Q: What is the oxidation number of the underlined element in the following compound? a) KMnO4 f)…
A: Oxidation number is defined as the number of electrons which an atom can share, lose or gain while…
Q: redox reaction? O yes O no 2Fe(s) + 3NICI, (aq) 2FECI, (aq) + 3Ni(s) reducing agent: oxidizing…
A: Redox A reaction in which oxidation and reduction takes place simultaneously is called a redox…
Q: Determine the oxidation number for the indicated elementin each of the following compounds: (a) Co…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: 7- The oxidizing agent in the following chemical reaction is +2 5Fe + MnO (aq) + 8H (aq) - Ma + 4H,0…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Identify the type of reaction, and balance it: 6. Qn + Fv,P Fv + On,P a type of reaction
A:
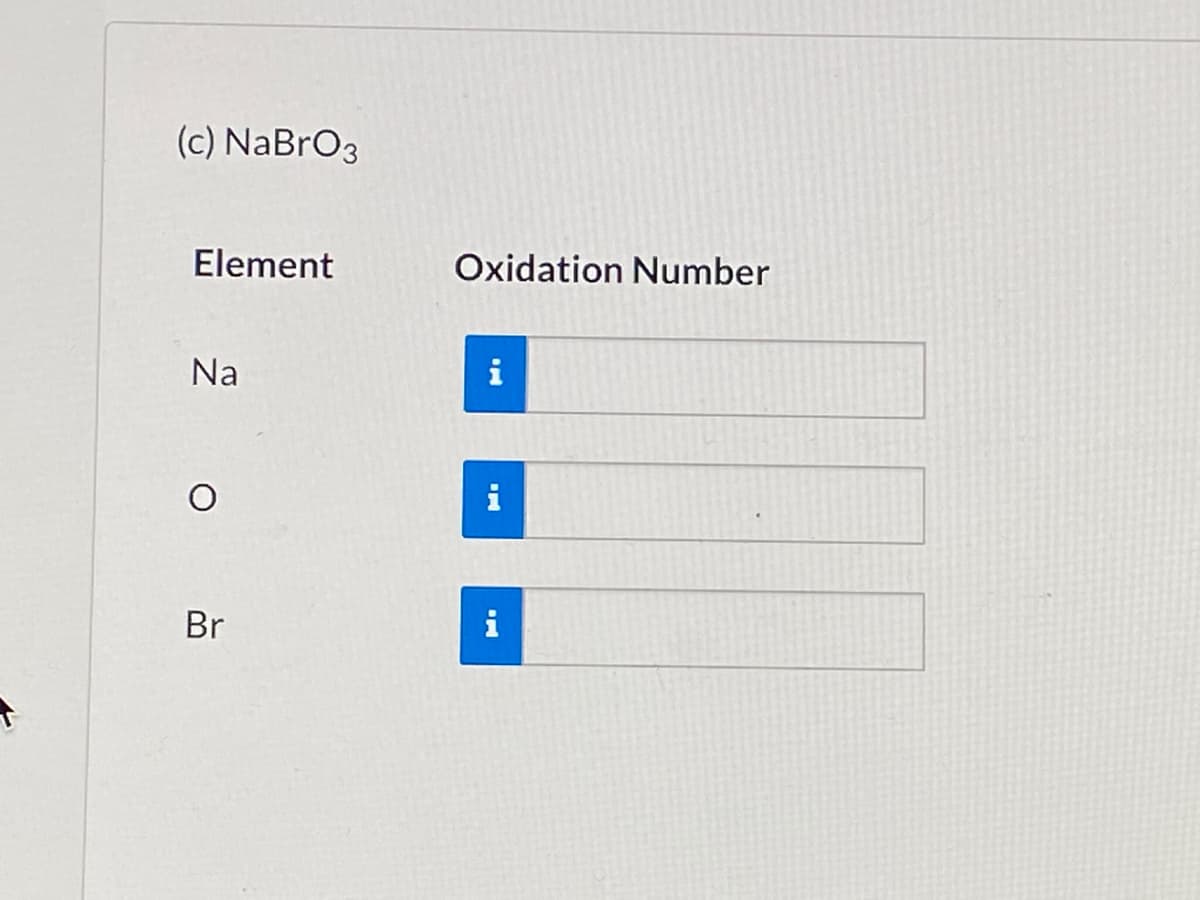
Q: (d) NaBrO4 Element Oxidation Number Na i i Br i
A:
Q: S + Pb+ 2H+Pb2+ + H2S In the above redox reaction, use oxidation numbers to identify the element…
A: Loss of electron oxidation, gain of electron reduction.In the given reaction, oxidation numbers of…
Q: Consider the following redox reaction: Ag⁺(aq) + Fe²⁺(aq) ⇌ Ag(s) + Fe³⁺(aq). How many moles of…
A: In the given reaction we can see that the Ag+ is being reduced to the Ag and Fe2+ is being oxidised…
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A: Oxidation state represent the residual charge on atom in a compound when all other atoms are…
Q: What is the oxidation number of iron, Fe, in Fe(NH4)2SO4?
A:
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A: We have to calculate the oxidation state of highlighted atoms.
Q: What type of reaction is represented by the following equation: C4HB(g) + 602(g)→4CO2(g) + 4H20(g)…
A: The given reaction is, C4H8 (g)+ 6 O2 (g) ----> 4 CO2 (g) + 4 H2O…
Q: oxidation number and balanced redox reaction NH3(g) + NO2(g) → N2(g) + H2O(l)
A: Balancing of a redox reaction using oxidation number method involves conservation of oxidation…
Q: Determine the oxidation states of the elements in the following compounds:(a) NaI(b) GdCl3(c)…
A: Introduction: We need to tell oxidation state of the following compounds . Oxidation state : It is…
Q: 4Cr(OH)3 + 3N₂0+ 60H4Cr + 6NO₂+ 9H₂0 to In the above reaction, the oxidation state of nitrogen…
A: We need to find oxidation state of N in the given reaction and the number of electrons transferred
Q: Identify the oxidizing agent and the reducing agent in the following reaction, and explain your…
A: Identify the oxidizing agent and the reducing agent in the following reaction, and explain your…
Q: Identify the oxidation number of the atom/s 1. NaCl 2. Cu2O 3. CaH2 4. S8 5. H2O2
A: We know that,. For neutral atom, some of oxidation number is equal to zero. If molecule present in…
Q: In the following redox reaction, what are the reducing agent, and the oxidizing agent, respectively?…
A: The reaction given is Cl2 (aq) + 2 Fe2+ (aq) → 2 Cl‾ (aq) + 2 Fe3+ (aq)
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A: Answer:- this question is answered by using the simple concept of calculation of oxidation state…
Q: Cr + 6NO3¯+ 6H*6NO2 + CIO;+3H20 In the above reaction, the oxidation state of chlorine changes from…
A: Find out the oxidation states of chlorine on both the sides Find the difference between the…
Q: 4.43 Arrange the following species in order of increasingoxidation number of the sulfur atom:
A: Suppose that oxidation number of Sulfur in each molecules is "a" Given molecules are part…
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A: The oxidation state of an atom is a number which gives us the number of electrons lost or gained (or…
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A: Write oxidation number of highlighted atom ---
Q: In the following reaction, the oxidation number of O changes from to Fe,O,+ 3CO → 2Fe+ 3CO, a)…
A: The oxidation number of O doesn’t change in fallowing reaction Fe2O3 + 3CO ——> 2Fe + 3CO2…
Q: Write the balanced NET ionic equation for the reaction when AgC₂H₃O₂ and Ni(ClO₄)₂ are mixed in…
A: Balanced molecular reaction is given by,
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A:
Q: Balance these equations for redox reactions occurring in BASIC MEDIUI the reducing agent and the…
A:
Q: 2. + H + Fecla → ar +. H + element oxidized
A: In the redox reaction, Oxidation and reduction takes place simultaneously. In Oxidation loss of…
Q: Classify the following reaction: ZnO (s) + C (s) → Zn (s) + CO (g) O Combustion reaction REDOX…
A: ZnO(s) +C(s) = zn(s) + CO(g) * The reaction in which one substance oxidised and other reduce is…
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A:
Q: 1. Consider the following redox reaction: MnO, + 5 Fe* + 8H* Mn2* + 5 Fe* + 4H,0 Which of the…
A: Oxidation number is the imaginary or fictitious charge developed on an atom if all shared electrons…
Q: (b) Cr2S3 Element Oxidation Number Cr i S i
