Q: In a chemical reaction 267 mL of oxygen gas at room temperature (25oC) and standard pressure used to...
A: According to Charles’s law, the volume occupied by a fixed amount of gas is directly proportional to...
Q: A sample of helium gas at 27.0 °C and 4.20 atm pressure is cooled in the same container to a tempera...
A: According to Gay-lussac's law: The pressure of a fixed amount of gas is directly proportional to abs...
Q: Will a B(OH)₃ precipitate form if 150.0 mL of 0.002 M BX₃ is mixed with 250.0 mL of pH 0 Ba(OH)₂ sol...
A: Ksp of B(OH)3 = 1.9 × 10-13 Molarity of BX3 = 0.002 M Volume of BX3 = 150 mL or 0.15 L BX3 dissociat...
Q: 4. Acid A has a pKa value of 5.71, and acid B has a pKa value of 5.30. Which of the two acids is the...
A: A strong acid is completely dissociated in an aqueous solution. On the other hand, a weak acid is no...
Q: Write the structural formula of the organic product for the given reaction between an alkyne and an ...
A: To write the structural formula of the organic product for the below reaction CH3CH2CCH→2. CH3CH2Br...
Q: 1. Balance the gaseous chemical equation:_C:Hs +_02 →_CO2+_H»Oa. 1,5, 3, 4 c. 2, 10, 6, 8 d. 4, 6, 8...
A: Since you have posted question with multiple sub-parts, we will solve first three sub-parts for you....
Q: Determine the overall orders of the reactions to which the following rate laws apply: (a) rate = k [...
A: The overall order of the reaction is equal to the sum of the powers of the concentrations of the rea...
Q: I need help on this problem
A: Boyle's law This law states that the absolute pressure exerted by a given mass of an ideal gas is in...
Q: Answer questions from number 2 (a and b), number 3, number 4, number 5 (a and b), number 6
A: We’ll answer the first question since the exact one wasn’tspecified. Please submit a new question sp...
Q: Consider the substrate shown. Identify the major organic product for each hydrogenation condition wi...
A: Pd/C and hydrogen will reduce alkynes all the way to alkanes – that is, two equivalents of H2 are ad...
Q: partial pressure of Ar is 0.400 atm. What is the partial pressure of Xe? Express your answer to thre...
A: Partial pressure of Xe ,Pxe = 0.600 atm
Q: A solute with a partition coefficient of 4.0 is extracted from 10.0 mL of phase 1 into phase 2. a) W...
A: Given information - partition coefficient, K = 4.0 Volume of phase 1, V1 =10.0 mL Volume of phas...
Q: A glass weighs 50.915 g in air and 23.950 g when immersed in water. What is the specific gravity?
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question s...
Q: Write the structures of all singly chlorinated products that form when 2,4-dimethylpentane is reacte...
A: 2,4-dimethylpentane on chlorination could result in the formation of three different products contai...
Q: What is the equivalent mass of the following: 1. 0.250 M HCl 2. 5.3 x 10-2 M H3PO4 3. 0.134 M NaOH ...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: Can i get help with this problem please
A: Welcome to bartleby!
Q: Addition of water to an alkyne gives a keto‑enol tautomer product. Draw the ketone that is in equili...
A: The isomers have the same molecular formula but a different structural formula. In the keto-enol iso...
Q: A sample of gas in a balloon has an initial temperature of 6 °C and a volume of 1190 L. If the tempe...
A: According to Charles's law volume of a definite amount of gas is directly proportional with absolute...
Q: What are the different methods of clarification of cane juice and chemistry involved in the clarific...
A: The purpose of the clarification process is to remove as many nonsugar components as possible, imp...
Q: Please solve faster, im waiting answers and i need answers now, thanks for solving Best regards
A: The equation for osmotic pressure P=C×R×T×i P = osmotic pressure (atm). C = molar concentration of s...
Q: A dilute aqueous solution of an organic compound soluble in water is formed by dissolving 2.35 g of ...
A: Osmotic pressure is given by the relationship: π=cRT ...
Q: (a) In an investigation of the influence of experimental variables on detection limits in electrothe...
A: A) Factors which influence sensitivity:- 1. Huge aerosol droplets 2. Huge volume of combustion gase...
Q: A volume of 18.0 L contains a mixture of 0.250 mole N2 , 0.250 mole O2, and an unknown quantity of H...
A:
Q: Sketch the following molecules or ions, include the important symmetry elements on the drawing and u...
A: The molecular structure of a molecule/ion provides information about the arrangement of the individu...
Q: High-pressure liquid chromatography (HPLC) is a method used in chemistry and biochemistry to purify ...
A: Recall the relation between Pascals(Pa) and torr 101,325 Pa=760 torr Given pressure for HPL...
Q: Draw the structures of 3‑methyl‑1‑butyne and 3,4‑dimethyl‑1‑pentyne.
A:
Q: What is the purest form of sugar?
A: First of all we will understand about the pure sugar. The pure sugar is the sweet crystalline materi...
Q: 8. Rank the following compounds from lowest to highest boiling point: CaHe. CaHsOH, CH:OCH).
A: The compounds are – C2H6 – Ethane is a type of an alkane. C2H5OH – Ethanol is a type of alcohol CH3O...
Q: Predict which of the following substituents on the benzene ring is likely to be ortho and para direc...
A: D option is correct. All other groups are meta directive like carboxylic acid, ketone, aldehyde, ...
Q: When acetic acid ionizes under normal conditions, both the departing proton and the acetate anion ar...
A: Acetic acid is a weak acid. Thus, when acetic acid is solvated by water molecules leads to its parti...
Q: f)) "Crystal field theory provides a basis for explaining the features of transition metal complexes...
A: Two statements based on coordination complexes, which are to be explained.
Q: Compare the basicitiess of the following compounds NH, NH, NH2 (1) (2) (3) CH3 NO,
A:
Q: Calculate the molarity of each of the following solutions: (a) 29.0 g of ethanol (C2H$OH) in 545 mL ...
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the ra...
Q: In an investigation of the influence of experimental variables on detection limits in electrothermal...
A: Electrothermal atomization uses graphite furnace has been the most common method for obtaining chrom...
Q: 1. 4-methylpentane 2. 3-ethyl-5-ethylhexane 3. 1,3-dimethylbutane
A:
Q: Please tell me which letter is correct and explain thoroughly thanks!
A: Covalent molecules are formed by sharing of electrons in between two or more atoms. In these molecul...
Q: The amount of caffeine in an analgesic tablet was determined by HPLC using a normal calibration curv...
A: On plotting the point in the graph,
Q: A certain gas is present in a 13.0 L cylinder at 1.0 atm pressure. If the pressure is increased to 2...
A: To find whether a gas obeys Boyle's law the following check should be made: Since, the mass of the...

