C5 Ha+80, > 5 CO2 +6 Ha0 Calcelate the #of moles of He0 produced if 3.0 moles A CSHI2 reacts? Cs Hiz+80z ->5 COz +6 HzO hmes → 山 3.0 3.0 24 15 18
Q: What is (are) the productis) of this reaction? 1. KMO U ZHO. HO OH OH IV
A: We have given the incomplete reaction and asked for the product. The product of the above reaction…
Q: 2. A student performs the NaBH4 reduction of the triketone shown below (note that the equation is…
A:
Q: Chemical Formula Prelab Questions Name: Ashlyn Neurton Section: O0 Prepare For It! Pre-Lab Questions…
A: 4.) Mole ration is calculated by dividing the least value of moles by each value. 5.) We use mole…
Q: what is the coefficient in front of the O2 when the equation is balanced (using only whole numbers…
A: Given, Consider the reaction: C2H6O(g) +O2(g) ------>CO2(g) +H2O(g) The coefficient in front of…
Q: Give the theoretical yield, in moles, of CO2 from the reaction of 0.60 moles of C8H18 with 40.00…
A:
Q: Cl2 9. H20 10. Br2 / CCI4
A: Reaction 9(nine) will result in chlorohydrin formation. There will be no reaction in 10(ten). The…
Q: 4Cr+302 2Cr203 5. How many grams Сr are needed to produce 58.3g Cr203 by this reaction ?
A: According to reaction, 4 moles of Cr gives 2 moles of Cr2O3.
Q: The percentage yield of product F using the above reaction was 70.0%. Calculate the amount of…
A: In a chemical reaction, the participating components such as reactant reacts stoichiometrically to…
Q: Balance the equation for the combustion of butanol C4H9OH + O2 ⎯→ CO2 + H2O
A: The combustion of butanol equation has to be balanced.
Q: When 2.50g Iron (II) hydroxide is treated with 2.50g of phophoric acid. 1.99g of iron(II) phosphate…
A:
Q: Assume that student reacts 1.55 mL of 2-methylcyclohexanol (d=0.93 g/mL) with 0.50 mL of 85 %…
A: To express the efficiency of a reaction ,you can calculate the percent yield . % yield = actual…
Q: balance the following: _________C4H10 + 13O2___________ → CO2 + 10HOH in the presence of heat.…
A: Topic- Balancing the chemical reaction used in Gas Chromatography (GC)
Q: 1.3 g naphthalene, 1 mL HNO3 and 1 mL H2SO4 were used to synthesize alpha nitro naphthalene.…
A: Given: Mass of naphthalene taken = 1.3 g Volume of HNO3 taken = 1 mL Volume of H2SO4 taken = 1 mL…
Q: Acetyl Salicylate, C9H8O4, reacts with Potassium hydroxide. 1.75g of acetyl salicylate were…
A: Given data,Mass of acetyl salicylate=1.75gVolume of KOH=50cm3=0.050dm3Molarity of…
Q: A mixture weighing 9.25 mg contained only cyclohexane, C6H12 (FM 84.159), and oxirane, C2H4O (FM…
A: The reaction of combustion of both the given compounds will be as follows: C6H12. +…
Q: 39-40. Consider the chemical equation below 2 C2H2 +5 02 (MW: C2H2 = 26; 02= 32; CO2 = 44; H20 = 18)…
A:
Q: Na2SO3Na2SO3 reacts with HClO4HClO4 according to the reaction…
A: The equation is given below.Na2SO3(aq) + 2 HClO4(aq) ⟶ 2NaClO4(aq) + H2O(l) + SO2(g)
Q: ( C 2 H 5 OH) is produced from the fermentationof sucrose ( C 12 H 22 O 11 ) in the presence of…
