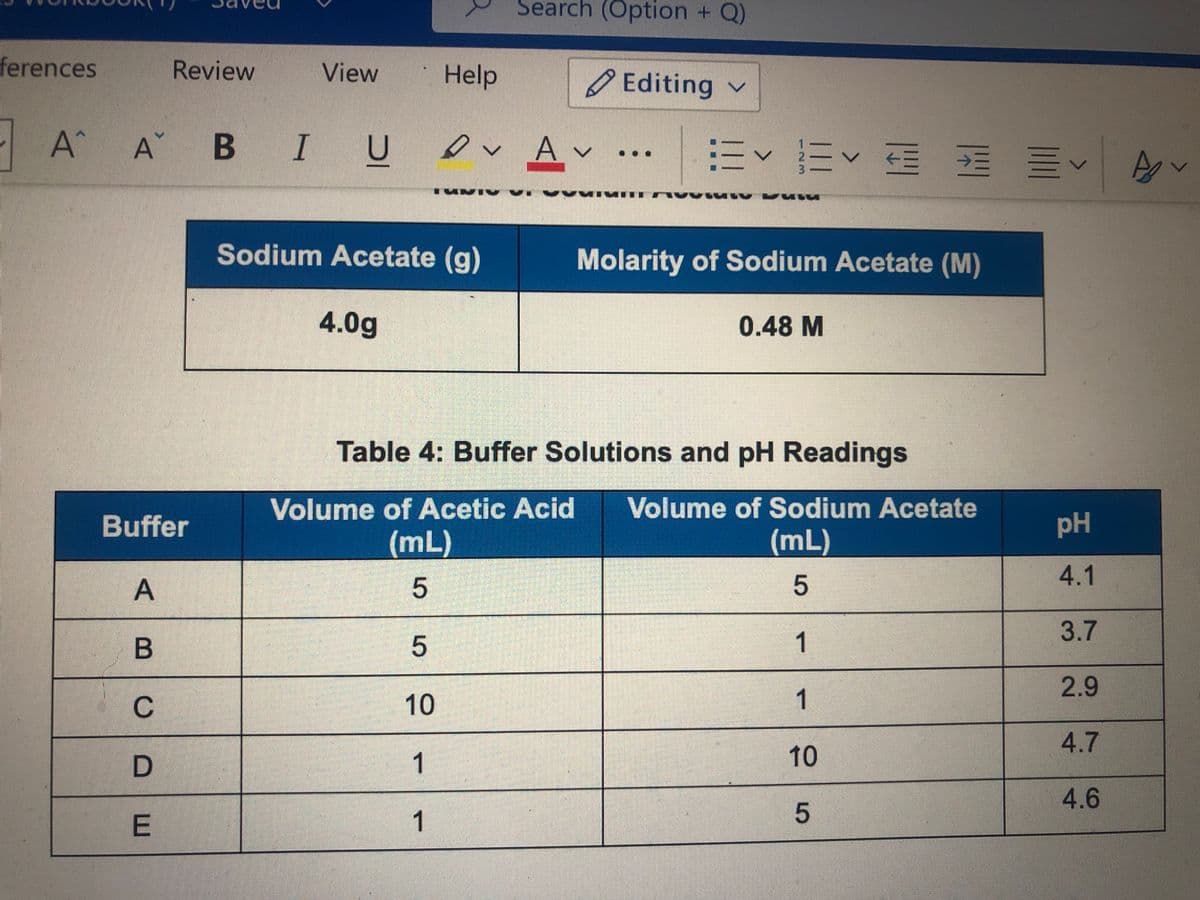
Buffer C has twice as much acetic acid as buffer B, but the same amount of sodium acetate a.describe the difference in pH in these two solutions b. Why do you think the pH is different, based on what you know about acid pH values?
Buffer C has twice as much acetic acid as buffer B, but the same amount of sodium acetate a.describe the difference in pH in these two solutions b. Why do you think the pH is different, based on what you know about acid pH values?
Chapter14: Principles Of Neutralization Titrations
Section: Chapter Questions
Problem 14.38QAP
Related questions
Question
Buffer C has twice as much acetic acid as buffer B, but the same amount of sodium acetate
a.describe the difference in pH in these two solutions
b. Why do you think the pH is different, based on what you know about acid pH values?
Transcribed Image Text:Search (Option + Q)
ferences
Review
View
Help
Editing v
A BI U Av
蛋 三、
Sodium Acetate (g)
Molarity of Sodium Acetate (M)
4.0g
0.48 M
Table 4: Buffer Solutions and pH Readings
Volume of Acetic Acid
Volume of Sodium Acetate
Buffer
pH
(mL)
(mL)
4.1
5
3.7
В
1
2.9
C
10
1
4.7
1
10
4.6
1
5
5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning