Q: Calculate the solubility of AgBrOz in water at 25 °C. You'll find K data in the ALEKS Data tab. ds.…
A: In this question, we want to determine the solubility of AgBrO3 in g/L unit You can see details…
Q: A student evaporated 150. mL of a saturated solution of MgC2O4. If 0.16g of solute remains,…
A:
Q: NEUTRALIZATION REACTIONS Can you help me with this one? The given problem is in the image that I've…
A: To Calculate: 1. The molecular weight of potassium biphthalate (C8H5KO4) 2. The mEq of potassium…
Q: Vinegar is a 5% (w/v) solution of acetic acid (HC2H3O2) in water. How many moles of acetic acid are…
A:
Q: What will change the value of Ksp for Mg(OH)2? O Heat the solution. O Add water to the solution.
A:
Q: An unknown liquid has a freezing point of 25.5oC, however, it absorbs H2O (as impurity) on exposure…
A: Given: unknown liquid freezing point (T0) =25.50C Sample freezing point (Ts) = 24.590C Kf of the…
Q: What is the normality of 0.050 M Cu2+ solution based on the reaction below? Cu2+ (ag)+ 2e- Cu (s)
A: Given that, Cu2+(aq) + 2e-= Cu(s) Concentration of Cu2+=0.050 M Normality=?
Q: Which of the salts below would cause the temperature to increase the most, when 5.00 grams of the…
A: Given-enthalpy of dissolution of MgCl2 = -160 KJ/mol Using the formula we have No. Of mole of Mgcl2…
Q: What is the molarity of 10.0 mL of 75% v/v ethanol
A: We have to find Molarity if 10 mL of 75% v/v ethanol. This means that in a 10ml solution, 7.5mL…
Q: 6.25 gm of Sodium Chloride dissolved in distill water , then the volume completed to IL , found that…
A: In aqueous Sodium Chloride when Silver Nitrate is mixed ,Insoluble Sodium Chloride salt is formed.…
Q: Which solution is the most concentrated? Convert all solutions to g/dL show work A 6.7% B 1:5 C…
A: Percentage concentration of solution (m/v) : It is used To define percentage of ratio of mass of…
Q: In the following reaction calculate and find the normality when it is 1.0 M H3PO4? H3PO4 + 2NaOH →…
A:
Q: The solubility of ZNCO, in water at 25 °C is measured to be 0.0013 Use this information to calculate…
A: Since, solubility should be in mol L-1 So, first of all we will convert its units and then, Ksp will…
Q: You are working in an aseptic unit making TPN bags. One of your adult patients requires 20mmol of…
A: Given the stock solution= 5% sodium chloride. Let the mass of the solution be 100g. Then the mass…
Q: The solubility of CaF, in water at 25 °C is measured to be 0.017 . Use this information to calculate…
A: Solubility of CaF2 = 0.017 g/L Ksp for CaF2 = ?
Q: Salubility product For iran CIII) Flouride, is -4.4X 10?. What is the molar salubliity Of Iran…
A: Ksp =SOLUBULITY PRODUCT STRUCTURE OF IRON(III) FLOURIDE FeF3
Q: What is the maximum solubility of silver carbonate, Ag2CO3, in g/L, if the solubility product for…
A:
Q: The equation for the dissolving of lead (II) chloride (including heat) is: PBCI2(s) + heat = Pb2-…
A: According to LeChatleir's principle "when a system at equilibrium is subjected to a change in…
Q: For the following reaction: 2 HCIO4 + Ca(OH)2 → Ca(CIO4)2 +2 H2O (1) What is the equivalent mass of…
A: Equivalent mass:- It is equal to the mass of a given substance which will combine with or displace a…
Q: Calculate for the molarity if 250. mL of solution contains 1.838 mol of solute
A: molarity of the solution is defined as the number of moles in the unit volume of the solution
Q: calculate the concentration (w/w%) when 15g of calcium chloride is dissolved in 125g of water.…
A: concentration (w/w%) = mass of solutemass of solution×100 where, mass of solute (calcium chloride) =…
Q: folarity of HC1 Standard Solution olume (mL) HC1 Standard Solution o. of moles HCl =No. of moles H…
A: We have to find the molarity of NaOH for given reaction in trial 2
Q: why is lead (II) sulfate more soluble in water than in any solution containing Pb2 + or SO4 2- ions?
A: The question is based on the concept of chemical equilibrium. we have to explain the High…
Q: What is usually the relationship between the volume needed to reach the end point (Vep) and the…
A: Equivalence point and end point are the terms associated with the titration of an acid with a base.…
Q: Many instant hot and cold packs (to soothe muscle pain and as first aid for sprains, respectively)…
A: The hot and cold packs which are used as pain relief purpose must product heat or cold to alleviate…
Q: What happens (increase or decrease), in the system at eq≡m, to: SiO2(s) + 4HF(g) SiF4(g) + 2H2O (g)…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: Is the dissolution of NH4Cl an exothermic or endothermic reaction?
A:
Q: Calculate the solubility of Fe(OH), in water at 25 °C. You'll find Kn data in the ALEKS Data tab. sp…
A:
Q: What willl be the Molar concertration of the 306.29 HCl solution prepared from 3 mL of concentrated…
A: To solve this problem we will first calculate mass of HCl . Then number of mole HCl will be…
Q: If given a 100% (v/v) ethanol solution at room temperature, calculate and prepare 100 mL of the…
A:
Q: 1) Dissolution and precipitation a) explain whether the dissolution of a salt is an exothermic or…
A: Here we have to explain dissolution of salt in aqueous solution is exothermic or endothermic.
Q: The solubility of CaF, in water at 25 C is measured to be 0.017 Use this information to calculate K,…
A:
Q: Question attached
A: Given, percentage of H2SO4 in water by volume = 85 % volume of 85% H2SO4 used in the…
Q: The dissolution of NH,Cl in water is an endothermic process. If the initial temperature of water is…
A: Endothermic reaction is reaction in which energy/heat is consume in reaction.
Q: The equation for the dissolving of lead (II) chloride (including heat) is: PBCI2(s) + heat Pb2+ (a…
A:
Q: Write Kc or Kp of the following: 1. 2N2O5 (g) ↔ 4NO2 (g) + O2(g)
A: A question based on equilibrium concept, which is to be accomplished.
Q: 25- How to prepare 70% (v/v) Ethanol in water? (Ethanol is absolute, Mwt 46 g/mol) A) By Diluting…
A: The concentration in v/v of some solute means the volume of solute that is present in a certain…
Q: Calculate the solubility of Mg (OH), in water at 25 °C. You'll find Kn data in the ALEKS Data tab. 2…
A: Given that Temperature = 25°C
Q: How many milliliters of water must be added to 250 mL of a 25% w/v stock solu- ion of sodium…
A:
Q: t 27° C the concentration of Mg2+ in a saturated solution of MgF2 is 1.17 *10^-3 molar. Is the…
A: The dissolution of an ionic compound in water results in a release in energy because the formation…
Q: A neutral solution of water at a particular temperature has a concentration of OH⁻ of 4.5 × 10⁻⁷ M.…
A:
Q: Use this information to calculate K L for BaSO4- The solubility of BaSO, in water at 25 °C is…
A: The solubility product of salt depends on the solubility of its constituent ions. It can be written…
