Q: how does x/0.6 cpme from? and x is moles or concentration in step 4?
A: Answer: This question is demanding the elaboration of certain values used in this question to solve…
Q: Gg.209.
A:
Q: A 44.4 mL sample of a 0.300 M solution of NaCN is titrated by 0.100 M HC1, K₁ for CN is 2.0 × 10-5.…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: A solution contains 6.44x10-3 M potassium phosphate and 1.08x10-2 M sodium carbonate. Solid zinc…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: The equilibrium constant for the reaction below is K=5.87×105 at 277.0ºC. 3 NO (g) ⇄ N₂O (g) + NO₂…
A:
Q: 3) Given the precipitation reaction: Ag+ (aq) + Cl(aq) = AgCl(s) the K value for this reaction is…
A: Solution: The reaction illustrated in: a) is heavily displaced to the right, that is products, and…
Q: The correct name for the compound given 0=C-CH₂-CH₂ H -N -CH₂-CH₂-CH₂ N-propylpropanamide…
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: Enter the chemical formula of a binary molecular compound of hydrogen and a Group 7A element that…
A: The question is based on the concept of chemical compounds. we need to identify a compound of…
Q: 1. For each or the following molecules, write M, 1º, 2º, 3º, V, or A, if it is a methyl, primary,…
A:
Q: Classify the following reaction as: addition, elimination, substitution, or rearrangement.
A:
Q: 4. The ester you drew in Question #1 has a melting point range of 32-35 °C. The methyl ester of…
A: We have to explain the term mono-unsaturated and draw the structure of methyl ester of…
Q: Draw the major organic substitution product or products for (2R,3S)-2-bromo-3-methylpentane reacting…
A:
Q: 3. What product is formed when HO(CH₂) CHO is treated with each reagent? a. NaBH, CH₂OH b. PCC c.…
A:
Q: The following molecules parent chain has the ROOT - pent: True False
A:
Q: A student performs a flame test on two white powders she finds in the lab. Each powder produces…
A: During a flame test, a sample is heated in a flame, causing the atoms to become excited and jump to…
Q: A concentration cell is set up with two nickel electrodes and two solutions of Ni(NO3)2. One cell…
A: Nernst equation gives the relation between cell potential, standard cell potential, and activity of…
Q: 13.Phosgene COCl₂ dissociates as follows: COC1₂(g) CO (g) + Cl₂(g). At 720 °C the Kc value is 3.63 X…
A: % dissociation of phosgene is calculated as per formula shown below.
Q: Draw the major product of the aldol addition reaction between two of these aldehydes with the…
A:
Q: 20.5 Provide reasonable mechanisms for the following transformations. L + Brg H", quench
A: Grignard reagent is the organo magnesium reagent which involves the C-Mg bonding. Due to presence of…
Q: Answer the following questions regarding the decomposition of arsenic pentafluoride, AsFs(g). (a) A…
A:
Q: 9.Suppose 1.000 mole of CO and 3.000 mol of Hydrogen are put in a 10.000 L vessel at 1200 K. the…
A: The equilibrium constant KC, is the ratio of the equilibrium concentration of the products to the…
Q: 1. Calculate the molar mass (molecular weight) of the following compounds a. K,PO b. Al(NO₂),
A: The molar mass of any compound is the total sum of molar mass of all the atoms or elements present…
Q: When 646. mg of a certain molecular compound X are dissolved in 60.0 g of benzonitrile (CH₂CN), the…
A: Given Mass of benzonitrile = 60 g Mass of X = 646 mg = 0.646 g Temperature (Tf) = -13.2
Q: How to calculate the concentration of sodium thiosulphate in titrimetric analysis of ascorbic acid…
A: Titrimetric analysis is a technique used in analytical chemistry to determine the concentration of a…
Q: From the graph, determine the solubility of KNO3 at 35° C. 2. From the graph, determine the…
A: To find out the solubility of KNO3 at 35°C by using the graph of gram of KNO3 per 100 g of water…
Q: Problem 7 What is the pH of a 0.10 M ammonia solution: NH3(aq) + H₂O(1) NH(aq) + OH (aq), K =…
A: We have to calculate the pH of the solution
Q: Butane is a common fuel used in cigarette lighters and camping stoves. Normally supplied in metal…
A: Given, The vapor pressure of butane at 25.0°C is 2.30 atm. The vapor pressure of butane at 126.0 °C…
Q: What is the Chemical equation for nickel iron alkaline and nickel cadmium battery
A: A question based on battery cells. Electrochemical equations representing overall cell reactions of…
Q: Give detailed Solution with explanation needed...if you don't give both sub parts then don't give…
A: We have been given two questions.In one question,we have been asked the name of given organic…
Q: 11. Which conjugate base should be the strongest out of these choices? (pKa values of their…
A: Useful Concepts: Stronger is the acid, weaker is its conjugate base. pKa and pKb are related as pKa…
Q: In the answer box provided, show a series of reactions that would produce the following molecule…
A: The series of reactions involves in the conversion of acetylene to the given ketone.
