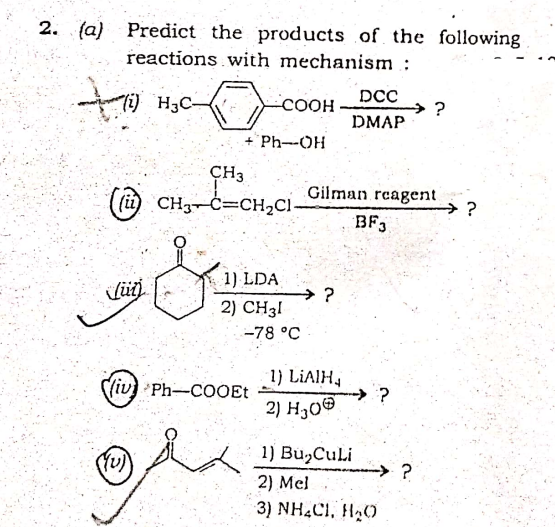
2. (a) Predict the products of the following reactions with mechanism : to DCC Ti) H3C- -COOH →? DMAP + 'Ph-OH CH3 (i CH3¬Ċ=CH2CI- Gilman rcagent →? BF,
Q: The reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO-…
A: In bromination reaction, bromine atom is introduced to a compound. Hydrogenation can be done using…
Q: 3. Provide the major organic product of the following reaction. H₂NNH₂, H (cat.)
A: When ketone react with hydrazine it will form hydrazone as the major organic product.
Q: Consider the following reaction (DPPE structure below): F. - B(OH)2 F Ni(OTf)2 DPPE K2CO3 (a) Give…
A: The name reaction of the given transmetalation is known as Suzuki-Miyaura coupling in which metal…
Q: What products would ypu obtain from reaction of 2,4-dimethyl-2-pentene with BH3, followed by H2O2,…
A: Alkene reactions with BH3 and H2O2 in presence of base are Hydroboration oxidation reaction.
Q: 3. The utilization of diols as intermdiates towards the ultimate oxidative cleavage of alkenes is an…
A: Aldehyde on reaction with Grignard reagent followed by THF to form secondary alcohol.
Q: d. 1. BuLi, THF, -78 C; 2. dil. HCI e. KH Ph PF6 f. Zn H3C. CH3 Br Br
A: Regioselectivity - The position at which the reagent prefers to affect and the reaction happens.…
Q: 4 pts Provide the structure of the major organic product. in the following reaction sequence. 1.…
A: Answer:- This question is answered by using the simple concept of chemical reaction of ketone with…
Q: (d) For the following reaction give the structure of the heterocyclic product formed and give a…
A: This reaction is an example of Heck Coupling.
Q: + NG CusO.SH,O MC
A: Detail mechanism is shown below
Q: a) Provide a mechanism that accounts for the product formed in the following reaction. OH CH +…
A: In both the reactions, carbocation rearrangement takes place through hydride shift
Q: A2. Complete the following by drawing the structure of the main organic product(s) AB-AG for each…
A:
Q: 6. 2-methyl-1,1-diphenylpropene can be prepared by Wittig reaction. a) Suggest any suitable carbonyl…
A:
Q: Explain the following observation. Treatment of alkyl chloride A with NaOCH,CH3 yields only one…
A: The last option i.e. switching from concentrated to dilute base shifts the mechanism from E2 to E1.
Q: 2. (a) Explain the reactivity of dimethyl- sulphoxonium methylide and dimethyl- sulphoxium…
A:
Q: 8. Provide the mechanism for the reaction of problem #7. Draw the skeletal structures of all the…
A: Given, The skeletal structures of the intermediates and final products are:
Q: 12. Based on the reaction scheme below, deduce and name structures A, B and C. H,, Pd A KMNO, В…
A: Ans
Q: Alkene itself in the Heck reaction can be a reducing agent, converting Pd(II) pre-catalyst to Pa0).…
A: HECK REACTION
Q: 1) 0,; CH,CI,; -78°C 2) Zn; ácido acético 1) O,; CH,CI,; -78°C 2) Zn; ácido acético 1) O,: CH,CI;…
A:
Q: -! Starting from ethane, outline a method of synthesis of meso 3,4 dibromohexane. SHOW ALL THE STEPS…
A: At first ethane is treated with one mole Cl2 under photolytic condition to get ethyl chloride. Two…
Q: 1.Provide the identity of Intermediate I and Intermediate Il in the following synthetic sequence,…
A:
Q: ÇOOH CH,OH CH,OH (1) LIAIH, (i) H̟O HNO, / H,SO, `NO,
A: The formation of alcohol from a carboxylic acid is a reduction reaction. It takes place in the…
Q: do both with mechanisn
A:
Q: Suggest a plausible-mechanism-for the synthesis of the compound shown below H30 OH
A: This synthesis is bassed on Dienone-phenol rearrangement followed by reduction of side ring.
