Q: ?nucleophiles اختر واحدة أو أكثر a. CH4 Ob. Br c. H20 d. NH3 e. H2C=CH2 (-)f. OH (+)g. Br h. BH3
A: The electron rich species are termed as nucleophiles. Nucleophiles can be neutral or negatively…
Q: B. CIRCLE (CROSS-OUT) the FASTEST (SLOWEST) Nucleophilic Substitution Br + H3C-O + H3C-O .CI
A: Rate of Nucleophilic substitution depends on the nature of leaving group. Better the leaving group…
Q: Draw the product of the attached reaction sequence, includingstereochemistry.
A: The given question is based on the prior knowledge of the hydroboration reaction. Hydroboration…
Q: Determine the mechanism of nucleophilic substitution for attached reaction anddraw the products,…
A: INTRODUCTION: SN1 reaction is a two step reaction in which first carbocation is formed that acts as…
Q: From each pair, select the stronger nucleophile. Q.) CH3OCH3 or CH3SCH3
A:
Q: H2N' ONH3
A:
Q: Rank the anions in order of increasing nucleophilicity in acetone: CH3S−, CH3NH−, I−, Br−, and…
A: Among CH3NH-, CH3S-, CH3O- , I- and Br- acetone.
Q: How to Form an Internal Alkyne by Two Sequential SN2 Reactions ?
A: The terminal alkynes are those alkynes in which the triple bond is present between the first and the…
Q: Name the PRODUCT compound and give it's molecular weight Q. Which is a better nucleophile methoxide…
A: Nucleophile takes a negative charge or lone pair of electrons on itself and attacks on a positively…
Q: Rank the following in terms of reactivity towards a nucleophile (1 = least reactive and 4 = most…
A: Electrophiles are reagents that are electron deficient. These attack reagents that are rich in…
Q: Rank the nucleophiles in following group in order of increasing nucleophilicity. H2O, −OH, CH3CO2-
A: Water is a weak nucleophile because it is neutral. Since acetic acid is more acidic than water, the…
Q: Draw a stepwise mechanism for the following reaction. (Hint: Conjugate addition can occur with…
A: Given reaction is,
Q: Which nucleophile gives the highest overall reaction rate in SN2 reactions? Select one: OF o all…
A: Given F- All give the same rate Br- I- Cl- Nucleophile gives higher rate of SN2 = To be…
Q: S. Classify the following reagents as either nucleophiles or electrophiles: Znt, CH,NH2, HS, OH, ,…
A: Since we answer only one question. So, we'll answer the first question. Please resubmit the question…
Q: Q3. Rank the compounds in each of the following groups according to their indicated property )…
A: An electrophilic aromatic substitution is a process where an atom or aromatic ring get replaced by…
Q: Complete the curved arrow mechanism for this reaction including the charges where necessary CO,Me -…
A:
Q: Which of the following correctly represents a nucleophilic attack? :o: :CN ö: : :CN ..O :
A: Nucleophile is reactive species that contains negative charge and attacks on electron deficient…
Q: -Cf3 Nucleophilic Attack
A:
Q: The reaction will proceed with mechanism because the secondary alkyl halide react with nucleophile.…
A: The given 2-bromobutane is a secondary alkylhalide as bromine is attached to secondary carbon atom.…
Q: 5. Tropone (A) is much stronger base (conjugate acid more stabilized) than the open chain,…
A: Comparison of basic nature of A and B.
Q: :ÇI: CH3 + KOTBU H,C ČH3
A: Here, strong base/nucleophile is present, so there is a possibility of substitution as well as…
Q: In a nucleophilic substitution reaction, complete inversion of atom is observed. Would the mechanism…
A: In substitution reaction in which one nucleophile is substituted with another nucleophile is called…
Q: CH, CH, THF/H;0 -: + Hc--CH, CH, + CH, CH, CH Use curved arrows to write the first step of this…
A: Nucleophilic substitution reactions can be defined as the chemical reaction which involves the…
Q: Consider the nucleophilic substitution reaction shown here. Based on the stereochemistry, does it…
A: Nucleophilic substitution reaction is the kind of reaction where electron-rich nucleophile attacks…
Q: Rate the below nucleophiles in terms of INCREASING nucleophilicity. Explain why. CH3OH CH;0 t-BuO-…
A: As the name suggests, nucleophiles are those reagents that possess attraction for the positively…
Q: 4) Draw all mechanistic steps (initiation, propagation, termination) for the following…
A:
Q: 4. Provide the reagent(s) needed to complete the following reaction scheme. Provide the complete…
A:
Q: Draw the products of attached SN2 reaction and indicate the stereochemistrywhere appropriate.
A: (1R,3R)-1-bromo-3-methylcyclopentane undergoes SN2 with cyanide ion to form (1S,3R)-3…
Q: Explain why quinuclidine is a much more reactive nucleophile thantriethylamine, even though both…
A: The chemical species that donates a pair of electron to form a chemical bond is called a…
Q: Consider the following groups of compounds. In each series circke the weaker (poorer) nucleophile.…
A: The nucleophilicity of a substrate depends upon many factors such as basicity, nature of…
Q: 3/5 5.1 each indicated reaction. Mechanisms. Using curved arrows, suggest a reasonable mechanism for…
A: The reaction of alkene with phosphoric acid leads to the formation of carbocation. The oxygen of -…
Q: Draw the products of attached SN2 reaction and indicate the stereochemistrywhere appropriate.
A: Introduction: SN2 reaction: SN2 reaction means substitution nucleophilic bimolecular reaction. In…
Q: Rank the species below in order of increasing nucleophilicity in protic solvent.
A: Nucleophile are species which have excess of electron density either have filled orbital or lone…
Q: "ropose a mechanism for the following reaction. но H,SO, + H,0
A:
Q: From each pair, select the stronger nucleophile. Q.) CH3COO- or OH-
A: The species, that make a covalent bond by donates a pair of electrons is known as nucleophile.…
Q: но. CH3 NaOH aq.
A:
Q: 1. reactivity towards nucleophilic acyl substitution CI HO, RANKING:
A: Nucleophile are those species which attacks on electron deficient species. In carboxylic acid and…
Q: H;C CH3 HN- -c-CH, A в a) Which of the above molecules (A or B) have a higher rate of reaction…
A: We have two compounds, we have to predict the rate of electrophilic aromatic substitution of the…
Q: Complete the following nucleophilic substitution reactions. In each reaction, show all electron…
A: In a chemical reaction; the substance which involves in conversion is said to be reactant whereas…
Q: Draw the products of attached SN1 reaction and indicate the stereochemistry when necessary.
A: The compound (S)-3-Bromo-3-methylheptane undergoes SN1 reaction with ethanol to form a racemic…
Q: 3) Write a detailed mechanism of: CH CH. HNOT H'SO NO
A:
Q: Rank the nucleophiles in following group in order of increasing nucleophilicity. −OH, Br−, F− (polar…
A: The given groups are -OH, Br-, F- To find: The increasing order of nucleophilicity
Q: Arrange in decreasing order according to their reactivity towards an electrophile. H A В
A:
Q: 5. Tropone (A) is much stronger base (conjugate acid more stabilized) than the open chain,…
A:
Q: Rank the nucleophiles in following group in order of increasing nucleophilicity. −OH, −NH2, H2O
A: The word 'nucleophile' comes from two words- nucleus and 'philos' means love. Nucleophiles are…
Q: Draw the products of attached SN1 reaction and indicate the stereochemistrywhen necessary.
A: The products of the attached SN1 reaction and indicate the stereochemistry when necessary has to be…
Q: Which of the following statements about nucleophilic reactions are generally true ? V SN2 reactions…
A:
Q: Draw the curved arrows and the product for each of the following nucleophilic addition steps. (a)…
A: Hi. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If you…
Q: Rank the nucleophiles in each group in order of increasing nucleophilicity.a. -OH, -NH2, H2Ob. -OH,…
A: Nucleophilicity increases with increase in basicity of species. Across a period from right to left…
rank the following
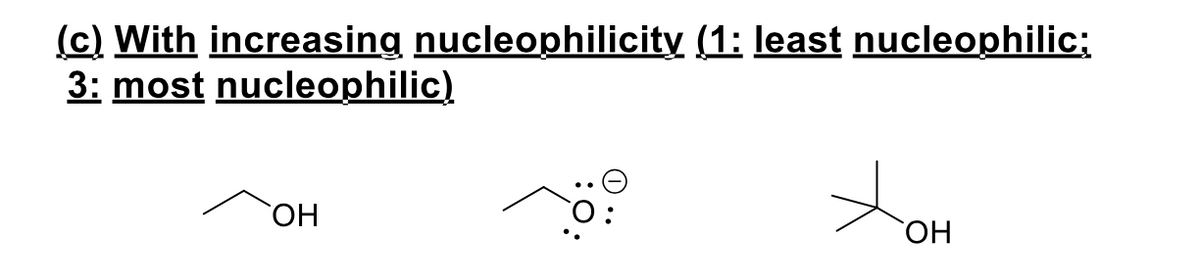
Step by step
Solved in 3 steps with 4 images

- I labeled one of the atoms incorrectly, but I am not sure which. Would the H in HBr be considered a nucleophile as well ?I drew out the molecule to identify any nucleophilic carbon atoms, but they all look to be neutral so I dont understand if there are any.From each pair, select the stronger nucleophile. Q.) CH3COO- or OH-
- In the box to the left of each reaction below, write the mechanism by which it occurs (could be SN1, SN2, or E1, or even 2 of them). Then draw the product(s).Complete the reactions given below, write down the type of mechanism (SN1, SN2, E1, E2)?Give 3 examples of a reaction mechanism of E1 that follows Zaitsev's rule.
- How do I get to my target molecule (1st image) with the following uncomplete reaction (2nd image)Name the PRODUCT compound and give it's molecular weight Q. Which is a better nucleophile methoxide or methanol? Suggest a reason why?3) Explain how and why the reaction below results in the observed regiochemistryand/or stereochemistry.