Q: Select one or more: O₂N-NO2 SF6 NO NH3 BH3 HCI H₂O
A:
Q: Design a synthesis of 3-buten-2-thiol from 3-buten-1-ol. Part 1 out of 8 SH A Choose the best option…
A: The retrosynthesis of any target molecule is a good method to analyse the easier synthesis pathway…
Q: 7. The molecule pictured to the right has which functional group? A. aldehyde B. alcohol C.…
A: Functional group represent a group of atoms or bonds inside a substance whic is responsible for the…
Q: Calculate the PH of a solution that is 0.0011M in HCl and 0.012M in HClO2. Express your answer using…
A: In the given question, we have to find the pH of solution.
Q: 17. Which statement regarding acid-base relationships is true? A. The pH of a solution and the…
A: Given statements are A. The pH of a solution and the concentration of hydroxide are directly…
Q: How many radon-222 nuclei (f/2= 3.82 d) decay per minute to produce an activity of 7.19 pci?…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Data vial + NaOH mass empty vial mass measured volume 100 liquid mass (V x density) NaOH used soln…
A: We need to calculate: (a) heat absorbed (b) enthalpy change (c) molar enthalpy change (d) % error
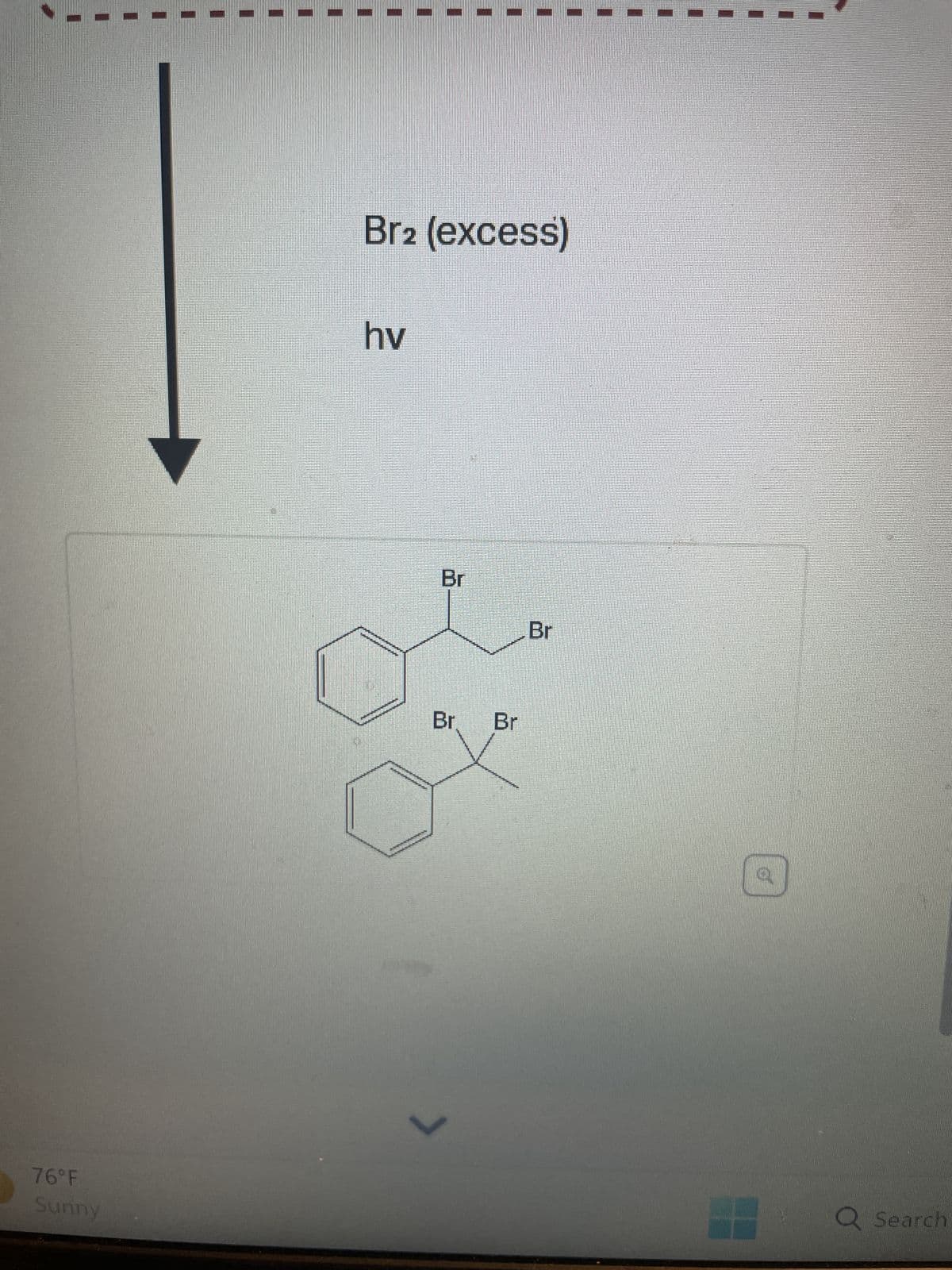
Q: Br2 (1 equiv) hv Br
A:
Q: Calculate the solubility of CaCO3 in water at 25 °C. You'll find Kp data in the ALEKS Data tab. sp…
A:
Q: Calculate the molar solubility of SrC₂O4 in a solution that has a fixed H30 concentration of (Ksp…
A: Given Ksp of SrC2O4(s) = 5.0*10-8 For H2C2O4, Ka1 = 5.60*10-2 , and Ka2 = 5.42*10-5 .
Q: 2 1. The salt, sodium carbonate, is composed of carbonate (CO₂) anion(s) 2. The salt, copper (II)…
A: Given than, Different salts are given,we have to determine number of ions when salt gets disociated.
Q: 1. In which species does bromine have an oxidation number 0? 2. Balanced molecular equation for…
A:
Q: The solubility of Mg(OH), in water at 25 °C is measured to be 0.0096 Use this information to…
A:
Q: Calculate the concentration of CuS in a solution in which H₂O+ is held constant at ( Kap (CuS) = 8.0…
A: Given: Ksp (CuS)=8.0×10-37 K1(H2S)=9.6×10-8 K2(H2S)=1.3×10-14 For problem (a) [H3O+]=0.25M and For…
Q: Give detailed Solution with explanation needed..please give answer sub parts
A: The following compound are recognized as acidic basic as per follow.
Q: ncorrect Question 17 What is the product(s) of the following oxidation reaction, if any? SH S S This…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: 18. Which statement(s) is/are true regarding chiral carbons? A. All chiral carbons must have a…
A: The chiral carbon atoms are carbon atoms that are attached to four different atoms or groups of…
Q: Calculate the solubility of BaCrO4 in water at 25 °C. You'll find Kp data in the ALEKS Data tab.…
A: Ksp of BaCrO4 = 1.17 × 10-10 To find out solubility of BaCrO4 , we need to write dissociation…
Q: Name and identify all phase transitions
A: Phase transitions refer to the changes that occur in the physical state of a substance, such as from…
Draw a possible
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Hand written soln only.Br2(at)+2NaI(aq) --> 2NaBr(aq)+I2(aq) Cl2(aq)+2NaBr(aq) --> NaCl(aq)+Br2 Which of the three halogens would you predict is the most stable, upon mixing with other halogens? Justify your answer.Group A: solution conductivity (MS) NaCl 6594 CaCl2 13188 AlCl3 16311 Group B: solution conductivity (MS) HCl 21809 Hc2H3O2 639 H3PO4 8805 For H3PO4, does the subscript "3" of hydrogen in this formula seem to result in additional ions insolution as it did for AlCl3in Group A? Explain.

