Q: Explain why a solution of ammonium nitrate is a weak acid. Give a balanced rection equation…
A: The objective of the question is to explain why a solution of ammonium nitrate behaves as a weak…
Q: solve this with step by step calculations
A: Approach to solving the question:For any solution to be a buffer solution, it should fall into one…
Q: Problems: 1. Predict the product of the following reaction and show the mechanism. O 0 1) KOiPr,…
A: Step 1:α− Alkylation of carbonyl (aldehyde or ketone) and ester:The α−H of the carbonyl/ester group…
Q: Draw structural formulas for organic products A and B in the window below. CI Li CH3CHCH3 pentane…
A: Step 1:Alkyl chloride reacts with Li to form alkyl lithium. Isopropyl chloride reacts with Li (in…
Q: 13. Consider the combustion of one mole of glycine, NH2CH2COOH(s), to form carbon dioxide gas,…
A: 13.1 The balanced reaction is:- 4C2H5O2N(s) + 9O2(g) → 8 CO2(g) + 10H2O(l) + 2N2(g) 13.2 We know:-…
Q: Phosgene decomposes to carbon monoxide and chlorine gas. COCl2 (g) CO (g) + Cl2 (g) If the reaction…
A: The question is asking about the effect of adding more phosgene gas (COCl2) to a reaction that has…
Q: Determine the temperature of a reaction if K = 1.2 x 10⁻⁶ when ∆G° = +18.1 kJ/mol.
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 7. Which of the following molecules exhibits E/Z isomerism? а A C D
A: For a molecule to exhibit E/Z isomerism:It must contain a C=C double bond.Each carbon atom of the…
Q: Don't use extra explanation solve correct
A:
Q: EtO. 2. Complete the road map by providing the structures of the products or the reagents as needed…
A:
Q: Determine the value of Kc for the reaction 2 A ⟷ 1 B for the following equilibrium…
A: Formula for calculating KcAccording to the question following is the reaction 2 A ⟷ 1 B…
Q: Enter electrons as e*. Use smallest possible integer coefficients for ALL reactions. If a box is not…
A: Step 1:Galvanic cell is an electrochemical cell that converts chemical energy into electrical energy…
Q: A sample of neon gas occupies a volume of 6.57 L at 67.0°C and 315 torr. If the volume of the gas…
A: The objective of this question is to find the resulting gas pressure when the volume and temperature…
Q: The following mechanism was proposed for the reaction with overall stoichiometry A + B → R A → X X+…
A: Step 1: The mechanism given for the reaction is A↔X X+B→R The intermediate X can be…
Q: A sample of argon gas occupies a volume of 6.57 L at 53.0°C and 0.880 atm. If it is desired to…
A: The objective of this question is to find the temperature of a sample of argon gas when its volume…
Q: The standard electrode potential (E) for the half-reaction Ni2+(aq) + 2e → Ni(s) is -0.257 V. What…
A: Step 1:In a galvanic cell, oxidation always takes place at anode and reduction always takes place at…
Q: For the reaction HCI (aq) + CO2 (aq) = Cl¯ (aq) + HCO3 (aq) identify one change that would shift the…
A: The objective of the question is to identify a change that would shift the equilibrium of the given…
Q: Draw the structure(s) of the major product(s) of the following reaction after workup to neutralize…
A:
Q: 2. A sample of table sugar (sucrose, C12H22O11) has a mass of 1.202 g.
A:
Q: 12H2O, Write the equation for the other half-reaction, called the dark reaction, for…
A: The objective of the question is to write the equation for the dark reaction, also known as the…
Q: Please don't upload any image just give me the answer with proper explanation
A: Approach to solving the question:Read alot of related books or research onlineDetailed…
Q: Calculate the mass (in g) of SiO2 that reacts completely with 15.0 grams of carbon according to the…
A: Given mass of carbon= 15.0 gMolecular mass of carbon = 12.0 g/molNumber of moles of carbon= given…
Q: 7. Copper metal crystallizes in a face-centred cubic structure. If the atomic radius of copper is 64…
A: The objective of this question is to calculate the volume of a unit cell of copper, which…
Q: Consider a 1mg sample of cobalt- 60(). What is the expected exposure rate 1m away from the source…
A: the problem appears to be about calculating the expected exposure rate from a 1 mg sample of…
Q: Draw the peptide Glycine-Alanine-Serine-Cysteine-Isoleucine-Glutamic acid Tryptophan-Valine. Circle…
A: This is an 8-amino acid peptide with the sequence…
Q: A 1.44-g sample of an unknown gas has a volume of 896 mL and a pressure of 742 mm Hg at 37.9 °C.…
A: The objective of this question is to calculate the molar mass of an unknown gas given its mass,…
Q: At 460 K, ∆G = -13.1 kJ/mol for the reaction 2 A (g) → B (g). If the partial pressures of A and B…
A:
Q: A chemistry graduate student is studying the rate of this reaction: 2SO3 (g) → 2SO2(g) + O2(g) She…
A: Time, ms[SO3], M00.090010.0.050320.0.028130.0.015740.0.00877Step 1: Plot the [SO3] vs. time. This…
Q: Write the reduction half-reaction for Fe(s)+2H+(aq)→Fe2+(aq)+H2(g)Fe(s)+2H+(aq)→Fe2+(aq)+H2(g)
A: The objective of the question is to write the reduction half-reaction for the given chemical…
Q: Solve.
