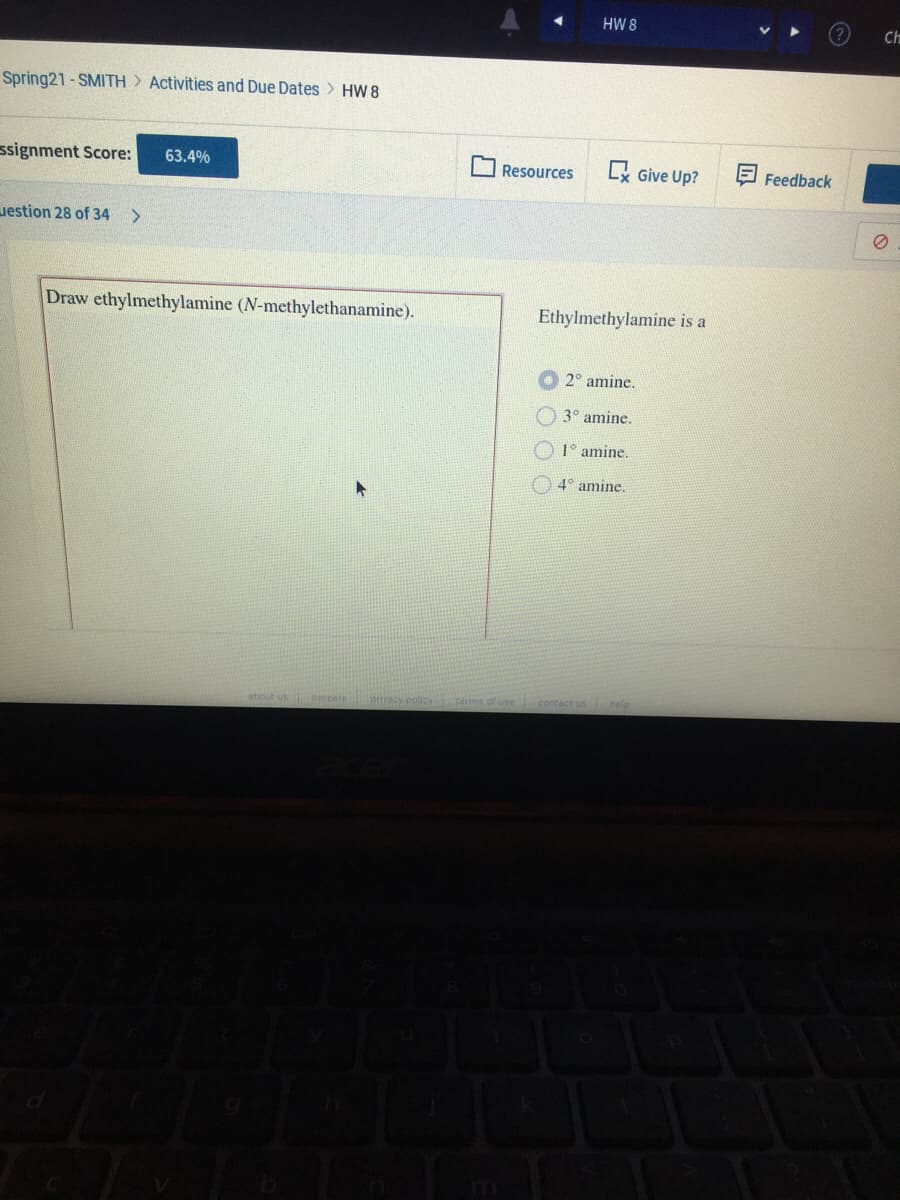
Draw ethylmethylamine (N-methylethanamine). Ethylmethylamine is a O 2° amine. 3° amine. O 1° amine. O 4° amine.
Q: If we attached an amine group (NH₂) to Carbon 4, what type of amine will be the result? A. Primary…
A: Amines are organic compounds that contain nitrogen atoms with a lone pair.
Q: The compound below is an aldehyde O a ketone a carboxylic acid
A: Aldehyde, ketone, and carboxylic acid are three different functional groups in organic chemistry. An…
Q: When you made soap, first you dissolved vegetable oil in ehanol. What happened to the ethanol during…
A: Soaps are sodium or potassium fatty acids (that is long carbon chain) salts, produced from the…
Q: Daclatasvir is formulated at the dihydrochloride salt. Which functional group in Daclatasvir will be…
A: Daclatasvir is one of the potent, first-in-class, and a selective inhibitor of the nonstructural…
Q: Arachidic acid has a lower melting point than ___________ because the former has a shorter…
A: Fatty acids contain a long hydrocarbon chain attached to a carboxylic group. Fatty acids vary from…
Q: Is sodium propanoate (the sodium salt of propanoic acid) as effective a soap as sodium stearate (the…
A: Soap is the cleansing or emulsifying agent, which helps to remove dirt.
Q: Order the following from polar to nonpolar. Acetone Nitromethane
A: A condition or state of a molecule or an atom consisting of negative and also positive charges,…
Q: enzyme Nitrite reductase has which of the elements as its constituent ?
A: Enzymes facilitate speed up chemical reactions within the human body. They bind to molecules and…
Q: Define the following terms:a. allyl groupb. epoxidec. SAM d. PAPSe. phase I reaction
A: In chemical structure, the group of atoms that replace the hydrogen atoms on the parent hydrocarbon…
Q: Which functional group is not contained in prostaglandin E1?* ketone secondary alcohol…
A: Prostaglandins are produced locally, in contrast to other hormones, which are produced by glands and…
Q: Classify the following as an Alcohol, Phenol, Thiol, Ether, Aldehyde or Ketone. ОН CH HÇ HC CH CH OA…
A: Organic compounds are the chemical compounds that deals with the bonding of one or more carbon atom…
Q: NH O purine O pyridine H. Consider compound 2. Compound 2 is classified as a purine NH pyrimidine…
A: Introduction: Compound one is pyrimidine. (Uracil) Compound two is a purine. (Guanine) Compound…
Q: List advantages and disadvantages to phenolic compounds.
A: Phenols are the chemical compounds which consist of a hydroxyl group (-OH) bonded to an aromatic…
Q: Which of the compounds below are aromatic?
A: Aromaticity of a compound is dependent on the following properties: the compound should be cyclic…
Q: Identify ALL the CORRECT statements regarding the compound shown below. SELECT ALL THAT APPLY A.…
A: Ans- Given structure is of Guanine. # Guanine is a nitrogenous base found in nucleic acids such as…
Q: In the Equation: HCO3- + HCl --------> Conjugate Acid + Conjugate Base a ) Which reactant…
A: An acid base reaction is a type of chemical reaction that involves transfer of one or more hydrogen…
Q: Identify the conjugate acids and bases in the following pairs of substances. (CH3), NH / (CH3)3N…
A: An acid is a molecule that can donate a proton to the solution. The presence of acid in a water…
Q: What type of compound is this? CH3CN Primary Amine Secondary Amine Tertiary Amine Quaternary…
A: A moiety or a specific group of atoms present in a molecule that possess their own characteristic…
Q: Which do you think has a higher boiling point, pentane or neopentane (2,2-dimethylpropane)? Why?
A: The boiling point is the temperature at which the pressure of the liquid changes to the state of the…
Q: Mention at least two simple chemical tests by which you can distinguish between benzaldehyde and…
A: Benzaldehyde will respond to the Tollen's reagent where as benzoic acid will not respond to it.
