Q: Use the information given to you below to solve/calcualte the rest of the table. Please show all wo...
A: Given : Formula of Hydrate: CuSO4*5H2O Weight of Hydrate and Evaporating Dish = 63.961 g Weight of E...
Q: SnO 2 + H 2 Sn + H 2 O balanced equations
A: A chemical equation is said to be balanced if the number of moles of different types of atoms are eq...
Q: Explain the Buffer Range and Buffer Capacity?
A: Buffer range : pH range in which buffer solution can resist the change in pH effectively is known as...
Q: The equilibrium constant for the reaction N2(g) + 3H2(g) ➡ 2NH3(g) is 1.0 x 10-1 at 5...
A: The change in temperature affect the rate of reaction. As the rate of reaction is changes the equili...
Q: What is the molality of a solution containing 7.78 g of urea ((NH2)2CO) in 203 g of water?
A: Given Mass of Urea = 7.78 gram Molar mass of urea = 60 gm/mole ( Fo...
Q: A compound with a molar mass of about 42 g/mol contains 85.7% carbon and 14.3% hydrogen by mass. Wri...
A: The molecular formula of a compound denotes the exact number and type of atoms present in it. This ...
Q: Theoritical and experimental values of lattice energies of a compound may differ Explain why the va...
A:
Q: Write the equations for the reaction between: H2O and HClO4 CH3CO2H and HCl
A: HClO4 + H2O → H3OClO4
Q: Pentene (C5H10) reacts with ethanoic acid to form pentyl ethanoate, the equilibrium CH3CO2H + C5H1...
A: Equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical equili...
Q: What is the equivalent value of T = 50° C in kelvins?We can use Equation (11.9) :T(K) T( C)+ 273 = 5...
A: The relation between temperature in degree C°C and degree Fahrenheit°F is, T(K)=T(°C) + 273.15
Q: Write IUPAC names for each compound. Specify the configuration of chiral centers
A: To write the IUPAC name of the below compound
Q: The progress of the reaction between Iodine ions and potassium iodate(V) can be followed by withstan...
A:
Q: Draw the Alkylating the β-Carbon via a Michael Reaction:
A: Michael reaction is also known as 1,4-addition reaction which has α,β-unsaturated carbonyl compound ...
Q: The data below was obtained for the reaction between Iodine and propanone in the presence of an acid...
A: From the given rate and concentration data we can determine individual order of substance as well as...
Q: What are the products of the following reaction?
A: The given amine is a secondary amine. Ketone undergoes protonation in the presence of acid and the r...
Q: Question is attached
A:
Q: Draw the organic product(s) formed when CH3CH2CH2OH is treated withfollowing reagent : H2SO4
A:
Q: Label each compound as chiral or achiral. Compounds that contain a single carbon common to two rings...
A: Form the given compounds, a and c are achiral due to the place of symmetry.
Q: What is the pH of a 0.310 M solution of HF (Ka = 6.8 × 10⁻⁴)?
A: Recall the expression of pH for weak acid as follows pH=12pKa-12logC Here HF is a weak acid with c...
Q: 4. Consider the following reaction: HBr 40°C a) Show a detailed mechanism for the reaction and give ...
A: Hello. Since the question has multiple subparts, the first three subparts have been solved for you. ...
Q: A solution is prepared by dissolving 396 g of sucrose (C12H22O11) in 624 g of water. What is the vap...
A:
Q: 4 Show how you would make the following ethers, using only simple alcohols and any needed reagents a...
A: The ether is a compound having an oxygen atom present between two carbon atoms. Alcohol is a compou...
Q: What absorbance corresponds to 1% transmittance? To 50% transmittance?
A: Given information, 1% transmittance and 50% transmittance
Q: Draw a structure for alkane. 2,3-dimethylbutane
A: Alkanes are saturated compounds which contain single bonds only. The general formula of alkanes is C...
Q: Pentene (C5H10) reacts with ethanoic acid to form pentyl ethanoate, the equilibrium CH3CO2H + C5H1...
A: The chemical equation for a reaction between pentene and ethanoic acid is CH3CO2H + C5H10 ----&g...