A: Sucrose on fermentation gives ethanol and carbon dioxide. Here we have to calculate the percent…
Q: The industrial synthesis of H2 begins with the steam-reforming reaction, in which methane reacts…
A: Theoretical yield :- The maximum amount of product that can be obtained as per balanced chemical…
Q: How many moles of calcium fluoride (78.07 g/mol) will dissolve in 10.0 L of water at 25°C? Ksp of…
A:
Q: NaOH (0.5 M) Start with 0.5 mL of [Co(OH,),]* (aq) + 2C1 (aq) Product 3 [Co(OH,)6]Cl, (0.1 M) NAOH |…
A: In product 3 when we mix 1/2 mol NaOH it will replace 2 molecule of H2O molecule So product 3 is…
Q: What is the maximum yield of aspirin (ASP) that is possible to be synthesized from 3.45 g of…
A: Aspirin is synthesize from Salicylic acid . Chemical Reaction :- Salicylic acid + Acetic…
Q: 3.728 g of p-aminophenol (MW = 109.13 g/mol) is reacted with 6.487 g of acetic anhydride (MW =…
A:
Q: Given 1 tonne of a mixture of 70% malachite, CuCO3.Ca(OH)2 and 30% emerald green, Cu(C2H3O2)2.3…
A: NOTE: The formula given for malachite is incorrect. The correct formula for malachite is…
Q: What is the theoretical yleld of chromilum that can be produced by the reaction of 40.0 g of Cry03…
A:
Q: rentropy Crav 10. Arrange the following reactions in order of increasing AS° value. 1. H₂(g) + F2(g)…
A:
Q: A student runs the reaction with 1.04 mL of o-chlorotoluene (d=1.073 g/mL) and 5.0 g of potassium…
A:
Q: Na2SO3Na2SO3 reacts with HClO4HClO4 according to the reaction…
A: Number of Mole-Ratio of given mass of substance to the molar mass of substance is called number of…
Q: Calculate the mass of excess reagent remaining at the end of the reaction in which 90.0 g of so2 are…
A: Sulfur dioxide reacts with oxygen to form sulfur trioxide. The equation for the balanced chemical…
Q: Calcium oxalate monohydrate [Ca(O2CCO2)·H2O, also written as CaC2O4·H2O] is a sparingly soluble salt…
A: Solubility of calcium oxlate in water = 7.36 × 10−4 g/100 mL. Ksp = ? Molecular mass of CaC2O4 .…
Q: 1.3 g naphthalene, 1 mL HNO3 and 1 mL H2SO4 were used to synthesize alpha nitro naphthalene.…
A: Given: Mass of naphthalene taken = 1.3 g Volume of HNO3 taken = 1 mL Volume of H2SO4 taken = 1 mL…
Q: Show your work: Balance the following chemical equation: C7H16O5 + O2 ---> CO2 + H2O
A: The given reaction is, C7H16O5 + O2 ---> CO2 + H2O
Q: What is the coefficient in front of potassium hydroxide when the equation is balanced using smallest…
A: The reaction given is S (s) + O2 (g) + KOH (aq) → K2SO4 (s) + H2O (l) In the above reaction,…
Q: For the reaction below, how many grams of C2H5OH can be produced from the decomposition of 33.65 g…
A: Given, mass of C6H12O6 = 33.65 g Thus, moles of C6H12O6 = Mass of C6H12O6Molar mass of C6H12O6…
Q: 8. Calculate the percentage yield of t-butyl chloride if we obtained 9.3g product from 16.7 mL of…
A:
Q: Consider the series of reactions to synthesize the alum (KAl(SO4 )2 · xH2O(s)). If you start the…
