What is the theoretical yleld of chromilum that can be produced by the reaction of 40.0 g of Cry03 with 8.00 g of aluminum according to the chemical equation below? 2AI - Cr203 - Al203+ 2Cr Multiple Choice 3089 77g 15.4g 2739 4999
What is the theoretical yleld of chromilum that can be produced by the reaction of 40.0 g of Cry03 with 8.00 g of aluminum according to the chemical equation below? 2AI - Cr203 - Al203+ 2Cr Multiple Choice 3089 77g 15.4g 2739 4999
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter20: Organic Chemistry
Section: Chapter Questions
Problem 37E: MTBE, Methyl tert -butyl ether, CH3OC(CH3)3, is used as an oxygen source in oxygenated gasolines....
Related questions
Question
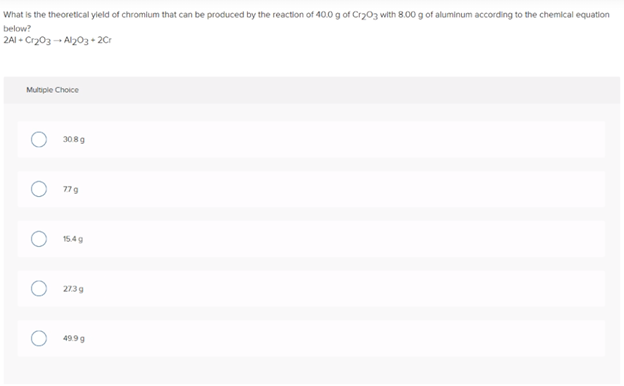
Transcribed Image Text:What is the theoretical yleld of chromilum that can be produced by the reaction of 40.0 g of Cry03 with 8.00 g of aluminum according to the chemical equation
below?
2AI - Cr203 - Al203+ 2Cr
Multiple Choice
3089
77g
15.4g
2739
4999
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning