Q: Add a lone pair to NH3. Add a formal charge, if applicable. Select Draw Templates More / || | N H…
A:
Q: What is the major product of the following reaction? NO₂ O₂N- O₂N- OCH3 CI O Multiple Choice O O…
A:
Q: Organic Chemistry HW: 2,6-dimethyloct-2-ene cannot be handwritten, explanation of print the…
A: 2,6-dimethyloct-2-ene We need to write the structural formula of above molecule (expanded or…
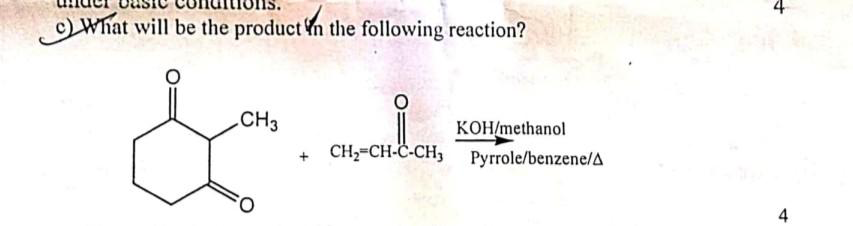
Q: 9. Provide the structure of the product in the following reaction. НО OH
A: The given reaction is a type of protection reaction where protection of ketone occur with the use of…
Q: actions with respect to the amino acid sequence) between residues on a single polypeptide chain…
A:
Q: The lattice energy of MgO is 3890 kJ/mol. The first and the second ionization energies (IE, and IE₂)…
A: The reaction involved is Mgs+12O2g→MgOs The values provided are: Lattice enthalpy of MgO = +3890…
Q: How does the coordination geometry of a metal center influence its reactivity in bioinorganic…
A: Introduction: The coordination geometry of a metal center has a profound impact on its reactivity in…
Q: Suppose the galvanic cell sketched below is powered by the following reaction: Fe(S)+2 AgNO3(aq) →…
A: A question based on electrode potential. An electrochemical cell is represented using that the…
Q: A chemist designs a galvanic cell that uses these two half-reactions: standard reduction…
A: Note: Since you have posted a question with multiple sub-parts, we will solve the first three…
Q: Complete the mass balance expression for a saturated solution of Ag₂CO3. Ignore any subsequent…
A: We have to write the Mass balance equation for a saturated solution of Ag2CO3
Q: Calculate the solubility at 25 °C of AgBr in pure water and in 0.12 M KCN. You'll probably find some…
A: From Aleks data resource the Ksp of AgBr is 5.35 x 10-13 AgBr ↔Ag++Br-At…
Q: Using group theory and symmetry selection rules, predict how many signals corresponding to M-Cl…
A: To find out the number of signal corresponding to M-Cl stretches in the infrared and raman spectra…
Q: Which of the following will favor the E-stereoisomer in an elimination? QTS CH3 Br TSO H KOH MeOH…
A:
Q: Consider the following hypothetical scenario: A chemist is attempting to synthesize a complex…
A: Introduction:In complex organic synthesis, unexpected byproducts can pose challenges to chemists…
Q: What quantity of energy does it take to convert 0.700 kg ice at -20.°C to steam at 250.°C? Specific…
A: Following steps will happen in converting 0.700 kg ice at –20 °C to steam at 250 °C 1) heating of…
Q: If 8mL of 60% aqueous sodium hydroxide was used in an experiment, what would be the molecular weight…
A: In the above question given that Volume of NaOH used = 8ml 60% NaOH was used in the experiment.…
Q: Which is not a resonance structure formed when the lone pair of electrons on the N gets delocalized…
A: In N, N-dimethylaniline, the lone pair on nitrogen undergo delocalization with benzene ring.
Q: Excess sodium is reacted with 1.00 L of chlorine gas that has a density of 1.88 g/L to form sodium…
A:
Q: Zinc hydroxide is sparingly soluble. Consider the cell below: 2+ Zn(s) | Zn(OH)2(s) | OH¯ (0.15M)||…
A: Answer:- This question is answered by using the simple concept of electrochemistry which involves…
Q: Draw the Lewis structure of CO. Include lone pairs and formal charges. Select Draw Templates More
A:
Q: What is the binding energy in kJ/mol nucleons for silver-109? 47 H+62 n ¹9 Ag The required masses…
A:
Q: Which molecule is the most reactive in an SN2 reaction? A) CH3CH2CH2CI B) CH3CH2CH2OH C)…
A: For the comparison of the rate of nucleophilic substitution reaction first of all we determine the…
Q: A 150. g sample of KCl is dissolved in enough water to make 1.00 liter of solution. How many…
A: Mass of KCl = 150 g Molar mass of KCl = 74.55 g/mol Moles of KCl = mass / molar mass…
Q: Determine the bond strength of A-A. The bond strength of C-C is 143 kJ/mol The bond strength of A-C…
A:
Q: For the following reaction, 25.4 grams of diphosphorus pentoxide are allowed to react with 13.6…
A: diphosphorus pentoxide s + water l → phosphoric acid aq Mass of diphosphorus pentoxide P2O5 = 25.4 g…
Q: 1) The solubility of MgF, in water at 25 °C is 0.13 g/L. Calculate the K, of MgF,
A:
Q: 7. The following observations were made during a chemical reaction involving three unknown metals.…
A:
Q: Derive the electron configuration and draw the orbital notation for sulfur, 16 electrons.
A: Electronic configuration for sulfur = ? Orbital notation for sulfur = ?
Q: Complete and balance the chemical equation
A: Balanced chemical equation can be define as the reaction in which number of atoms of all the…
Q: Erwin Schrödinger Orbitals which result from solving the Schrödinger wave equation can be…
A:
Q: Calculate the solubility of N₂ in water when the partial pressure of nitrogen above water is 1.16…
A: Henry Law the amount of gas that is dissolved in a liquid is directly proportional to the partial…
Q: Suppose the galvanic cell sketched below is powered by the following reaction: Fe(S)+2 AgNO3(aq) →…
A: A galvanic cell is composed of two half cells: cathode half cells and anode half cells. In such type…
Q: mL of 0.02 M NaOH added pH Reading 0 2.1 1 2.1 2 2.1 2.1 2.3 2.3 2.4 2.5 3 4 5 6 7 8 9 10 11 12 13…
A: The question is based on the titrations. we are titrating a strong acid with a strong base. we need…
Q: Explain the trends in boiling points shown in the graph below. Ensure that you discuss the…
A: Alkanes are the hydrocarbons, that is, compound formed by the carbon and hydrogens. When these…
Q: Describe the mechanism shown for a double zinc aminopeptidase. The image shows the catalysis pathway…
A: The enzyme is question is a double Zn amino peptidase. As its name suggests, it is an enzyme that…
Q: Which of the following reactions are consistent with the reaction profile shown below? Potential…
A: The diagram that shows the potential energy changes during the chemical reaction is considered as…
Q: If a student decreased the temperature for this reaction: C₂H4(g) + H₂(g) - heat C₂H6(g) what would…
A: This principle is used to predict the effect of a change in conditions on a chemical equilibrium.…
Q: A sample of zirconium experiences a temperature change of +790.5 K while absorbing 150820 joules of…
A: Temperature change (∆T) = + 790.5 K Heat absorbs (q) = + 150820 J (positive…
Q: How many equivalents of base need to be added to fully titrate all acidic protons in…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: you took 40.7 grams of KCl and dissolved it in 0.375 L of water, what would be the molar…
A:
Q: If the [COCI412 anion has a molar susceptibility of 6.26 X 10-3 (the corrected value (XM')) at 25…
A: Effective magnetic moment and molar susceptibility are related as μeff = 2.84χm.T B.Mμeff=…
Q: An equilibrium mixture contains 0.350 mol of each of the products (carbon dioxide and hydrogen gas)…
A:
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free…
A: Given,The cell reaction:N2H4(aq) + 4 OH-(aq) 4 Cu2+(aq) → N2(g) + 4H2O(l) + 4 Cu+(aq)From the ALEKS…
Q: How does the molecular structure of a compound affect its chemical reactivity and reaction rate?
A: The molecular structure of a compound is a key determinant of its chemical reactivity and reaction…
Q: A A certain liquid X has a normal freezing point of -5.60 °C and a freezing point depression…
A: This is colligative property, Depression in freezing point. Colligative properties depends on no.…
Q: Draw the curved arrows and the resulting resonance structure for the following structure. Include…
A: The given structure is shown below We have to draw the curved arrow resonance structure of the…
Q: 9. For the information NHƯg) H₂(g) -2H(g) N₂(g) 2N(g) AH" -1172.8 kJ AH-435.930 kJ AH-945.408 kJ the…
A:
Q: Consider the generic Lewis dot symbol for an element. Which element could this symbol represent? N C…
A: In the Lewis dot structure of element, the dots represent the valence electrons in the element.
Q: Determine the polarity of the following compounds.
A: Polar compound: A polar compound is usually formed when one end of the molecule possess positive…
Q: 5. Name the following compounds using IUPAC nomenclature a) b) sa CI
A: The following steps are required for IUPAC naming- First of all identity the longest carbon chain…
Step by step
Solved in 3 steps with 2 images

- 2) What is the main product for each of the following reactions? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (h) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ?What alkene is needed to synthesize each 1,2-diol using [1] OsO4 followed by NaHSO3 in H2O; or [2] CH3CO3H followed by −OH in H2O?Which structure will not yield cis- or trans- isomers after a reaction with H2/Lindlar catalyst or Na/NH3?
- Which of the following is the product of the reaction shown in Image 17? a. (C6H5)3COH b.C6H5CH2OH c.(C6H5)3CH d.(C6H5)2CHOHWhat is the major product formed when 3-methyl-1-butene is treated with Hg(C2H3O2)2 in a THF/ethanol mixture followed by NaBH4 in base?2. How many substitution product/s is/are formed when metabromo anisole is treated with ammonia?A. 0-no reactionB. 1C. 2D. 3