Q: 9) What is the equivalent mass of Mn0?- in the following reaction? Mn0- + 5Fe 2+ + 8H Mn 2+ 3+ + 5Fe…
A: 9) Given unbalanced chemical reaction is - MnO42- + 5Fe2+ + 8H+ <==> Mn2+ + 5Fe3+ + 4H2O…
Q: B :Calculate [OH] for 0.5 liter solution containing (5.35) gm of NH4CI Ammonium Chloride ,Know that…
A:
Q: The value of ∆G°f for Mg+2 is -454.8 kJ/mol. The value for ∆G°f,(298) for MgCl2*6H2O is -2114.64…
A: Change in Standard Gibbs Free Energy (ΔG): It is the difference in Change in Standard Gibbs energy…
Q: Calculate the solubility of Fe (OH), in water at 25 °C. You'll find K. data in the ALEKS Data tab.…
A:
Q: -4 g The solubility of PbCrO, in water at 25 °C is measured to be 1.7 × 10 Use this information to…
A: Given: The solubility of PbCrO4 in water at 25 oC = 1.7×10-4 gL We have to calculate the Ksp for…
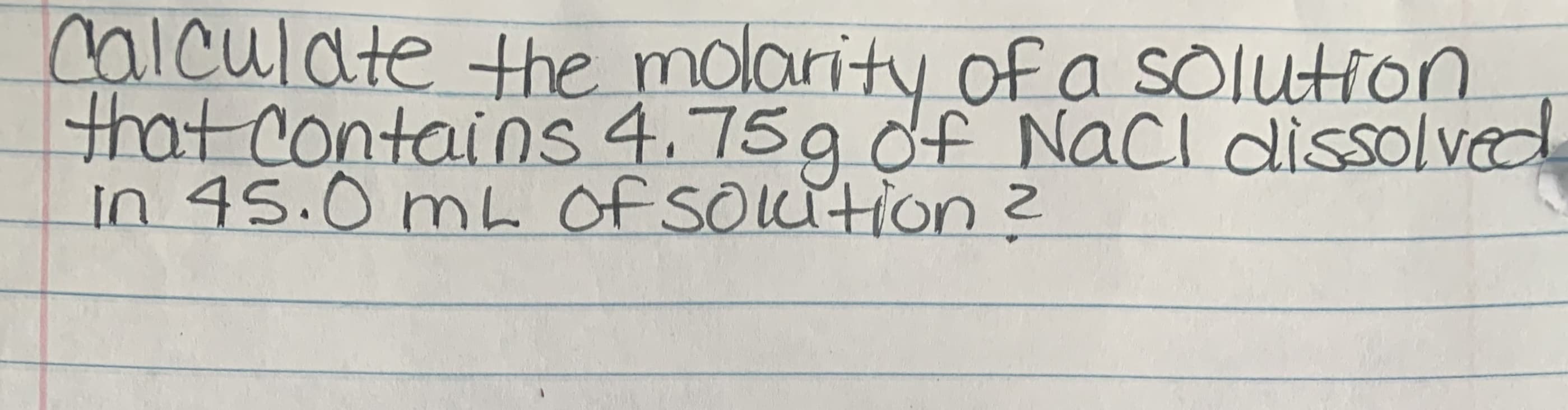
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- A solution is made by dissolving 0.455 g of PbBr2 in 100 g of H2O at 50C. Based on the data in Table 8-1, should this solution be characterized as a. saturated or unsaturated b. dilute or concentratedWhen 4.75 g of sodium hydroxide (NaOH) was dissolved in 50.00 g of water a value of 11.00oC was obtained for ΔT.Calculate the molarity of the sodium hydroxide solution.f dissolving 1.5 g of a solute into 100 mL of water caused the temperature of the solution toincrease by 4.7 ⁰C, what would the change of temperature be if 1.5 g of the solute were dissolvedin only 50 mL of water? Explain.
- Calculate for the mass of glyceraldehyde (C3H6O3) needed to prepare 250 mL of each of the following solutions?a. 78% (w/v)b. 1.65 Mc. 890 ppmHow many grams of Ag2CO3 will be soluble in 250 ml of 0.180 M AgNO3? (Ksp of Ag2CO3 = 5.95 x 10-10)The freezing point of a 0.100 M acetic acid (CH3COOH) solution is determined to be-0.1878 C. Estimate the percent dissociation of acetic acid in this solution. Is it a strong orweak electrolyte? Note that acetic acid produces acetate (CH3COO-) and hydrogen (H+) ionswhen it is dissolved in water.
- Q: 0.001 percent (v/v) ethanol solution is considered Choices: saturated supersaturated concentrated dilute1) How many grams of MnI2 are there in 244 grams of an aqueous solution that is 27.9%(w/w) MnI2? ____g 2) How many grams of iron (II) chloride,FeCl2,are required to make a 6.28%(w/v) aqueous solution in a 500.mL volumetric flask? ____gIn standardizing a solution of NaOH against 1.431g of KHC8H4O4, the analyst uses 35.50 mL of the alkali and has to run back with 5.12 mL of acid ( 1mL ≎0.0031g Na2O) What is the normality of NaOH?
- What is the molarity of a solution in which 150g of CaO are dissolved in 1900mL of solution? (Use thefollowing given information: overline MW:Ca=40g/mol , O=16g/molA. 1.41 mol/LB. 0.0014 mol/LC. 0.0014 MD. 1.41 m4 in which of the following solutions would solid AgI be expected to ve least soluble at 25c? 0.1 m sri2 0.1 m nai 0.1 m hi 0.1 m kno3Calculate Ksp value for Ce(IO3)4, solubility = 1.8x10-4 mol/L