Q: How do some pairs of metals form alloys with structures that depend on the relative composition of t...
A: An alloy is a metallic substance composed of a metal with at least one other metal metal or nonmetal...
Q: Which of the following is more reactive and why? Explain using periodic trends. a) Lithium, sodium, ...
A: Reactivity of Group 1 and 2 elements increases when we move down the periodic table. a . Among the ...
Q: A common illicit synthesis of methamphetamine involves an interesting variation of the Birch reducti...
A: A common illicit synthesis of methamphetamine involves an interesting variation of the Birch reducti...
Q: a 5.000g sample that contained NaCl and BaCl2 *2H2O was heated. After heating the sample had a weigh...
A: Given : Mass of sample of NaCl and BaCl2*2H2O = 5.000 g And mass of sample after heating = 4.688 g
Q: Given the following redox potentials I2(aq) + 2e ➡ 2I-(aq) E=+0.54V S2O62-(aq) + 2e- ➡ 2...
A:
Q: Write the general formula for alkynes.
A: Hydrocarbons are organic molecules formed by carbon and hydrogen. It is further classified into satu...
Q: The decomposition of N2O5(g) follows 1st order kinetics. N2O5(g) → N204(g) + ½ O2(g) If 5.95 mg of N...
A: P
Q: For compound, indicate which group on the ring is more strongly activating and then draw a structu...
A: In the given compound there are NHCOCH3 and COCH3 group attached to the benzene ring. NHCOCH3 is a a...
Q: Specify the configurations (R or S) of chiral centers a and b in the chem3D structure below. F a bal...
A: Carbon a = R Carbon b = R By using priority according to CIP rules , absolute configuration of car...
Q: A weak monoprotic acid is titrated with 0.100 M NaOH. It requires 50.0 mL of the NaOH solution to re...
A: Given: Concentration of NaOH = 0.100 M Volume of NaOH = 50.0 mL (1 L = 1000 mL) ...
Q: Temperature a. sink water: 25.4°C °F b. ice-water: 0.2 °C = °F boiling water: 100.3°C = °F %3D с.
A: There are different units to measure temperature. Some of them are degree Celsius (°C) , Kelvin (K) ...
Q: Give reagents and conditions for the following reaction. Ph Ph, Br Br Br Ph Br Br Ph Br Ph Ph
A: A palladium-catalyst reaction in which the carbon group of hexabromobenzene is coupled with a carbon...
Q: For item 1-2 please refer to the problem on the picture. Please answer and choose the correct answer...
A: The balanced chemical equation is 16 Al + 3 S8→8 Al2S3 16 moles of Al react...
Q: The periodic table is a result of the periodic law. What observations led to the periodic law? What ...
A: In the periodic table, there are elements arranged according to the increasing atomic number.
Q: This question is concerned with acid/base. The ionic product of water may be taken as 10-14 mol2dm-6...
A: Introduction: The Titration is provided a graph that is called titration curve which contains the vo...
Q: Exactly 41.00 mL of 0.100 M NaOH is used to titrate a 25.00 mL sample of HNO3 . What is the concentr...
A: Volume of NaOH = 41.00 mL Concentration of NaOH = 0.100 M Volume of HNO3 = 25.00 mL
Q: A sample of glucose, C6H12O6, contains 1.250 * 1021 carbonatoms. (a) How many atoms of hydrogen does...
A: 1 molecule of glucose contains 6 atoms of C, 12 atoms of H and 6 atoms of O. Molar mass of glucose ...
Q: The data below was obtained for the reaction between Iodine and propanone in the presence of an acid...
A: Let us consider the order of the reaction with respect to CH3COCH3, I2, and H+ are x, y, and z respe...
Q: Specify whether the configuration of the chirality center in the structure is R or S. (Other terms u...
A: Configuration of chiral center is S. Configuration of chiral center is determined by priority of gro...
Q: Name the two monosaccharide units of which trehalose is composed
A: Trehalose is also known as Mycose or Tremalose
Q: Balance the following equation KCIO3----> KCI+O2 a) how many grams of KCI are produced from 250 gram...
A:
Q: Define phospholipids ? Why it is a major component of the cell membrane ?
