Buffers are aqueous solutions that tend to resist changes in pH when small amounts of strong acid (H+) or base (OH-) are added. Which of the following are buffers in the biological systems? O A) Carbonic acid bicarbonate buffer system O B) Phosphate buffer system OC) Protein buffer system O D) All of these.
Buffers are aqueous solutions that tend to resist changes in pH when small amounts of strong acid (H+) or base (OH-) are added. Which of the following are buffers in the biological systems? O A) Carbonic acid bicarbonate buffer system O B) Phosphate buffer system OC) Protein buffer system O D) All of these.
Chapter9: Parenteral Medication Labels And Dosage Calculation
Section: Chapter Questions
Problem 1.2P
Related questions
Question
78 79
please answer all
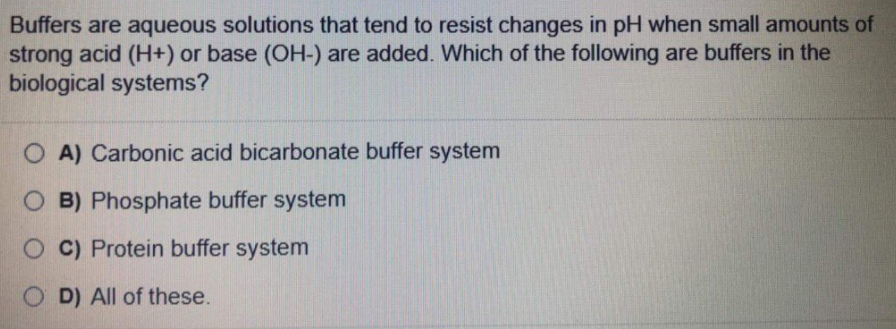
Transcribed Image Text:Buffers are aqueous solutions that tend to resist changes in pH when small amounts of
strong acid (H+) or base (OH-) are added. Which of the following are buffers in the
biological systems?
O A) Carbonic acid bicarbonate buffer system
O B) Phosphate buffer system
O C) Protein buffer system
O D) All of these.

Transcribed Image Text:Neutralization reaction is the combination of OH and H* forming water and the salt. If
someone has accidentally taken muriatic acid, what will you do?
O A) Let him drink 8 glasses of water.
O B) Let him take milk of magnesia (Mg(OH)2).
O C) Let him take a shower.
O D) Let him take vinegar (5% acetic acid).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you



