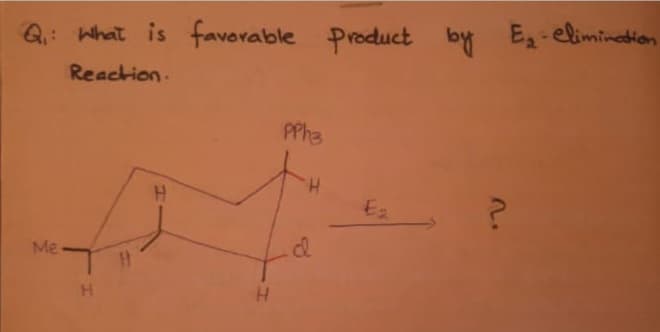
Q: Draw the MAJOR products A - E for the following reactions. i. KMNO4, HO", heat SOCI, A В ii. H;O" i.…
A:
Q: Which reactant(s) would form the products in the following reaction? Lxs NANH- NH) 2. H,0 b. Od. All…
A:
Q: What is the product of this reaction? excess OH OH но- но он ???? рyridine OH OH OH HO но он но. но…
A: Given is acetalization reaction.
Q: Give the major product of the following reaction. Zn(Hg), HCI CI heat AICI3, heat CI I There is no…
A: When an atom in the aromatic ring is replaced by an electrophile is known as electrophilic aromatic…
Q: 14C labelled LOTS RCO „H 6. product; Product of this reaction is : no label LOCOR -OCOR (a) (c) both…
A:
Q: Alkyl halides undergo elimination reactions with Brønsted–Lowrybases. The elements of HX are lost…
A:
Q: 애 H2SO4 180°C
A:
Q: 1. Draw the structure of the intermediate products in the following reactions: (C,Hz)jP-CH CH3 AICI,…
A: Steps: Friedel-Crafts acylation Wittig reaction Addition of HBr to alkene Elimination Nucleophilic…
Q: Find He appoprilate reagens for etficlent counveuieh of Starting meterial to podust a) OH OH b) OTs…
A: The reagent(s) in each reaction is shown below along with mechanism also
Q: O fmd out the frost (M) tor the ow toansihon ometals forllowing Comyoun ds Cll them obey 18 e ete…
A:
Q: Which is the best route to the compound? Me Me MeO O The following reaction scheme: AICI3 H2NNH,…
A: The best sequence is that in which we get maximum yield of product is best Sequence.
Q: Give the major product(s) of the following reaction. РСС он -? HO. H. OMe HO. There is no reaction…
A:
Q: он HO ČHO ABC DEF GHI
A: ->Reaction of ether with HI produce alkyliodide and alcohol. ->For strength of acids we see…
Q: он Mẹ 4. HSO4 → Product and name of the reaction is : NH – C – Me NH – C – Me (Hoffmann bromamide…
A: Beckman rearrangement is a rearrangement of oxime functional group in presence of acid.
Q: (a) OH, H,SO, (cat.) Но
A: Hi. You have posted seven questions in a single session and we are entitled to answer first…
Q: Are all of these reachchs regiosciective and how do you Aind it ? 1) 1,3 dimethylcycloher-1-ene…
A: Regioselective reactions are those reactions where one site is preferred over another. Because…
Q: Rank the following alkyl halides in order of increasing SyI reactivity. Br Br B: Br C: А: A B C…
A: In SN1 reaction formation of carbonation takes place so the reactivity of that alkyl or Aryl halide…
Q: Which isomer of 1-bromo-2-tert-butylcyclohexane reacts faster when refluxed with potassium…
A:
Q: OH H2SO4 H20 4-methylpentan-2-ol b. Which alkene product will will be favored and why? Discuss this…
A: In given reaction, a secondary alcohol (4-methyloentan-2-ol) is treated with sulfuric acid solution.…
Q: 1) LIAIH, H2N- 2) H,O* SOCI, но
A:
Q: Which of the following reagents is CORRECT for the reaction below. HO O KMNO4 O H2CrO4 O…
A:
Q: Draw all resonance structure contributors that result from ortho position of the nucleophile. Raw aL…
A:
Q: 1. Draw the structure of the intermediate products in the following reactions: (CH)Þ-CH CH3 AICI, ?…
A:
Q: Br TMSCI 1. Mg, Et20 Bu4N F Но EtzN 2. THE 3. HС (aq) Page 7
A:
Q: 2. (a) Predict the products of the following reactions with mechanism : to DCC Ti) H3C- -COOH →?…
A:
Q: What is/are the major product(s) of the following substitution reaction? Br CH₂OH (substitution) Vam…
A: 39) Tertiary alkyl bromides undergo nucleophilic substitution reaction in SN1 reaction mechanism.In…
Q: elect the major product of the reaction:" ONa Br HO (a) (b) (c) (d) (e)
A: The answer to the following question is-
Q: which involve by migration of atom or group from k Which involve interconversion reaction. 6.…
A: 6)The reaction which involve migration of atom or group of atom from one atom to another is called…
Q: 1. Draw the structure of the intermediate products in the following reactions: (C,H),P-CH CH3 AICI,…
A:
Q: Identify the minor elimination product in the reaction below. H,O A а. Ob.
