Q: Calculate AH and AStot when two copper blocks, each of mass 10.0 kg, one at 100°C and the other at 0...
A: Solution Given that Mass of each block= (M1) = (M2) = 10 kg Initial tempe...
Q: In order to keep as much HCHO dissolved in the water in the embalming machine reservoir as possible,...
A: For gases At low temperature solubility is more. And HCHO is a gas so to have a great solubility ,...
Q: Chemistry Calculate the dissociation factor (i-value) of NaH2PO4 * 1OH20 (m.w. 300) if it undergoes ...
A: Degree of Dissociation = 65% Molecule is :- NaH2PO4.10H2O
Q: View Policies Current Attempt in Progress The standard enthalpy of combustion, A H¿ ,of graphite is ...
A:
Q: 400g of NaF are disolved in 2 kilograms of water. The solutions density in this condition is 1.2g/ml...
A:
Q: Hydrofluoric acid, HF, has a K, of 6.8 x 10. What are [H,O* ]. [F], and [OH]in 0.960 M HF? [H,O*]-0....
A:
Q: The molar heat of combustion, A H, of sucrose is -5643.4 kJ mol . If 0.250 moles of sucrose is combu...
A:
Q: q = energy transfer into system by heat flow -w = work done by system may be applied to the actual C...
A:
Q: 1. Complete the table below, giving the compound's formula and name. F S2. N3. NaF Na+ sodium fluori...
A: Solution Given the compound Find formula and name = ?
Q: COMBINED GAS LAW A gas has the initial conditions of: 21.2 Liters 279 K 1.03 atmospheres If its temp...
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: (f) ZNCO; (g) NaHSO4 (h) Ba(C10,)2 (i) MgO, G) CrBr3
A: The given inorganic molecules have cation and anion. When we write name of compound, balanced charge...
Q: The equilibrium constant for the reaction Naz(g) - 2Nalg) is 2.47 at 1000. K. Calculate the value of...
A:
Q: Which of these correctly describes an anion ? the number of protons is greater than the number...
A: Which of these correctly describes an anion ?
Q: in the experimental of determination liquid diffusion coefficient Dab where A (liquid solution of ca...
A:
Q: 1. A 10 L balloon contains helium gas at a pressure of 655 torr. What is the new pressure when the v...
A: 1. Given that, a 10 L balloon contains helium gas at a pressure of 655 torrs. We have to calculate t...
Q: b For the reaction CaBr2 (aq) + K2SO4(aq) write the complete ionic equation. (Be sure to specify sta...
A: Given, CaBr2(aq) + K2SO4(aq) -------->
Q: Ammonium perchlorate (NH,CIO,) Is the solid rocket fuel used by the U.S. Space Shuttle. It reacts wi...
A: Ammonium perchlorate decomposes into nitrogen, chlorine, oxygen, and water. The equation for the bal...
Q: Consider the reaction N2(g) + O2(g) = 2NO(g) If the equilibrium partial pressures of N2, O2, and NO ...
A:
Q: Steam reforming of methane ( CH, ) produces "synthesis gas," a mixture of carbon monoxide gas and hy...
A: Given, Initial pressure of CH4 (g) = 0.93 atm Initial pressure of H2O (g) = 2.7 atm Equilibrium ...
Q: a. NaOH + (NHa),PO4 → Na,PO, + H20 + NH, b. CH12 + O2 → CO2 + H20 C. MgaN2 + H20→ Mg(OH)2 + NH3
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for yo...
Q: A. How many mL of a 0.171 M aqueous solution of nickel(II) nitrate, Ni(NO3)2, must be taken to obtai...
A: A. Given, concentration of aqueous solution of nickel(II) nitrate, Ni(NO3)2 = 0.171 M Mass of Ni(...
Q: What is the pH of a solution containing 0.367 mol L-1 CH3NH2 and 0.169 mol L-1 CH3NH3+?
A:
Q: 12 The molar heat of combustion, A H;, of sucrose is -5643.4 kJ mol 1, If 0.250 moles of sucrose is ...
A: Given : 12. ∆Hc° of sucrose = -5643.4 KJ mol-1 Moles of sucrose = 0.250 moles Temperature = 16°C...
Q: Calculate the mass of ammonia (NH,) that contains a trillion (1.000 × 102) hydrogen atoms. ΝΗ. Be su...
A:
Q: Predict the reactants of this chemical reaction. That is, fill in the left side of the chemical equa...
A: Given products are :- NaF(aq) and H2O(l) Here NaF(aq) is Salt and H2O is water
Q: The chemical structure of lactic acid (C,H,O, is shown below. Highlight each atom that is in a hydro...
A:
Q: The standard enthalpy of combustion, A H:, of graphite is -393.51 kJ mol1. Calculate the change in s...
A: Given, Standard enthalpy of combustion of graphite = ΔH0c = -393.51 KJ/mole Calculate the standard...
Q: you only have the chemical equation, what information do you have? In order to convert between grams...
A: A chemical equation tells about the stoichiometric ratio between the reactants and products.
Q: 2. An alcohol solution contains 35.0 g of 1-propanol (C3H7OH) and 150 g of ethanol (C2H50H). (a) Cal...
A: Consider the given information is as follows; Mass of 1-propanol = 35.0 g (solute a...
Q: The standard enthalpy of combustion, A H: , of graphite is -393.51 kJ mol1. Calculate the change in ...
