c. Is the predicted trend for alkyl chlorides the same as or different than the predicted trend for alkyl bromides? Briefly elaborate. d. The type of alkyl bromide is the same in tubes 4 and 7 but one is acyclic and the other cyclic. Should there be any difference in the predicted rate of SN2 reaction of these alkyl bromides? Briefly elaborate. 2a. Describe the predicted trend between the rate at which the SN2 reaction occurs and the leaving group for each of the following entries: 1 and 2
c. Is the predicted trend for alkyl chlorides the same as or different than the predicted trend for alkyl bromides? Briefly elaborate. d. The type of alkyl bromide is the same in tubes 4 and 7 but one is acyclic and the other cyclic. Should there be any difference in the predicted rate of SN2 reaction of these alkyl bromides? Briefly elaborate. 2a. Describe the predicted trend between the rate at which the SN2 reaction occurs and the leaving group for each of the following entries: 1 and 2
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter19: Carboxylic Anhydrides, Esters, And Amides
Section: Chapter Questions
Problem 19.6P
Related questions
Question
c. Is the predicted trend for alkyl chlorides the same as or different than the predicted trend for alkyl bromides? Briefly elaborate.
d. The type of alkyl bromide is the same in tubes 4 and 7 but one is acyclic and the other cyclic. Should there be any difference in the predicted
2a. Describe the predicted trend between the rate at which the SN2 reaction occurs and the leaving group for each of the
following entries:
1 and 2
3 and 4
5 and 6
b. What general conclusion can be made concerning the rate at which the SN2 reaction occurs and the leaving group?
3. Alkyl iodides were not used in this part of the experiment. Why? Explain.
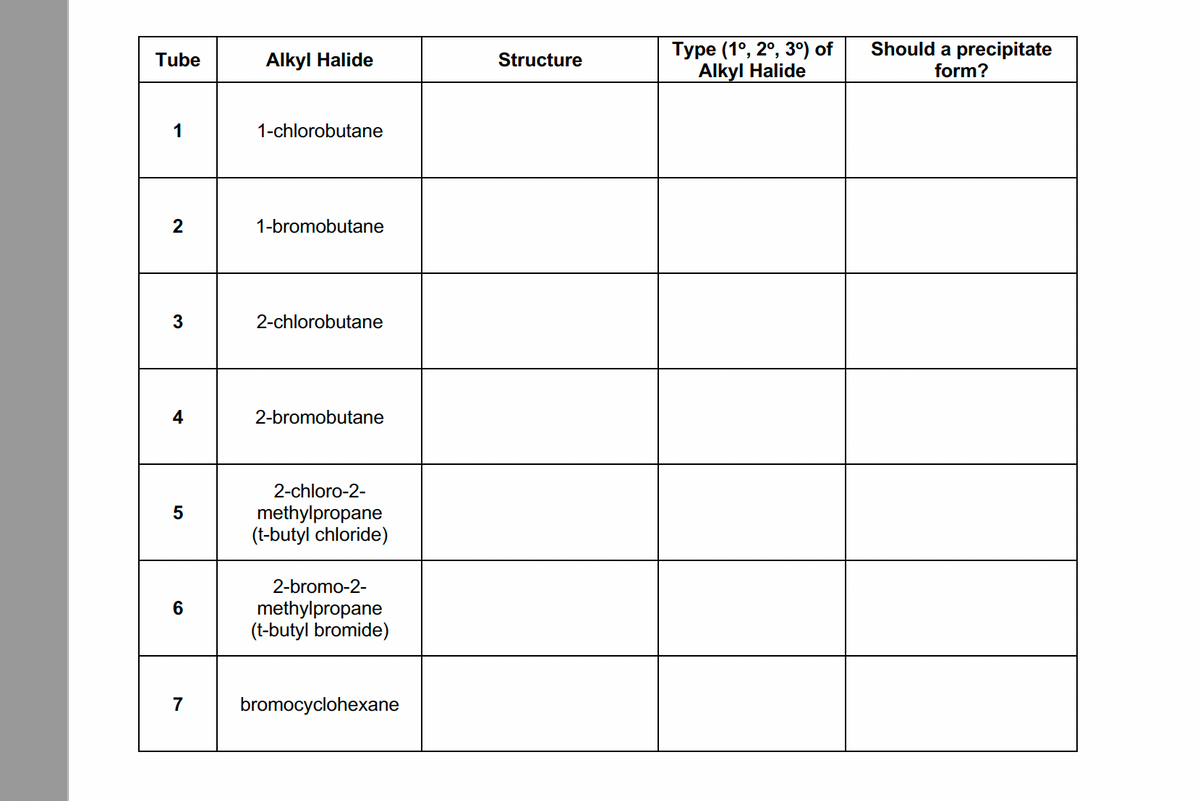
Transcribed Image Text:Should a precipitate
Туре (1°, 2°, 3°) of
Alkyl Halide
Tube
Alkyl Halide
Structure
form?
1
1-chlorobutane
2
1-bromobutane
2-chlorobutane
4
2-bromobutane
2-chloro-2-
methylpropane
(t-butyl chloride)
2-bromo-2-
methylpropane
(t-butyl bromide)
7
bromocyclohexane
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning