Calculate the concentration of silver in each sample. Are there any problems? How will you address each of the identified problems? Standards Absorbance Ag (ppb) 0.00 0.000 2.00 0.013 10.0 0.076 20.0 0.162 30.0 0.245 50.0 0.405 75.0 0.598 100.0 0.748 Samples Absorbance 08002 0.079 08003 0.058 08004 0.129 08005 0.082 08006 0.797 08007 0.063 08008 0.009 08009 0.028 08010 0.136
Calculate the concentration of silver in each sample. Are there any problems? How will you address each of the identified problems? Standards Absorbance Ag (ppb) 0.00 0.000 2.00 0.013 10.0 0.076 20.0 0.162 30.0 0.245 50.0 0.405 75.0 0.598 100.0 0.748 Samples Absorbance 08002 0.079 08003 0.058 08004 0.129 08005 0.082 08006 0.797 08007 0.063 08008 0.009 08009 0.028 08010 0.136
Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.1QAP
Related questions
Question
To each digestion flask 200 mL aliquots of 10 different drinking water samples were added. Silver was extracted by a co-precipitation procedure and collected by filtration. The precipitate was digested in 200 μl hot concentrated nitric acid and diluted to 5 mL in a dilute phosphate solution. Standards were prepared according to the following table. The samples were analyzed by graphite furnace AAS. Calculate the concentration of silver in each sample. Are there any problems? How will you address each of the identified problems?
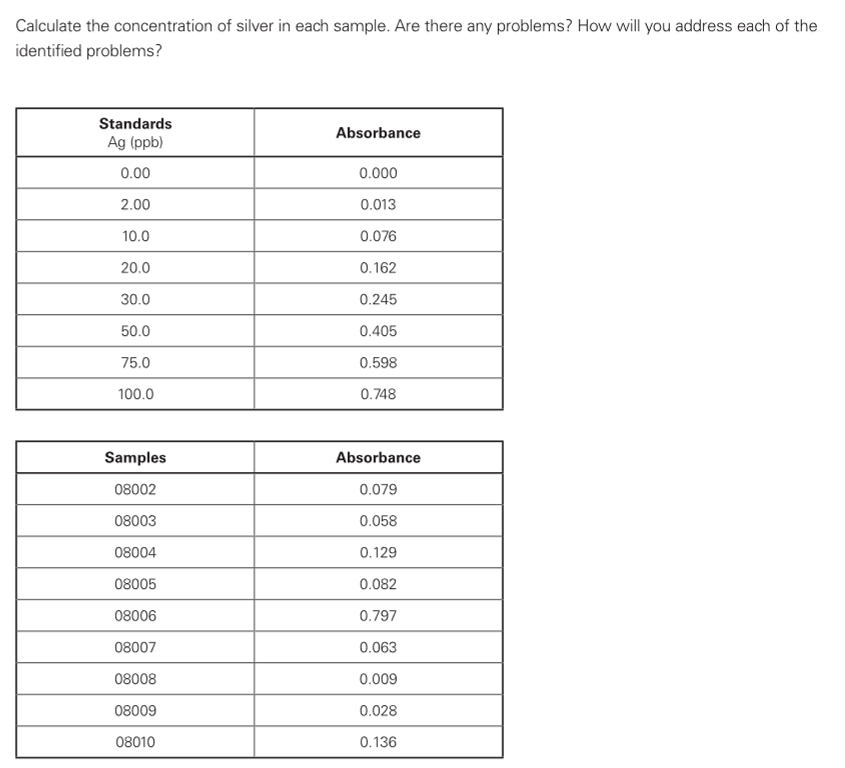
Transcribed Image Text:Calculate the concentration of silver in each sample. Are there any problems? How will you address each of the
identified problems?
Standards
Absorbance
Ag (ppb)
0.00
0.000
2.00
0.013
10.0
0.076
20.0
0.162
30.0
0.245
50.0
0.405
75.0
0.598
100.0
0.748
Samples
Absorbance
08002
0.079
08003
0.058
08004
0.129
08005
0.082
08006
0.797
08007
0.063
08008
0.009
08009
0.028
08010
0.136
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 1 steps

Recommended textbooks for you

