Calculate AH for the reaction: 2 N2(g) + 5 O2(g) →2 N2O5(g) using the following three equations: H2(g) + O2(g) → H₂O(1) N2O5(g) + H2O (1) → 2 HNO3(1) 12 N2(g) + 3/2 O2(g) + ½ H2(g) → HNO3(1) AH°=-285.8 kJ ΔΗ° = -76.6 kJ AH°=-174.1 kJ
Calculate AH for the reaction: 2 N2(g) + 5 O2(g) →2 N2O5(g) using the following three equations: H2(g) + O2(g) → H₂O(1) N2O5(g) + H2O (1) → 2 HNO3(1) 12 N2(g) + 3/2 O2(g) + ½ H2(g) → HNO3(1) AH°=-285.8 kJ ΔΗ° = -76.6 kJ AH°=-174.1 kJ
Chapter17: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 53E: From data in Appendix 4, calculate H, S, and G for each of the following reactions at 25C. a. CH4(g)...
Related questions
Question
100%
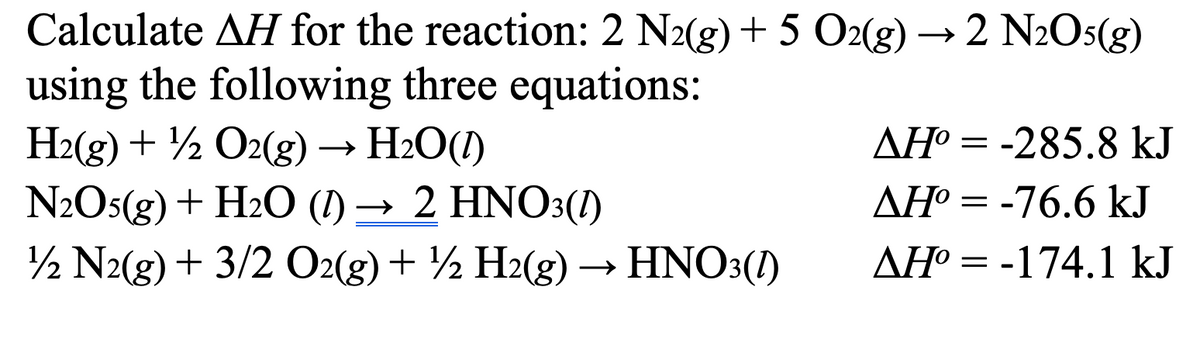
Transcribed Image Text:Calculate AH for the reaction: 2 N2(g) + 5 O2(g) →2 N2O5(g)
using the following three equations:
H2(g) + O2(g) → H₂O(1)
N2O5(g) + H2O (1) → 2 HNO3(1)
12 N2(g) + 3/2 O2(g) + ½ H2(g) → HNO3(1)
AH°=-285.8 kJ
ΔΗ° = -76.6 kJ
AH°=-174.1 kJ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning