Calculate AHxn in kJ/mol of NaOH, if the initial temperature is 25.00 °C and the final temperature is 31.22 °C, when 100 mL of 1.000 M NaOH is mixed with 100 mL of 1.000 M of HCI. (Heat capacity = 4.184 J/ °C.g, density of solution = 1.01 g/mL) You must show your work! Cor tho toolhor press ALT+F10 (PC) or AI T+FN+F10 (Mac).
Calculate AHxn in kJ/mol of NaOH, if the initial temperature is 25.00 °C and the final temperature is 31.22 °C, when 100 mL of 1.000 M NaOH is mixed with 100 mL of 1.000 M of HCI. (Heat capacity = 4.184 J/ °C.g, density of solution = 1.01 g/mL) You must show your work! Cor tho toolhor press ALT+F10 (PC) or AI T+FN+F10 (Mac).
Chapter6: Thermochemistry
Section: Chapter Questions
Problem 104AE
Related questions
Question
100%
2
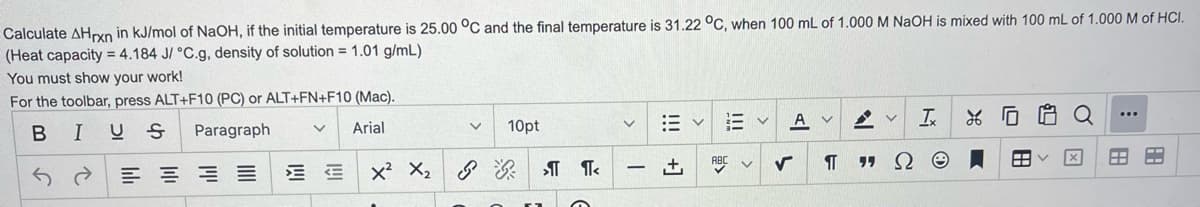
Transcribed Image Text:Calculate AHxn in kJ/mol of NaOH, if the initial temperature is 25.00 °C and the final temperature is 31.22 °C, when 100 mL of 1.000 M NaOH is mixed with 100 mL of 1.000 M of HCI.
(Heat capacity = 4.184 J/ °C.g, density of solution = 1.01 g/mL)
You must show your work!
For the toolbar, press ALT+F10 (PC) or ALT+FN+F10 (Mac).
I U S
Paragraph
Arial
10pt
田
E E x X2
+,
>
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning