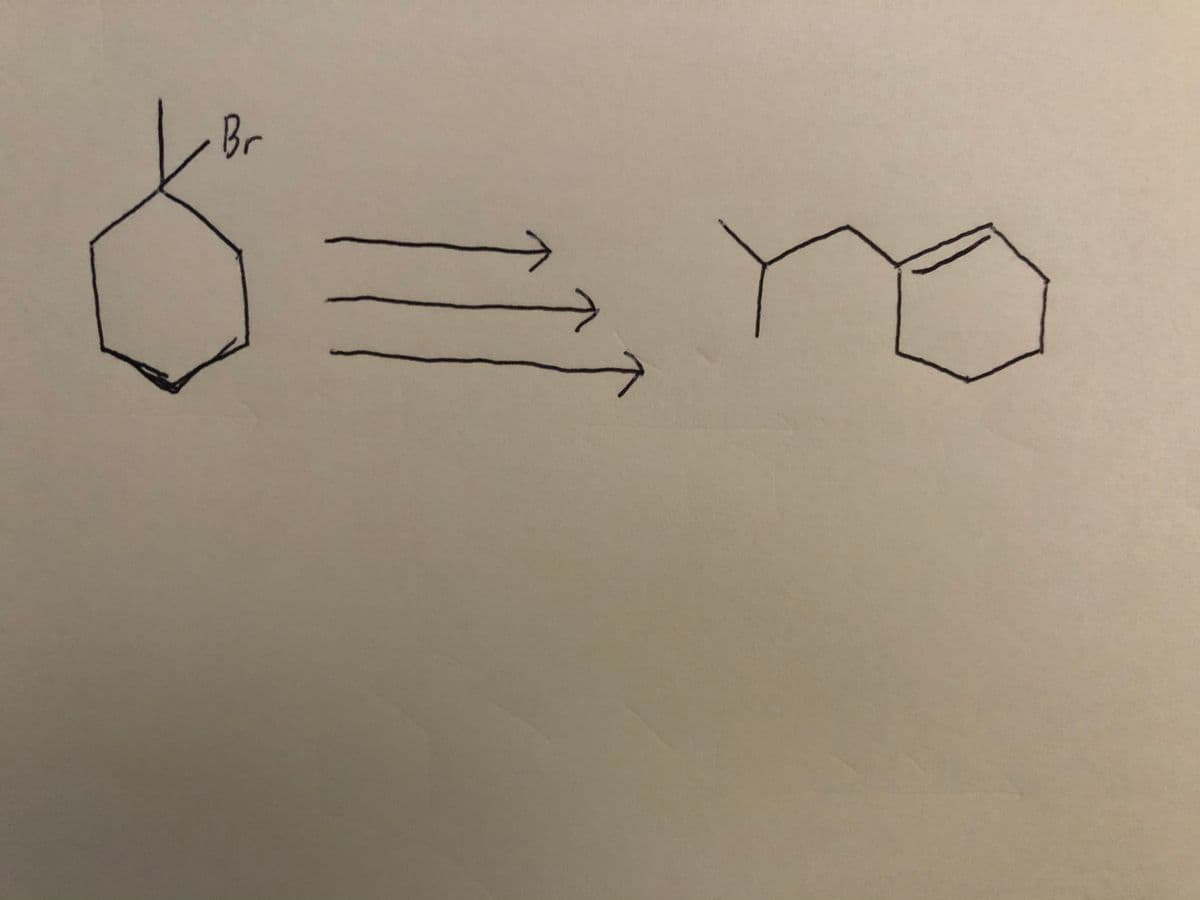
Step by step
Solved in 2 steps with 2 images