Step by step
Solved in 2 steps with 2 images

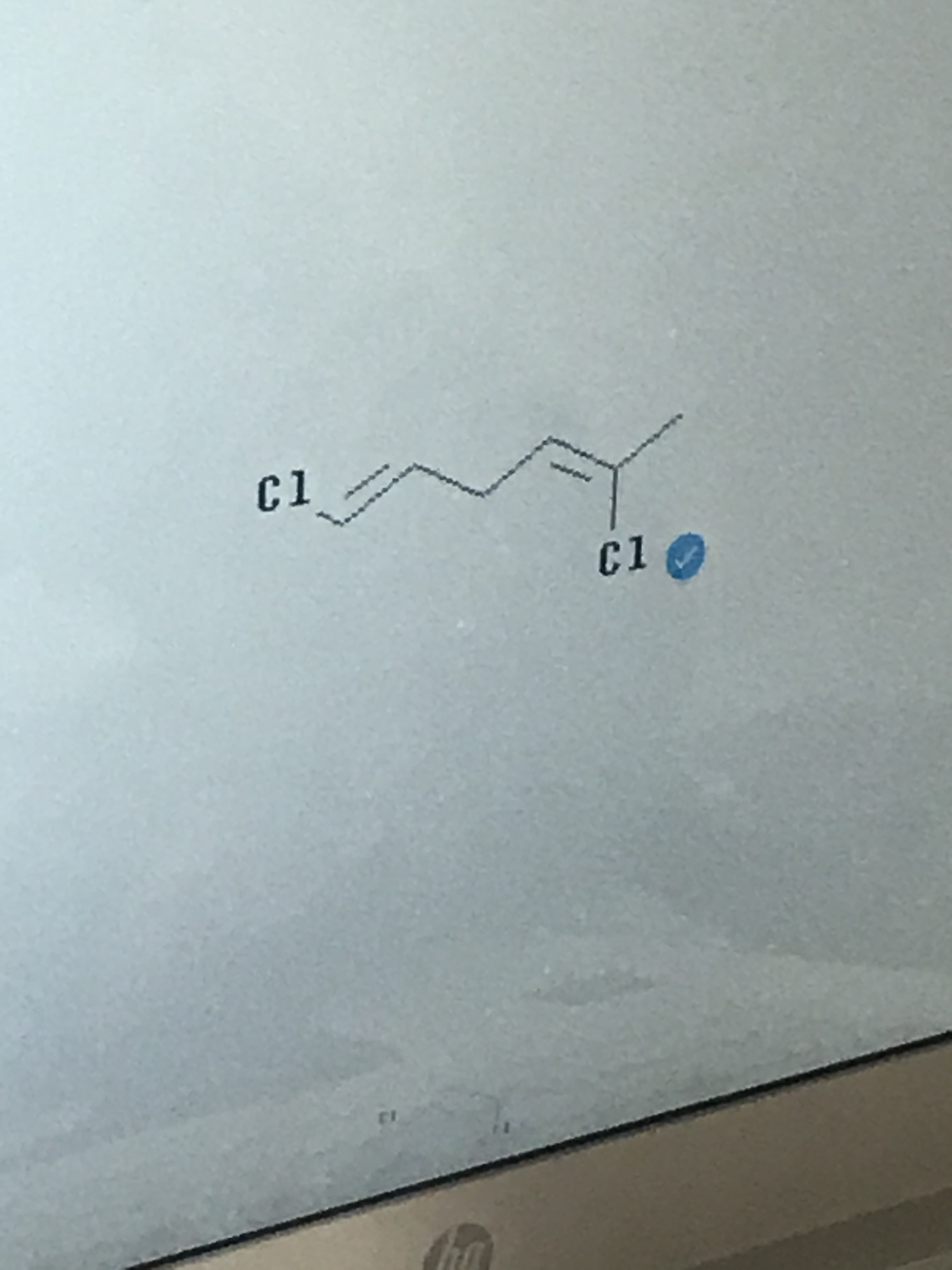
- Please provide the correct iupac name for the following compound. will vote up.Which of the following are molecular formulas? Select all that apply. Group of answer choices C3H6O3 C2H6O C2H4O2 C3H9O2 CH2O[II3] Instructions: Give the IUPAC names for each of the following compounds: (refer to the photo below) Name:
- Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference.Question 37: Give the preferred IUPAC name of the product of the response in Figure 37. [Use of lowercase letters. Do not use spaces except where necessary in the official name.] *Please provide the correct systematic/IUPAC name for the following compounds. will vote up for both.
- What is the name of the following compound? Is Ethoxycyclopentane correct?Question from at-home practice: How many and what types of carbon atoms are present in your compound? (carbonyl, aromatic, alkene, etc.)Question 30- Give the IUPAC name for the following: (Please show all of your work so I can understand going forward, also LOOK AT ATTACHED IMAGES for the picture so you can solve for the IUPAC name)