A: Given term, Phospholipids
Q: The solubility of CO2 in water at 25°C and 1 atm is 0.034 mol/L. What is the solubility of CO2 in a ...
A: where p is the partial pressure of the solute in the gas above the solution, c is the concentration ...
Q: H₂ may act as a reducing agent or an oxidizing agent, de-pending on the substance reacting with it. ...
A: Using sodium(Na) as the other reactant : 2Na(s) + H2(g) → 2NaH(s) Here we can see that the oxida...
Q: Which of the following statements is TRUE? If Q < K, the reaction proceeding to the left is ...
A: Reaction quotient(Q) is the ratio of concentrations of products at numerator with the concentrations...
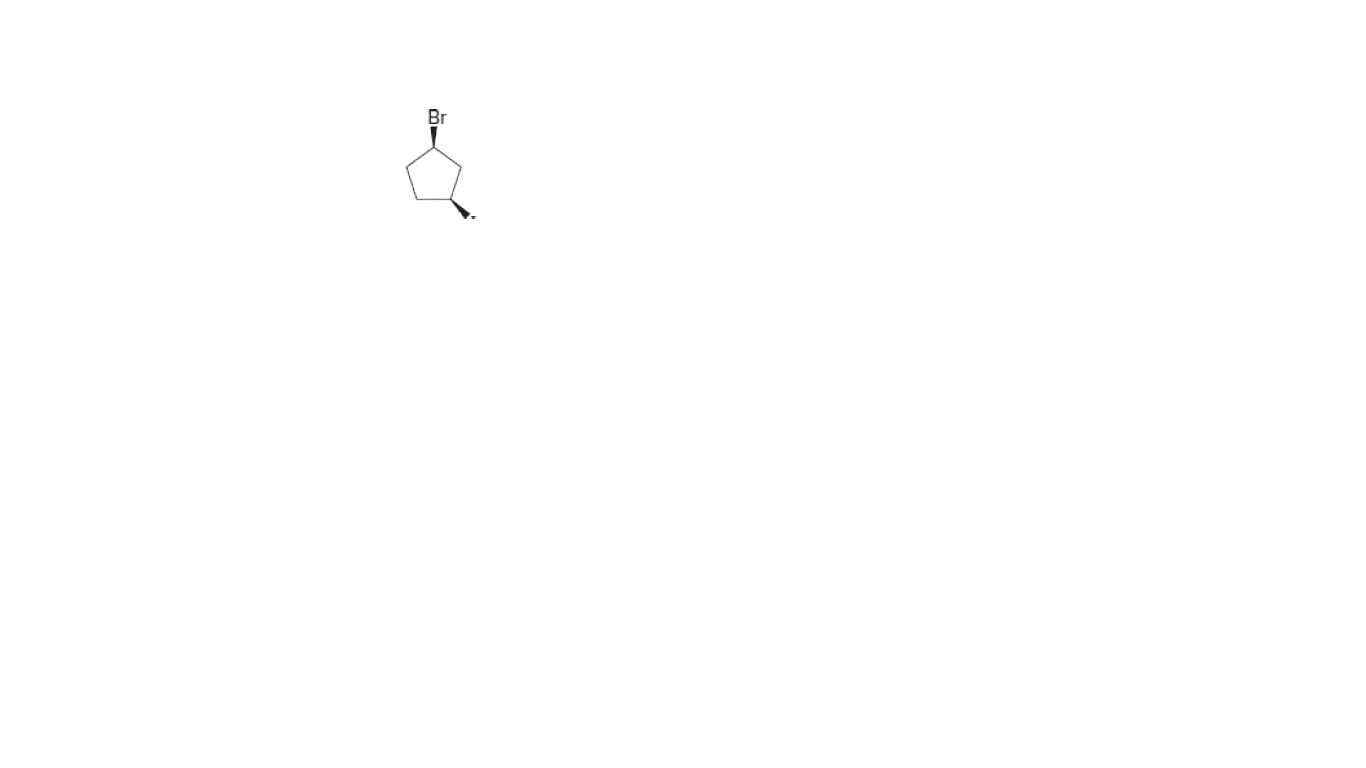
Give the IUPAC name for attached compound.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images