Q: What coefficients must be placed in the following blanks sothat all atoms are accounted for in the…
A: As per the law of conservation of mass, mass can neither be created nor destroyed. This also applies…
Q: In the following reaction in aqueous solution, the acid reactant is and its conjugate base product…
A: An acid–base reaction occurs when one or more hydrogen ions, H+, are exchanged between species that…
Q: Which of the following statements concerning the titration curve of glycine is correct? 2.0 E D 1.5…
A: Glycine is a hydrophobic amino acid with an alpha-carboxylic group and an alpha-amino group. Glycine…
Q: 2. Of the following amino acids, which contains an alcohol? a b HN. HN. HN. HN. HO, HO, HO, SH NH.…
A: Introduction: Amino acids are biomolecules comprise of two functional groups; an amino group (-NH2)…
Q: H What is this molecule? N-C-C OH R Name this molecule Explain
A: Protein is made up of a monomeric unit called amino acid and there are 20 amino acids that code for…
Q: Which of the following sugars is a non-reducing sugar? CH,OH CH,OH H/H H/H H. H (1) но OH H -어 он ÓH…
A: Carbohydrates are biomolecules that contain carbon, hydrogen, and oxygen atoms in the ratio (CH2O)n.…
Q: What are the effects of the following solvents when mixed with lipids? What type of hydrolysis is…
A: Lipid metabolism is the synthesis and breakdown of lipids in cells. This process entails the…
Q: Classify the alcohol with the following structure: NHCOCH, CH,OH Select one: a. none of the other…
A: Step 1 The carbon next to the carbon with which the alcohol is attached determines whether it is…
Q: alkyl halide
A: Alkyl halides (haloalkanes) are a kind of chemical compound which are obtained from alkanes and…
Q: are fragrant carboxylic acid derivatives. O Nitriles O Esters O Amides O Anhydrides O Acid chlorides
A: The derivatives of carboxylic acids are nitrile, acid chloride, acid anhydride, amide, and esters.…
Q: H3C- -CH3 CH3 H3C CH3 Но ČH3 -CH3 A B D H2N NH2 OH CH3 -CH3 -CH3 H3C- CH3 H3C- H3C CH3 H;C ČH3 F G H
A: A functional group is a substituent in a molecule which governs it's characteristics of chemical…
Q: Why do you think compounds containing the heavy metal lead are poisonous?
A: Heavy metals are chemical elements that have high density and very toxic or poisonous even at their…
Q: a. What is a heterocyclic amine? What was the most interesting example you found out about? Why was…
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: Which of the following is true of thymine and uracil? A Thymine has a methyl group at C-5 and Uracil…
A: Answer : Option D is correct - thymine has methyl group but uracil doesn't have it.
Q: Below is the structure of butyl alcohol. Based on the results of the experiment you just performed,…
A: Butyl alcohol is known to effect cell permiability.
Q: Identify the Lewis acid in the following reaction: A. BF3 B. F C. BF4 D. None of these is an acid.…
A: Boron trifluoride (BF3) is a Lewis acid because it is an electron-deficient molecule that may take…
Q: NH2
A: The given structure has three functional group.
Q: Explain the widely used drugs contain a benzene ring ?
A: Benzene Ring : It is an organic chemical compound with the molecular formula C6H6. The benzene…
Q: A secondary alcohol will be obtained if we attached -OH group to w carbons? Check all that applies.…
A: An alcohol is an organic compound with a hydroxyl (OH) functional group attached to a carbon atom of…
Q: 4. -C-O-C- A compound that contains the above alcohol alkoxy group antioxidant dehydration reaction…
A: Biological molecules are composed of different functional groups which include hydroxyl, methyl,…
Q: Tetrabromofluorescein is a purple dye often used in lipsticks. if the dye is purple, what color does…
A: The dye is a colored substance that constructs a chemical bond with the substrate to which it would…
Q: Which of the following would be expected to form hydrogen bonds with water? Choose all that apply. H…
A: Hydrogen bond is a type of intermolecular force. It exists in molecules in which highly polar bonds…
Q: Which of the following compounds would be most soluble in carbon tetrachloride, CCl4? a. NH3 d.…
A: Carbon tetrachloride is an organic compound. It has no flammability, used in refrigerators, fire…
Q: Which of the sugars in the following figure is/are D sugars? Choose all correct answers. CHO CHO CHO…
A: First question: D-configuration refers to any chemicals that can be linked to (+)-glyceraldehyde…
Q: Which of the following shows B-D-Gulose? A) B) CH2OH CH2OH OH OH OH OH H. OH OH H H ÓH C) D) E)…
A: Carbohydrates are polyhydroxy aldehydes or ketones which are associated with reducing property and…
Q: 1. Alkyne 2. Amines 3. Alcohol 4. Carboxylic Acid 5. Carboxylic Ester 6. Ester 7. Amine 8. Benzene…
A: Alkane: represented by CnH2n+2. They are saturated hydrocarbon. Example : Methane, found in coal…
Q: If the dehydration reaction of an alcohol is successful, what changes would be seen in the IR…