Q: 2. Predict the products of the reduction of a. LIAIHĄ with b. NaBH4
A: LiAlH4 is very reactive while NaBH4 is a mild reducing agent. LiAlH4 reduces almost all the commonly…
Q: Give the major organic product(s) or reagents needed. Show stereochemistry Where approp OH K,CrO7,…
A: In organic reaction we can convert one functional group into other carrying out certain reaction…
Q: A. B. C. D. Br Br heterolysis 心。 Br Br Br ① Br
A:
Q: CH; 0 CH3-CH-C-CI HNO, Fe, HCI A AICI3 H2SO, Pd/H2 Br2. FeBr. KMNO, D
A: Friedel-Craft acylation reaction: Benzene gives an aromatic electrophilic substitution reaction. The…
Q: Be the product of the reaction scheme below? TosCI NaNH(CH3) pyridine DMSO Na ONa
A: The question is based on the concept of organic reactions. We have to identify the product.
Q: Reaction of acetic acid, CH3CO2H, with isotopically labeled CH318OH and catalytic sulfuric acid…
A: The reaction of an acid with alcohol is known as the esterification reaction.
Q: a. Complete the following reactions and outline the mechanism for the formation of the product: CH3…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: (a) Provide mechanisms for formation of both A and B, starting from bromobenzene. Please name each…
A: Mechanisms for formation of both A and B is given below
Q: MeO2C -CO2H Pd(OAc)2, IOAC O a) Acetoxylation Ob) Acetylation Oc) lodination Od) Coupling
A:
Q: a. Complete the following reactions and outline the mechanism for the formation of the product: CH3…
A: Hello. Since your question has multiple questions, we will solve the first question for you. If you…
Q: 2-Give the structures, including stereochemistry of compounds A and B in the following sequence of…
A:
Q: When diethyl succinate (1) is treated with NaOEt in EtOH, the major product is diethyl…
A:
Q: 3. What product would you expect to obtain from a nucleophilic substitution reaction of (S)-2-…
A:
Q: 4) Provide the structure of the major organic product of the reactions below. (CH;CH,),N NaOCH CH,…
A: In this question, we want to draw the structure of major product. Reagent and starting material are…
Q: Give two syntheses for (CH3)2CH¬O¬CH2CH3, and explain which synthesis is better
A: The given compound is ether. It can be synthesized by Williamson synthesis. The best yield is…
Q: b) Ozonolysis of an unknown alkene yields the diketone shown in the scheme below. Suggest the…
A: This is the ozonolysis reaction of Alkene.In this reaction ozonide is an intermediate form and PPh3…
Q: 4. Tryptophan can also form adducts with arenediazonium ions, although the reaction is not…
A: Here the chemical reaction is:
Q: (3E , 5Z ,7E)-3,5 ,7 - Decatrine undergoes thermal clectrocyclic ing closure . The proauctls formed…
A: An electrocyclic reaction is a type of pericyclic rearrangement where the net result is one of the…
Q: N CH3 CH3 1. NBS, AIBN 2. NaOH ?
A:
Q: R OH 1. NaH MOTS PCI; Mg/dietil éter, H,O
A:
Q: エ HICIH I-CIH エーU-エ Jーエ H-CIH エ
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: Outline a full mechanism for the solvolysis reaction below. Use curly arrows. Identify the structure…
A: Ethanol is a polar solvent. The polar solvent stabilized the carbonation by dipole-ion interaction.…
Q: TMSCI HO, (CH3CH2)3N
A: Here the Et3N acta as the base which 1st deprotonates the OH and then the alpha H atom with respect…
Q: -s-OH Methanesulfonic acid Isopropyl cyclopentanecarboxylate H20 ,A
A:
Q: 2. (a) Give the product of the following reaction: Br t-BUOK + (b) Using the FMOS of the reacting…
A: The Frontier Molecular Orbital (FMO) approach deals with the HOMO (highest occupied molecular…
Q: 3. Juggest plausible arrew-pushing mechanism for the following reaction. a OH So mot % A Bu n Bu O:…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Carbonic anhydrase catalyzes the hydration of carbon dioxide: H2O + CO2 -> HCO3- + H+The following data is available: (image) - Calculate the Michaelis Menten constants - In the presence of sulfanilamide the Michaelis Menten constants for the reaction are Vmax: 7.57e-5M/s and Km: 20M. What type of compound is sulfanilamide and what is its mechanism of action?When pyrrole is added to a dilute solution of D2SO4 in D2O, 2-deuteriopyrrole is formed. Propose a mecha-nism to account for the formation of this compound.Addition of PPh3 to a solution of Wilkinson’s catalyst, [RhCl(PPh3)3], reduces the turnover frequency for the hydrogenation of propene. Give a plausible mechanistic explanation for this observation.
- please provide the machanisms of 1a, 1e, 1fSuppose we aempt the conversion of fumaric acid to deuterated malic acid with BD3·THF,followed by oxidation with D2O2in NaOD(aq). Show all the possible stereoisomers (as Fisherprojections) that may be formed, and draw the mechanistic pathways (showing stereochemistry)that lead to these possible productsState mechanism- Reduction of a Nitrile with LiAlH4 ?
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estroneOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…Virgin coconut oil contains about 46-48% of lauric acid (CH3(CH2)10COOH). In thepresence acid catalyst and an alcohol, propose the mechanism of this reaction.

