Q: Nitration of benzene is carried out by which of the following reactive ?species NO3 O HNO3 O "NO2 O…
A:
Q: H3C-O H3C-0-Li 2. HBr OH2 Br + a = Proton transfer e = Electrophilic addition h = SN1 Nucleophilic…
A: For the reaction mechanism. Mechanism:
Q: 1. H20 H;C- H3C CH, aqueous H,SO, он 2. -CH3 `CH3 h = SN1 Nucleophilic substitution i= Sy2…
A:
Q: Propose mechanism consistent with the following reactions a and b. HBr HBr ? b) регоxdes
A:
Q: What is the major product in the following series of reactions? a b C D d e 1) H2SO4 (dilute) 2)…
A: In presence of diluted H2SO4 and H2Cr2O7 Ketone is formed. Then phMgBr carry out SN2 reaction.…
Q: 1. AICI, + HCI 2. H2SO4 он H20 130° a = Proton transfer e = Electrophilic addition h = Sy1…
A: Organic reactions are classified mainly as addition (if a reagent gets added to the carbon carbon…
Q: Aromatics can be converted into nitroaromatics upon treatment with a mixture of nitric and sulfuric…
A:
Q: 4.33 Predict the products of the following reactions. Indicate regioselectivity where relevant. (The…
A:
Q: Arrange the following in increasing reactivity towards nucleophilic addition. 0₂N- Select one: O a.…
A: The carbonyl compounds give nucleophilic addition reactions. The carbon atom of the carbonyl carbon…
Q: When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40%…
A: The structure of buta-1,3-diene is shown below. The two compounds formed when Br2 is added to…
Q: Which of the following reagents is CORRECT for the reaction below. HO O KMNO4 O H2CrO4 O…
A:
Q: MC02 How many of the compounds shown are not expected to be converted into Grignard reagent on…
A: How many of the compounds shown are not expected to be converted into Grignard reagent on reaction…
Q: Predict the major product for the following reaction. Na,Cr207 H2SO4, H2O ?
A:
Q: a. Propose a mechanism for the following reaction. (Hint: An electrocyclic reaction is followed by a…
A: a) In the given reaction the reactant was treated with the ethylene molecule under thermal…
Q: Synthesize the given compound using both Suzuki and Heck coupling reaction: BI U TE 10 11 77…
A: Question belong to organometallic compound that is cross coupling reaction mechanism have mentioned…
Q: 22. Which of the following WILL NOT act as a diene reactant in a Diels-Alder reaction? B) A) A) C)…
A:
Q: Q4. Discuss the reagents, conditions, and mechanism of the following reactions. a) Kolbe Reaction b)…
A:
Q: a) Propose an efficient synthesis for the following reaction: b) Write the mechanism and predict the…
A: The conversion of a reactant to a desired product requires the chemical reaction of that reactant…
Q: Give the structure of compounds A to F in the following series of reactions: CH; 0 CH3-CH-C-CI HNO3…
A: Acyl group is meta directing. Amine group is ortho/para directing.