A: The limiting reagent in a chemical reaction is a reactant that is present in smaller quantity and…
Q: Number of grams of Ca3(PO4)2 and grams of KCI is....can be produced by mixing 5g of CaCl2 with 8g of…
A: To calculate the grams of the product obtained. First of all we have to determine the limiting…
Q: 1. a) Definę the term mole b) A carbonate of metal X has the formular X,CO. The carbonate reacts…
A: Since you have posted multiple questions, we will solve first question for you. Please repost other…
Q: The structure for 2-(4-fluorophenyl)-3-phenyl-1,3-thiazolidin-4-one is shown above. It has a molar…
A: Given: Compound: 2-(4-fluorophenyl)-3-phenyl-1,3-thiazolidin-4-one. Molar mass of the compound = 273…
Q: One mole of a compound weighs 2.93x10-22. Determine the molar mass (g/mol). Group of answer choices…
A: NOTE : In questions, there is a small typo error. It will be mass of 1 molecule of compound and not…
Q: Sodium trioxalatocobaltata(III) triahydate is prepared by the following reactions:…
A:
Q: 1) Fill in the following data table with the missing quantities NO₂ H₂O H₂SO4 + NaNO₂ KI NH₂…
A: For H2SO4 : We know, density = Mass / Volume Mass = Density * Volume = 18 M * 2 mL…
Q: Calculate the mass of excess reagent remaining at the end of the reaction in which 900 g of SO2 are…
A:
Q: The equations are not balanced. Which equation would have the same coefficients in the same order as…
A:
Q: How many litters of HCI [5.2M] would react completely with 325g of Na2CO3? [1 N22CO3 + 2 HCI → 2…
A:
Q: What is the MAIN product of the chemical sequence below? 1) NaOEt, EtOH, heat 2a) BH3-THF 2b) H2O2,…
A: Given reactant :- Reagents 1. NaOEt,EtOH , Heat 2. BH3-THF , 3. H2O2
Q: 9.240 g of p-aminophenol (MW = 109.13 g/mol) is reacted with 6.173 g of acetic anhydride (MW =…
A:
Q: 2.SO2 t Oz → 2S03 If 42019 of 80z reacts with 28-6g Df 0z be how produce!? many grams of SDg oil
A:
Q: 19. From the following reaction scheme, "CHs HCI FeBr, When 4 g of ethanoyl chloride was used, 3.24g…
A: Moles of ethanoyl chloride = 4 / 78.5 Moles of ethanoyl chloride = 0.051 moles Moles of…
Q: 24. How many moles of Fe203 are produced when 0.278 moles of Fe is reasted with O2? 4 Fe • 3 02 →2…
A:
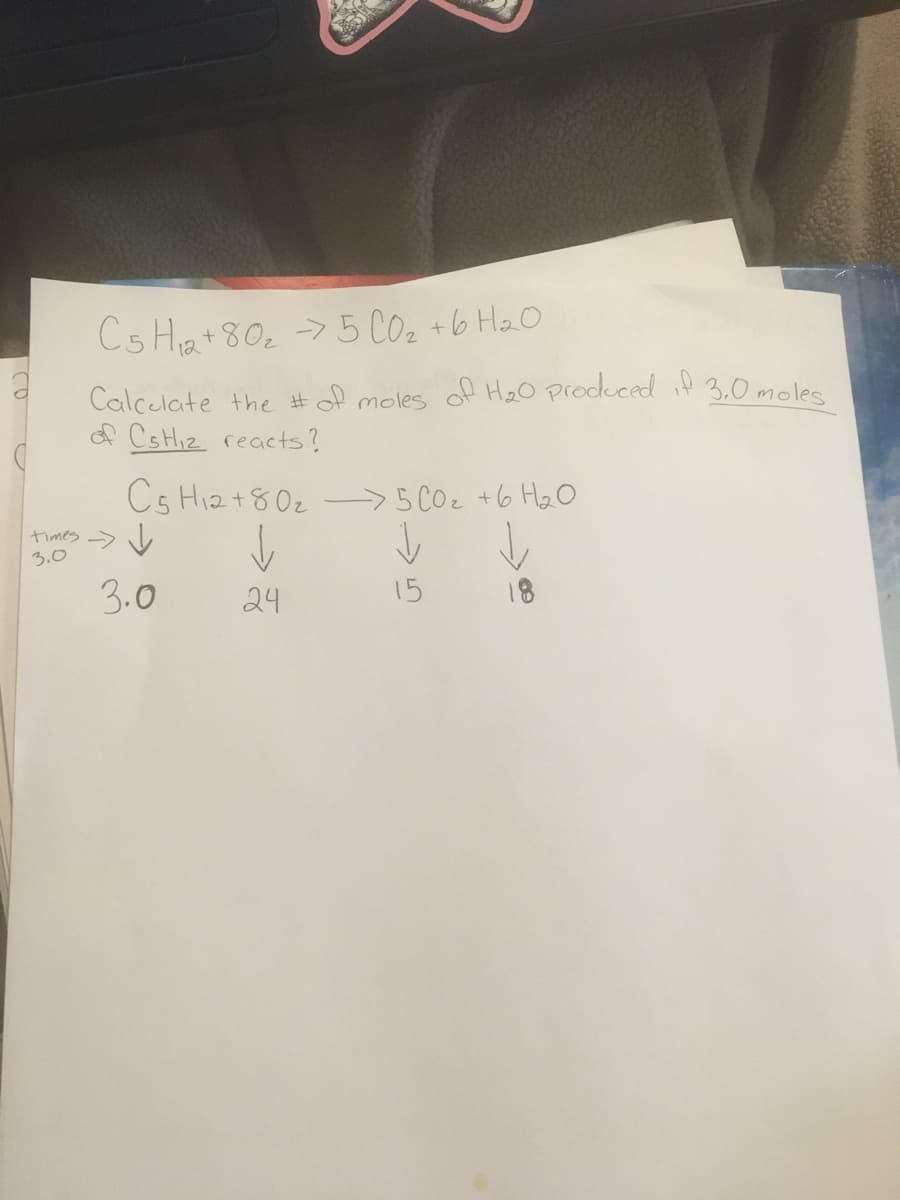
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- When the reactionP4O10 + KOH → K3PO4 + H2Ois balanced, how much KOH is required toreact with 3 moles of P4O10?1252. WG compound weighing 1980 h pressure, yielding An unknown organi d 360 mg of water- 12 my s fully combusted at hig ‘9’30 g of carbon dioxide an hat is its empirical formula? A) CHO, B) CHO, ©) CHO, D) CH,0,Write the empirical formula for the hydrated KAl(SO4)2, based on moles of anhydrous KSI(SO4)2= 0.046 mol molar mass of H2O= 18g/ mol moles of H2O= 0.0444mol ratio pf moles H20 to moles of anhydrous KAI(SO4)2= 9.65/1 . Show all work including units. Hint: if the ratio of moles of H2O to moles of anhydrous KAl(SO4)2 was 4, then the empirical formula would be: KAl(SO4)2•4H2O.
- A well is punched into a slab of gelatin and a solution ofequimolar amounts of NaCl and KCl is placed in it. After anhour the concentration of Na+ and K+ present in the well ismeasured. Which ion, Na+ or K+, would be present ingreater abundance in the well? Explain your answer.Sample: Saline 0.900% (m/v), in sodium chloride (NaCl). Data: M.A. (g/mol): Na = 22.9898; K = 39.0983; Cr=51.9961; Ag = 107.8682; Cl = 35.453; N=14.0067; O = 15.9994. Material available in the laboratory's warehouse: Reagents: distilled water; standardized solution of silver nitrate (AgNO₃), at a concentration of 0.09980 mols/L; Potassium chromate solution (K₂CrO₄) 1%; ammoniacal ferric alum solution [Fe(NH₄)(SO₄)₂] and; nitrobenzene (or cooking oil, alternatively); 0.1000 mols/L potassium thiocyanate (KSCN) standard solution. Glassware: beakers of all sizes available on the market; 50, 100, 150, 200 and 250 ml Erlenmeyers; 10.00 and 25.00 mL volumetric pipettes; 15.00 mL burette; 30.00, 50.00 and 100.00 mL volumetric flasks Calculate the molarity of saline, in terms of the NaCl concentration (concentration in mols/L). (a) 0.900 mols/L (b) 5.84 mols/L (c) 0.154 mols/L (d) 0.0154 mols/LMass of Na2CO3.H2O (g) = 2.12g (g) Mass of the CaCl2.2H2O (g) = 1.98g Mass of the top funnel + filter paper (g) = 15.85g Mass of top funnel + filter paper + CaCO3 collected (g) = 17.81g CaCl2 + Na2CO3 ==== CaCo3 + 2NaCl Theoretical yield in moles and grams? Moles of reagent in excess left unreacted? Mass of precipitate? Experimental yield? Percent yield?
- For the reaction C2H4 + 3O2 ---> 2CO2 + 2H2O, does this mean that ten (10) grams of CO2 produced reuired 0.345 mol O2?In the common process for the manufacture of nitric acid, sodium nitrate is treated with aq. sulphuric acid containing 95% H2SO4. In order that the resulting "niter cake" may be fluid; it is desirable to use sufficient acid so that there will be 34% H2SO4 by wt in the final cake. This excess H2SO4 will actually be in combination with the Na2SO4 in the cake, forming NaHSO4, although for purposes of computation it may be assumed to be free acid. The cake will contain 1.5% water and the reaction will go to completion, but 2% of the HNO3 formed will remain in the cake. Assume that the NaNO3 used is dry and pure. Calculate per 100 kg of NaNO3 charged: (a) wt and % composition of the niter cake (b) wt of aqueous acid to be used (c) wt of water vapor and nitric acid distilled from the niter cake.Assume that 1.00 mL (1.02 g) of crude product mixture is obtained from the reactionbefore the washing steps, and the distribution constant of the product mixture in brine isK = (Cmixture/Cbrine) = 50.0. Assuming that after mixing the product mixture volume isstill 1.00 mL (i.e., the volume lost is small), and the volume of the first brine wash layeris 2.00 mL. What mass (grams) of product mixture is lost (dissolves) in the first brinewash? (See Technique 13.2, p 53.) Show work.
- 43. In the preparation of Rochelle salt (MW = 282.83 g/mol), 7.2 grams of monohydrated Sodium carbonate (MW = 124.01 g/mol) dissolved in 80 mL water was made to react with 20g of Potassium bitartrate (MW = 188.10 g/mol). What is the amount of Rochelle salt obtained from Monohydrated Sodium carbonate? Rochelle's Salt:2 KHC4H4O6 + Na2CO3.H2O + 6 H2O -> 2 KNaC4H4O6.4H2O + CO2 ↑ a 38.77 g b 28.74 g c 36.48 g d 32.84 gWhen Na3PO4 and Ca(NO3)2 are combined , the following reactions occurs:2PO4 3- (aq) + 3 Ca2+ (aq) ------ Ca3PO4)2How many grams of Ca3(PO4)2 (MM = 310.18 g/mol) are obtained from 15.00 ml of 0.1386 M Na3PO4 are mixed with 20.00 ml of 0.2118 M Ca(NO3)2 ?Is it conceivable to have both a limiting and an excess reactant in the presence of a perfect one to one ratio?