A: Answer- Data given- standard enthalpy of combustion of graphite is -393.51 kaj mol-1. Kindly refer a...
Q: Question 29 How many electrons are transferred in the given redox reaction? Zn(s) + 2AGNO3(aq) → 2Ag...
A:
Q: (A) CsBr (B) MgCl2
A: A compound is formed by the electrostatic interaction between the cation and the anion. The cation i...
Q: using the debye-huckel limiting law, calculate the activity coefficient of sulfate ion with u = 0.22...
A:
Q: Ib/ft at atmospheric pressure. 2. Calculate the quantity of heat in kJ to be transferred to 9.5 lb o...
A: Answer 2. Calculate quantity of heat in kg Q = mCVdT Q= ?...
Q: The standard enthalpy of reaction, AH rxn, for the reaction, CaO(s) + SO3(g) → CaSO4(s) is -401.5 kJ...
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: In an analysis of the following reaction at a certain temperature, Br,(g) + Cl½(g) = 2BrCl(g) the eq...
A:
Q: CaO(s) -635.5 NaHCO3(s) -947.7 Ca(OH)2(s) -986.59 Na,CO3(s) -1131 – 1432.7 - 1575.2 CaSO,() NACI(s) ...
A:
Q: ve IUPAC names
A:
Q: Which of the following is an example of a nonspontaneous process? O A hot skillet cooling Electropla...
A: Non spontaneous reaction is the reaction which require additional energy to occur.
Q: What is the name, starting materials, and intermediate for the attached molecule? Please type the n...
A: The given bicyclo compound can be numbered as below:
Q: The following syntheses have flaws in them. What is wrong with each? (a) .CH3 1. Cl2, FeCl3 .CO2H 2....
A: 1) Methyl group is a moderate electron donating group and it directs the incoming electrophile ortho...
Q: The reaction by which it goes into solution is: Mg(OH)2(s) Mg2+(aq) + 2 OH−(aq) a. Formulate the ...
A:
Q: 4. For each acid, give the formula of the anion bonded to hydrogen, then give the formulas of the ac...
A: Given : Acid are the compounds which gives H+ ions.
Q: The standard enthalpy of combustion, A H; ,of graphite is -393.51 kJ moli. Calculate the change in s...
A:
Q: Sove for the (F) freezing point of urea and (G) Percent Error. Given: A. Weight of urea = 2g B. W...
A: The depression in freezing point used to calculate is, ∆Tf=-Kfm∆Tf→freezing point depresssion Kf→c...
Q: For a process with AH 0 O AS > 0 O Never. ОДН> 0 Save for Later Attempts: 0 of 1 used Submit Answer...
A:
Q: Given the mechanism, drow Hhe ncduct 9 SN b. et SN
A: P.S.: According to the company guideline we have to answer only the first question, please kindly po...
Q: Calculate the work, in joules, done by a gas as it expands at constant temperature from a volume of ...
A: As the volume of gas is expanding from 2.00L to 5.00L isothermally . Therefore, it is work done by t...
Q: Which of the following is an example of a nonspontaneous process? O A hot skillet cooling O Solid wa...
A: Spontaneous reactions are those reactions that occur by itself and non spontaneous reactions cannot ...
Q: e pH if 0. cid?
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
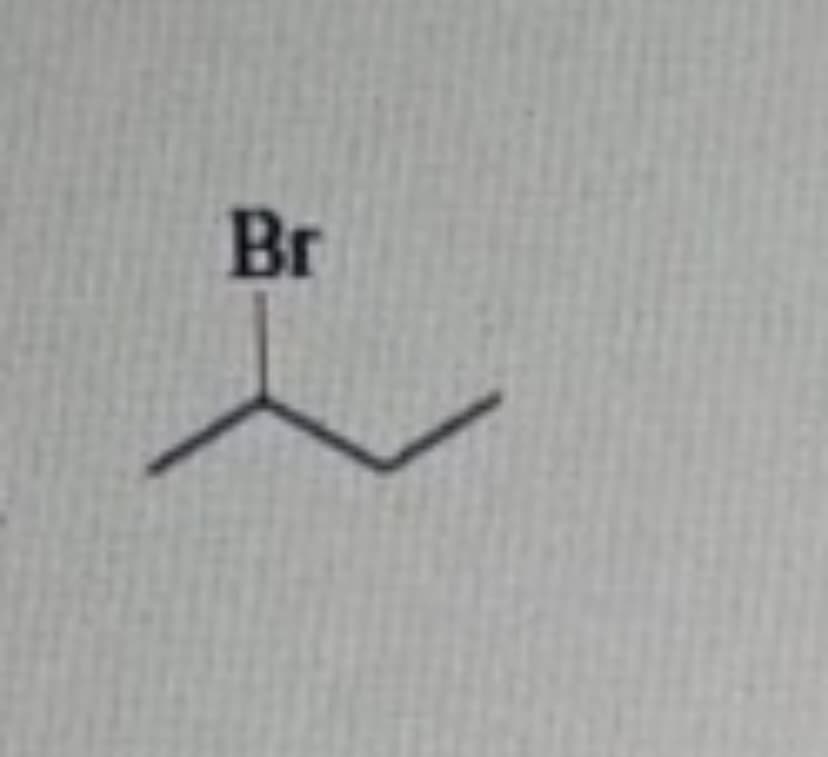
Step by step
Solved in 2 steps with 1 images