Q: . Terminal alkynes react with Br2 and water to yield bromo ketones. For example: `CH;Br Propose a…
A:
Q: Which of the following compounds is the most reactive towards electrophilic aromatic subsitition by…
A:
Q: Rank the following nucleophiles correctly arranged with respect to increasing nucleophilicity in SN2…
A: (B) A < D < C < B Negatively charged species is stronger nucleophile as compared to neutral…
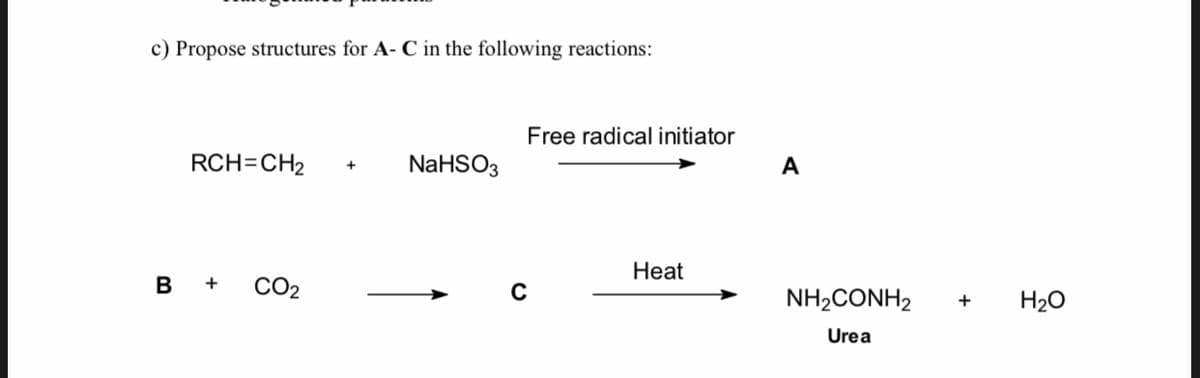
Q: Give the structures of products or reagents: A-C. N° heating A N' Br (1) Pd(PPH3)2Ch, THE (2) H3O*…
A:
Q: Propose a mechanism to account for the following finding: Use scratch paper. O18 || С. 18 ОН 18 HCI,…
A: The reaction between carboxylic acid and water in the presence of acid leads to a change in the…
Q: Explain, please 3.) Predict the products of the following. (17.41) (a) 1. LIAIH, 2. D20 ct (b) 1.…
A:
Q: What is the expected major product for the reaction below? EGMU 2020737654 t-BUOK Br ? t-BUOH 4.…
A: This reaction is an elimination reaction . H and Br should be anti to each other. As it is a anti…
Q: You must prepare the most stable possible alkene starting from the starting material given below and…
A: Here the given reactant. We have to determine the major product from the following reactant…
Q: Question is attached as a picture. Thank you!
A: Diels-Alder reaction:During this reaction, three pi bonds will get broken; two sigma bonds and one…
Q: Which of the structures below (A - D) is the major product (Y) of the following reaction scheme? 1.…
A:
Q: Suggest a mechanism and predict the major product for th 1) LIAIH, 2) H0
A: When ketones reduced by strong Oxidising agent like lithium aluminum hydride (LiAlH4) then finally…
Q: Give the Reagents. O KMN04 O 1) LIAIH4, 2) H3O+ O periodinane O 1) MgCI, 2) H3O+
A: Oxidation is defined as the removal of electrons, addition of oxygen and removal of hydrogen.…
Q: 5. Predict the products of the following reactions: basi loriosi 16W 39129 to ab a. b. C. d. 910…
A: All the reactions are Nucleophilic substitution reactions. Here in all the given reactions…
Q: 2. Propose mechanisms for the following reactions: a. Cl-substitution in [(n²-C2H4)PTCI] by Br in…
A:
Q: The following are several isomers of C10H14 with two fused six-membered rings. A B D E G H (a)…
A: Ans(a): The Diels-Alder reaction is a pericyclic reaction in which a diene reaction with enophiles.…
Q: (b) Propose how you could synthesize following compounds from suitable alkene. OH (ii) (ii)…
A: Organic reaction mechanisms:
Q: Which bromide below is most likely to lose Br and form an aromatic carbocation where the positive…
A: After releasing Br- it’ll generate a carbocation and aromatic or 4n+2π systems are highly stable…
Q: [ 18] A Hydrogen attached to which of the following carbons would be most acidic ? (a) (c) (b) → (d)…
A: Acidic strength depand stability of conjugate Anion
Q: 5) Provide the products necessary to complete the synthesis below. Indicate what mechanism was used…
A: The above organic transformation is given below
Q: Identify the expected major product of the following reaction. Na,Cr207/H2SO4/H2O ? excess но HO HO,…
A: 1- In this question, we will identify the major product. You can see details explanation and final…
Q: 1. HCI CO2CH3 AIBN H2 OCH3 70° CH3 poly(methyl methacrylate) (Plexiglass, Lucite) 2. a = Proton…
A:
Q: In Nicolaou's total synthesis of taxol (Ref.: J. Am. Chem. Soc. 1995, 117, 653) the carbonyl group…
A: This is an example of conversion of ketone to vinyl Lithium via tosylhydrazone intermediate.
Q: Draw the starting materials you could use to synthesize the following organic molecules, using only…
A: Heck Reaction is the Pd catalyzed cross coupling reaction between activated alkenes and aromatic…
Q: Identify (A) in the following reaction. 2H2 Pt (A) KMNO4 Warm conc. 0 || С — С—о—н |CO,H + HO | CO2H…
A: Correct answer is option (b)
Q: 4.21 Name the following alkenes, and predict the products of their reaction with (i) KMNO4 in…
A: Here, we have to write the names of the given alkenes. Also, we have to predict the products when…
Q: Identify the main expected product when the epoxide below reacts with PhMgCI, followed by acid…
A: 1. Epoxide is opened with Grignard at less substituted side (Sn2 reaction). 2. 6 pi electrons are…
Q: Which is the first step of the reaction below? protonation of benzene NO2 HNO3 protonation of nitric…
A: Note - Since you have asked multiple questions, we are bound to solve only the first question for…
Q: In Nicolaou's total synthesis of taxol (Ref.: J. Am. Chem. Soc. 1995, 117, 653) the carbonyl group…
A: In this question, we will discuss about the mechanism and then give the name of the Reaction. You…
Q: T A TOpice TReferences) OH 1. H,PO, H20 MeO 80 Meo 2. HI a - Proton transfer g- SyI Nucleophilic…
A: 1) This reaction is basically elimination reaction and it is going in acidic medium. 2) This…
Step by step
Solved in 2 steps with 1 images

- Aromatic iodination can be carried out with a number of reagents, including iodine monochloride, ICl. What is the direction of polarization of ICl? Propose a mechanism for the iodination of an aromatic ring with ICl.The polyurethane foam used for home insulation uses methane-diphenyldiisocyanate (MDI) as monomer. The MDI is prepared by acid-catalyzed reaction of aniline with formaldehyde, followed by treatment with phosgene, COCl2. Propose mechanisms for both steps.Identify the pericyclic reaction benzocyclobutane E undergoes on heating, give astructure for intermediate H, and provide a mechanism that accounts for itsformation.
- What is the expected product of the reaction scheme below?Following is a balanced equation for bromination of toluene.(a) Using the values for bond dissociation enthalpies given in Appendix 3,calculate ∆H0for this reaction.(b) Propose a pair of chain propagation steps and show that they add up to theobserved reaction.(c) Calculate ∆H0for each chain propagation step.(d) Which propagation step is rate-determininExplain the mechanism-Carboxylation—Reaction of RMgX with CO2
- Acetoxybenzene (PhOC(=O)OCH3) is much less reactive than ethoxybenzene (PhOCH2CH3) in electrophilic aromatic substitution reactions. Suggest an explanation for this result, based on an analysis of the inductive and resonance electronic effects of the two substituents on the stability of theWheland intermediate for para substitution by an electrophile E+Unknown X, C5H9Br, does not react with bromine or with dilute KMnO4. Upon treatment with potassium tert-butoxide,X gives only one product, Y, C5H8. Unlike X, Y decolorizes bromine and changes KMnO4 from purple to brown.Catalytic hydrogenation of Y gives methylcyclobutane. Ozonolysis–reduction of Y gives dialdehyde Z, C5H8O2. Proposeconsistent structures for X, Y, and Z. Is there any aspect of the structure of X that is still unknown?Tributyltin hydride (Bu3SnH) is used synthetically to reduce alkyl halides, replacing a halogen atom with hydrogen. Free-radical initiators promote this reaction, and free-radical inhibitors are known to slow or stop it. Your job is todevelop a mechanism,
- The following compound undergoes Benzilic Acid Rearrangementto yield ahydroxyacid salt. Proposea mechanism for the reaction, writethe major product,and provide an explanation as tothe preference of migration of one R group over the other.When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.) Propose a mechanism to account for formation of both A and BCompound A gives the product(s) below on oxidative cleavage with KMnO4 in acidic solution. Propose a structure for A.