A: Biochemistry is a branch of biology that deals with the study of chemical processes within living…
Q: Which one of the following substances would you expect to be most soluble in water? A) Butanol B)…
A: solubility in water depends on various factors one of which is the hydrogen bonding formed by the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- TRUE OR FALSE 23. Increased temperature increases Km. 24. Competitive irreversible inhibitors cannot be displaced by the original substrate.n the stages of caffeine isolation why calcium chloride, dichloromethane and toluene are usedCan yiu answer the following.: 1. Basuc of sifnal trwnsduction pathway in alcohol 2. Specific of cellular response and organismal response in alcohol
- Exposure to ethanol _____ the daphnia’s _____. This change would be evidence to classify ethanol as a _____.A person (70kg) eating locally caught fish from water contaminated with 100ppb TCE and 100ppb aniline. The bioconcentration value for TCE and aniline in fish is the same at 1.06mg TCE (or aniline) per kg fish, respectively. Fish is only exposed to these contaminants in its environment. About 54g of fish is consumed in 350days/yr for 30years. Assume bioavailability value of 1. The potency factor for TCE is 1.1 × 10$# kg.day/mg and reference dose is 1.00 × 10$% mg/kg.day. Whereas the potency factor for aniline is 5.7 × 10$" kg.day/mg and reference dose (based on drinking water) is 1.60 × 10$& ??/L. Unlike aniline, TCE is not a carcinogenic compound. There are 70yrs in a lifetime and 365day/yr. - Using chronic daily intake calculation, determine the non-cancer risk (number of people in one million) for the above case.Methadone elimination has very large patient to patient variability. A slow metabolizer of methadone who metabolized at t1/2 = 40hr. Is given a starter dose. They will first reach what is considered steady state at about: 1. 2.5 days 2. 6.5 days 3 4.5 days
- PLEASE READ EVERYTHING SENT... I keep getting questions Half done because there is no patience to read it all. How does someone Draw this Chemical Equation? I would like to draw a picture This was the information given by ChatGBT and it wasn't helpful to me so wondering if someone would be able to help draw a picture:The chemical equation for the oxidation of benzoin to benzil using sodium hypochlorite as the oxidizing agent can be represented as follows: (CHECK PICTURE SENT)This equation shows the conversion of benzoin (C14H12O2) to benzil (C14H10O2) in the presence of sodium hypochlorite (NaOCl), resulting in the formation of sodium hydroxide (NaOH) and water (H2O) as by-products.PLEASE DONT CANCEL. ILL UPVOTE WITH ALL OF MY ACCS. Make a drug study study about cetirizine (diphenhydramine hydrochloride) drug and fill up the corresponding details in the table. - The drug study shall be patient-based, and based on the informations provided below. - Nursing responsibilities would be in ADPIE form. MAIN DRUG: cetirizine (diphenhydramine hydrochloride) INFORMATIONS: Cetirizine Age: 18/F Occupation: Basketball player Weight and BMI: 50 KGS/22 Blood pressure: 100/70 Heart rate: 60 Respiratory rate: 15 Temperature: 36.2 Diagnosis: Allergic reaction with eating shrimp with facial angioedema Chief complaint: Swollen ears and mouth Prescription: Cetirizine PO 5 mg OD1. first coupling enzyme in enzymatic method for bicarbonate analysis a. malate dehydrogenase b. bicarbinate oxidase c. carbonic anhydrase d. pyrivate kinase e. none of the choices 2. color of the flame produced when using in AAS in potassium determination a. orange b. none of the choices c.. yellow d. red e. purple 3. preferred additive for chloride determination .a. EDTA b. sodium heparin c. none of the choices d. sodium citrate c. potassium oxalate
- Which of the following substances does not participate in the CalvinBenson cycle? a. ATP b. NADPH c. RuBP d. PGAL e. O2 f. CO2Please answer the all questions. I'll definitely give a like. Eddard has been stung by a swarm of bees. Given his allergies, he self-administered adrenaline intramuscularly, though was rushed to the emergency room for further assessment. He was administered a cocktail comprising aminophylline, hydrocortisone and chlorphenamine. a. What would the implication be if the dose of aminophylline was accidentally doubled? Justify your answer. (1) b. Should Eddard not have had access to injectable adrenaline, would an oral administration of chlorphenamine have resolved his symptoms? Justify your answer.d.) Dosage forms available in the market (ex. tablet) and matter classification (ex. solid/liquid/gas – homogeneous/ heterogeneous E.)Dosage and regimen (ex. take #1 500 mg tablet every 4 hours) f.)Pharmacologic Use or Indication. of a precipitated sulfur or the one at the third row only? (thank you so much to the experts who's going to answer this and has answered me with the first part this is extremely helpful for me ? sorry for this note ?)