A: Oxidation number of Cr : +3
Q: sets the oxidation numbers P+5HNO3~H3PO4+5NO2+H2O
A: Oxidation state or number is number of electrons gain or lose by compound/ element. . Total charge…
Q: What is the oxidation number of the underlined element in the following compound? a) K2Cr2O7 b)…
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost…
Q: Identify the oxidation number of the underlined element. MnO4
A: Oxidation number: • Oxidation number of atoms in pure element or Atoms in free or uncombined…
Q: For the following conversion, mark the correct agent as Oxidising Agent(OA) or Reducing Agent(RA).…
A: If the chemical species undergoes chemical reaction by losing of electron, then the species itself…
Q: 4Bi + 6NO,+ 9H,0→3N20 + 4Bi(OH)3+ 60H¯ In the above reaction, the oxidation state of bismuth changes…
A: The oxidation number or oxidation state refers to the number of electrons gain or loose by an…
Q: Some chemical reactants are listed in the table below. Complete the table by filling in the…
A: a) FeO: Since there are total 6 valence electrons in O. Hence O makes ion O2- to complete its octet.…
Q: (i) 5AI + 3V +5> 5AI +3 + 3V (ii) 2Ra + 02→ 2RAO
A: Mass number is nothing but the sum of protons and neutrons in an atomic nucleus and is denoted by…
Q: (a) Sr(BrO3)2 Element Oxidation Number Sr i Br i i
A:
Q: redox reaction? O yes O no 2N, (3) + 50,(e) → 2N,0,(2) reducing agent: oxidizing agent: redox…
A:
Q: e) OO0
A: Reaction of acid with base gives water and salt. Reaction is called as neutralization reaction. In…
Q: From the following reaction: MnO2 + 4H* + 2Cl → Mn2+ + Cl, + 2 H20 (a) Which atom is reducing? fill…
A: Explanation to the correct answer is given below
Q: is 2Na(s)+Cl2=2NaCl(s) a redox reaction?
A: In redox reaction , oxidation and reduction occurs simultaneously. The substance which undergoes…
Q: Give the oxidation number of an atom in the compound a. Mn in Mn04- b. Cr in CrO2 2+ c. Fe in Fe304…
A:
Q: In the following chemical reaction, which species is the reducing agent? 2 10, (aq) + 12 H"(aq) + 10…
A: Oxidation is defined as the process in which there is increase in the oxidation state while in…
Q: The proper assignment of oxidation numbers to the elements in the polyatomic ion SO32- would be…
A: In sulfite ion SO32-, the oxidation state of the oxygen atom is -2. Oxidation number of element in…
Q: (b) NaBrO2 Element Oxidation Number Na i Br i
A: Oxidation number can be determined by the electron acceptance or electron loosing character of an…
Q: The reaction Na3PO4(aq) + 3 AgNO3(aq) –Ag3PO4(s) + 3 NaNO3(aq) is best classified as a(n) A)…
A: Ans
Step by step
Solved in 2 steps

- I only need: Classification: ( SFA, MUFA, PUFA) Classification: ( ESSENTIAL OR NON ESSENTIAL)) The potency of antagonists is frequently defined in terms of either their IC50, ortheir Ki. Explain what these two terms mean.Part C (solvents labeled C): How does the number of hydrogen bonding sites within a molecule affect the relative strength of IMF’s?
- An increasing percentage of amphetamines available onthe illicit-drug market originate from ___________ druglaboratoriesThe potency of agonists is frequently defined in terms of their affinity and theirefficacy. Explain what these two terms mean.Draw the products that form when the thioester CH3CH2CH2COSCoA is hydrolyzed with water. Differentiate products by greater or lesser molecular weight.