A: The objective of the question is to understand the properties of hydrocyanic acid (HCN) and…
Q: Which reaction will occur if each substance is in its standard state? Assume potentials are given in…
A: Approach to solving the question: I believe that this is the complete question together with its…
Q: Chemistry
A: Step 1: Step 2: Step 3:
Q: Construct the wave function, psin,l,m(r, theta, phi), for the 3s, 2p and 3p and 3d orbitals of the…
A: The objective of the question is to construct the wave function for the 3s, 2p, 3p and 3d orbitals…
Q: A 0.760-g sample of an unknown gas has a volume of 759 mL and a pressure of 399 mm Hg at 34.2 °C.…
A: The objective of this question is to calculate the molar mass of an unknown gas given its mass,…
Q: 4) i) Fill in the reagents to complete the synthetic sequence below, and draw the final product in…
A:
Q: Don't use guidelines answer just specific answer correct don't use ai okk. Just solve accurate not…
A: Here's a table of the one-letter abbreviation of peptides. This was the reference for the answer…
Q: O II III O IV OV What is the structure of (1S,2R)-1-ethyl-2-methyl-1,2-epoxycyclohexane? .... www IV…
A: The objective of the question is to determine the structure of the compound…
Q: Place the methods listed in the table below in order of increasing sensitivity for the detection of…
A: To order the methods by increasing sensitivity for the detection of tellurium (Te), we need to…
Q: Determine E° for a galvanic (voltaic) cell if AG° = -6.3 kJ/mol and n = 3. (F = 96,500 J/(V⚫ mol))…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 1. Identify the following amino acid and a pH at which the structure would predominate: A. Y, pH…
A: The shown molecular structure is of tyrosine, which is also represented by one letter Y, among the…
Q: . Write balanced nuclear equations for the following:A) Formation of 48Ti through positron…
A: The objective of the question is to write balanced nuclear equations for the formation of 48Ti…
Q: CI + ΑΙ CH2C12 4 + HCI
A: Step 1: Step 2: Step 3: Step 4:
Q: Don't provide hand writing solution
A:
Q: Which of the following are true of the products of the following reaction? ピ excess HBr Copyright…
A:
Q: Draw the major product for this reaction. Ignore inorganic byproducts. 1. disiamylborane, THF 2.…
A:
Q: Draw the structure(s) of the major organic product(s), including counterions, of the following…
A:
Q: Using this reaction energy diagram, determine the activation energy for the reverse reaction. 140…
A:
Q: - Find the limiting reagent - Find the theoretical yield of both KHSO4 and Cr2(SO4)3 in grams - Find…
A: Step 1: Here, we are given following reaction:3 H2C2O4(aq) + K2Cr2O7(aq) + 5 H2SO4(aq) → 2KHSO4(aq)…
Q: Pure benzene freezes at 5.50°C. A0.9905 gram sample of an unknown nonelectrolyte is dissolved in…
A: Freezing point of pure benzene Tf0 =5.50°CMass of benzene 15.388 gram = 15.388 ÷1000 Kg=0.015388…
Q: dont provide handwriting solution .....
A: The reaction mechanisms of the given schemes will be -
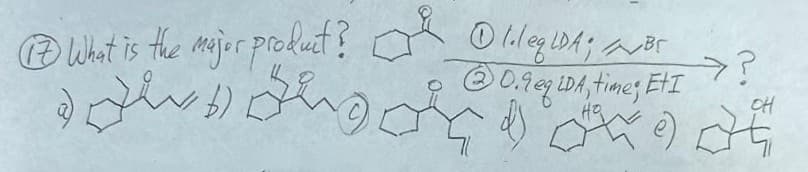
Step by step
Solved in 2 steps with 2 images

- Which is (are) able to oxidize Cl - ?(a) F2(b) F -(c) Both Br2 and I2(d) Both Br - and I -#29b except NO3 it should be NO2The molecular weight of hemoglobin is 64,500 daltons (= g mol-1). A. How much oxygen can be bound per g of hemoglobin? Express the values as mL per g of hemoglobin, where mL is the volume of O2. R =0.082 L atm mol-1 oK-1, oK =273 + oC. The molar volume of O2 is given as V = nRT/P where P = 1 atm and n= 1 mol. (Hint: 1.0 mol x 0.082 L atm mol-1 oK-1 x 273.2 oK / 1 atm = 22.4 L mol-1) (Hint: The number of moles of O2 that can be bound per g of hemoglobin is 4 x the moles of Hb in 1 g). B. The blood in a person contains 15 g of hemoglobin per 100 ml blood. How much oxygen does the blood contain at 95 mm Hg if hemoglobin is 98% saturated and the solubility of O2 is 0.003 mL dL-1 mmHg-1? (Hint: Total O2 content = HbO2 + free dissolved O2) C. Suppose the venous PO2 is 40 mm Hg, and that hemoglobin is 75% saturated at this point. What is the oxygen content of venous blood? D. Suppose the cardiac output is 5 L min-1. Calculate the oxygen delivery to the tissues from B and C. (Hint: Rate of…
- Give typed full explanation A stock solution of lithium phosphate (Li3PO4, MM= 115.794361 g/mol) is prepared by dissolving 295.8 g Li3PO4 in deionized water to a final total volume of 1.000 L. Suppose a 1.000-mL aliquot is taken from this stock solution and transferred to a new container to which deionized water is added until the total volume is 50.00 mL. A new 1.000-mL aliquot is taken from this 50.00 mL solution and transferred to a third container to which deionized water is added until the total volume is 25.00 mL. Calculate the number of individual lithium ions (Li+) in the final 25.00-mL solution. Note that lithium phosphate is a strong electrolyte that completely dissociates in water (note that (aq) means the species is a solute in an aqueous solution): Li3PO4 (aq) 3 Li+ (aq) + PO43– (aq) Report your answer to two significant figures. Use scientific notation. Example: 1.0*10^23 (this notation means 1.0 1023)Excessive nutrients dumped into waterways can create “algae blooms” that rapidly consume all of the dissolved O2 and trigger production of enough H2S to create “dead zones” like the ones that exist off the coast of Denmark and in the Gulf of Mexico immediately south of the Mississippi River delta. a. Since Group 16 element substitution is a recurring theme in that Group’s chemistry –e.g., oxygen substituting for Se and Te in the production of H2Se and H2Te, respectively –maybe molecular oxygen (O2) could be bubbled into these dead zones and react with the H2S.Write out a balanced chemical equation for the reaction between H2S and O2 to form elemental sulfur (S8) as a product that would precipitate out. b. Using bond dissociation energies, calculate if this reaction would be thermodynamically favorable. c. If your answer to part (b) is “yes”, do you think that simply bubbling O2 into these waters would work? Justify your answer.Why are the carboxylic acid groups of the amino acids so much more acidic (pK a ~ 2) thana carboxylic acid such as acetic acid (pKa = 4.76)?
- keAssignment/takeCovalentActivity.do?locator=assignment-take n progress Q Search 40 eaction is Submit Answer In the following equation for a chemical reaction, the notation (s), (I), or (g) indicates whether the substance indicated is in the solid, liquid, or gaseous state. 2NO(g) + 2H₂(g) →→→ N₂(g) + 2H₂O(l) + energy Identify each of the following as a product or a reactant: NO(g) H₂0(1) H₂(g) N₂(g) When the reaction takes place energy is V2 7 L Retry Entire Group = 18 + Use the References to access important values if needed for this question. fg 0 OWLv2 | Online teaching and lear X hp 9 8 more group attempts remaining f10 MO f11 PAI + 112 G ins prt sc Ⓡ ✰ FRIEVIDUS Next> ^ @ ¹0 delete backspace home lock 5:- 5/14For photosynthesis to occur, 2801 kJ/mole of energy is required. Add the ΔH to the correct side of the equation below: 6 CO2 (g) + H2O (l) → C6H12O6 (aq) + 6 O2 (g)ACID DEPOSITION: what are the methods used to reduce the incidence of acid deposition and photochemical smog; e.g., reducing sulfur content in fuels, using catalytic converters in automobiles, smokestack scrubbers

