By using "arrow-pushing diagram", draw the mechanism for each of the following reactions and predict the reaction as SyI, Sy2, El or E2. (i) OH conc. H,SO, 120 °C CH3 (ii) Br NaOCH; CH,OH (iii) -CH, CH,CH,OH warm
By using "arrow-pushing diagram", draw the mechanism for each of the following reactions and predict the reaction as SyI, Sy2, El or E2. (i) OH conc. H,SO, 120 °C CH3 (ii) Br NaOCH; CH,OH (iii) -CH, CH,CH,OH warm
Chapter11: Reactions Of Alkyl Halides: Nucleophilic Substitutions And Eliminations
Section11.SE: Something Extra
Problem 27MP
Related questions
Question
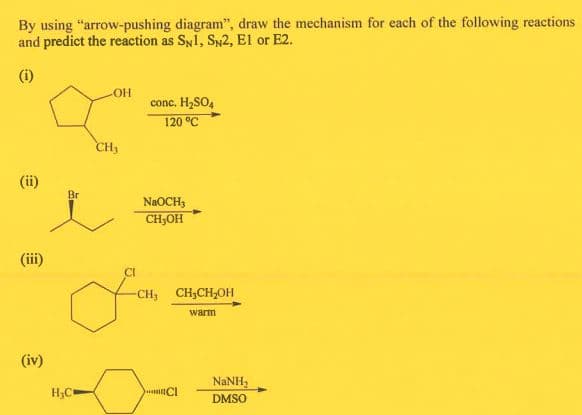
Transcribed Image Text:By using "arrow-pushing diagram", draw the mechanism for each of the following reactions
and predict the reaction as SNI, SN2, El or E2.
(i)
OH
conc. H,SO,
120 °C
CH3
(ii)
Br
NAOCH3
CH,OH
(i)
CI
-CH; CH;CH;OH
warm
(iv)
NaNH,
H;C
DMSO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