A:
Q: OH ? Me OH ?. HO, OEt ? Br OH HO, SH Me OEt ? OMe OEt O OH CN
A:
Q: Reactions Draw the stractwes of all productf) from the fllowing ctions 1) 2 Br 2)
A: When alkyne react with Br2 it give addition on triple bond. When O2 react with hydrocarbon it is…
Q: What is the product for the reaction below? CH;CO2H - ? HN Select one: O a. O b. Oc.
A: Here we are required to predict the product of reaction between ketone and secondary amine
Q: ank the carbo OlUlI 3. Each of the carbocations below will spontane rezIrangement product.
A:
Q: Give a clear handwritten answer....explain
A:
Q: Complete the reaction by identifying the major product/s of the reaction: CHgS CI CI (r) TV O IV O…
A:
Q: Which compound is the dominant product in the reaction shown below? KMNO4 .COOH (A) H*/H,O (B) COOH…
A: In presence of KMnO4, side chain of benzene is getting oxidised to form carboxylic acid.
Q: Reacti on Scheme:2ndrxn Ha sula (care) + Hzo HO n-butano I acetic acid n-buty acetate
A:
Q: ggest an efficient synthesis for the following product starting from the correspondi rting material…
A:
Q: Which of the following is a PRODUCT-FAVORED reaction? H3O* H20 HSO4 so,2- H2SO3 HSO3 H3PO4 H2PO4 HF…
A: In this question, we will Identify which reaction is Product favored means it will moving forward…
Q: What is(are) the predicted products of the reaction shown? -OCH2CH3 excess KOH LOK LOH LOK HO…
A: ->Ester on hydrolysis in basic medium form carboxylate ion and alcohol.
Q: Draw the products of attached reaction
A: Hydrolysis is a reaction in which the hydration of the compound is done to beak the bond.…
Q: 2. CidssiFy the Fallowing reagerts es either nucleaphiles or e lectraphiles: Zn, CH3MH, HS, OHa, CH3…
A:
Q: B. Draw structures for the products in the following reactions. CN CN CN Jo 70⁰ 1. PrMgBr 2. H₂O* 1.…
A: PrMgBr is the nucleophile here LiAlH4 is reducing agent I2, NaOH,∆=> iodoform reagent.
Q: What is the product of the E1cb reaction below? ОН NaOH ??? A O A O B
A:
Q: What product(s) would be formed in the reaction below? OH H2SO4 ? O a. Ob. c. O d. Two of them O e.…
A:
Q: 8.77 Substitution vs. Elimination: Identify the major and minor product(s) for each of the following…
A:
Q: 19.) Draw all possible products for the reactions below. Show the mechanism to support your an wer…
A:
Q: H202 HQ write the mechanis af each reaction)
A:
Q: aw the major products for the ff. reactions: МСРВА --> cat. OsO4 NMO + HBr –- H20 + Br2 --> 4-
A:
Step by step
Solved in 2 steps with 1 images

- provide the mechasim using the reagents shown ( asap)Explain the following result. Acetic acid (CH3COOH), labeled at its OH oxygen with theuncommon 18O isotope (shown in red), was treated with aqueous base, and then the solution was acidified. Two products having the 18O label at different locations were formed.Explain the following result. Acetic acid (CH3COOH), labeled at its OH oxygen with theuncommon 18O isotope (shown in red), was treated with aqueous base, and then the solution was acidied. Two products having the 18O label at different locations were formed.
- Draw a mechanism of the reduction of 188Re-perrhenate (Re(VII)) to Re(V) by using SnCl2.Identify the organic functional groups and reaction type for the following reaction.The reactant is a(n)a. beta pyranoseb. beta furanosec. alpha pyranosed. alpha furanoseThe product is a(n)a. alpha furanoseb. alpha pyranosec. beta furanosed. beta pyranoseThe reaction type is:a. oxidation (benedict's)b. esterificationc. mutarotationd. reduction (hydrogenation)e. hemiacetal formationTreatment with NaBH 4 converts aldose U into an optically inactive (meso) alditol V. Ruff degradation ofU gives W, whose alditol is optically inactive. Ruff degradation of W forms D-glyceraldehyde, thesimplest aldose. Upon Kiliani-Fischer synthesis, U is converted to two aldoses, X and Y. X is oxidized toan optically active aldaric acid Z. Y is oxidized to an optically inactive aldaric acid. Draw the structuresof D-glyceraldehyde, V, W, X, Y, and Z. Structure of compound U is shown below.
- PLZ GIVEN MECHASIM IN DETAILAn oligosaccharide isolated from an organism is found tocontain two glucose residues and one galactose residue.Exhaustive methylation followed by hydrolysis producedtwo glucoses with methoxy groups at positions 2, 3,and 6 and galactose with methoxy groups at positions2, 3, 4, and 6. What is the structure of the originaloligosaccharide?The attached isomerization reaction, drawn using D-glucose as startingmaterial, occurs with all aldohexoses in the presence of base. Draw astepwise mechanism that illustrates how each compound is formed.
- An aldopentose "O", is oxidized with HNO3 to a diacid "P" which is optically active. The compound "O", is also degraded to an aldotetrose "Q" which undergoes another oxidation to an optically inactive diacid "R". Assuming that "O" has the configuration D-(4R); what are the structures of "O" to "R"?Explain this result: Acetic acid (CH3COOH), labeled at its OH oxygen with the uncommon 18O isotope (shown in red), was treated with aqueousbase, and then the solution was acidified. Two products having the 18Olabel at different locations were formed.Draw the products of attached reaction and indicate stereochemistry aroundstereogenic centers.



