Chapter6: Oral Medication Labels And Dosage Calculation
Section: Chapter Questions
Problem 9.4P
Related questions
Question
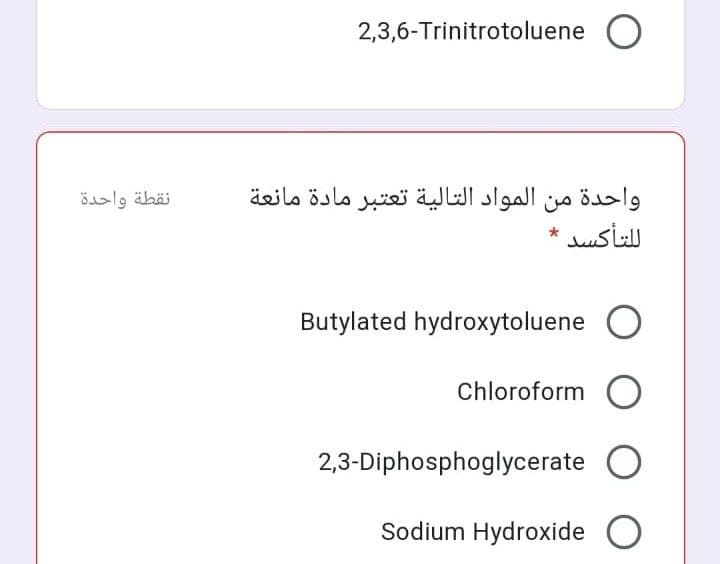
Transcribed Image Text:2,3,6-Trinitrotoluene O
واحدة من المواد التالية تعتبر مادة مانعة
ل لتأكسد
نقطة واحدة
Butylated hydroxytoluene
Chloroform O
2,3-Diphosphoglycerate
Sodium Hydroxide O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you



